Press release
Human Papillomavirus Testing Market Witnesses Asia-Pacific & North America Growth | Roche, Qiagen, Hologic
The global human papillomavirus (HPV) testing market is entering a decisive phase of transformation. Once positioned primarily as a supplementary diagnostic tool alongside cytology-based screening, HPV testing is increasingly becoming the frontline strategy for cervical cancer prevention. Governments, healthcare systems, and diagnostic providers are shifting toward molecular screening models that prioritize earlier detection, broader access, and higher clinical accuracy.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=9339
For healthcare executives, diagnostics manufacturers, investors, and public health planners, the implications extend well beyond test volumes. HPV testing is evolving into a population-scale infrastructure market shaped by reimbursement models, national screening mandates, automation capacity, and digital care delivery. As countries confront rising cancer burdens and healthcare inequities, the commercial and public health value of scalable HPV diagnostics is becoming more difficult to ignore.
Quick Stats: Human Papillomavirus Testing Market at a Glance
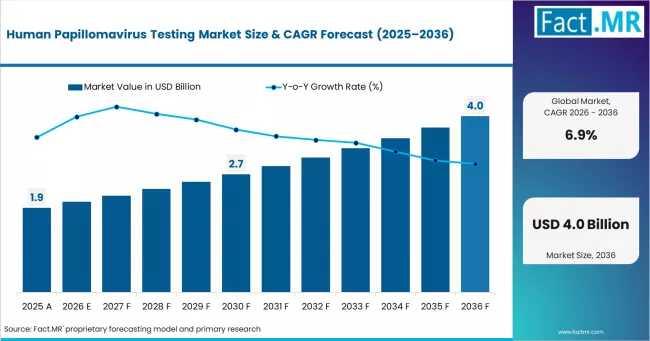
Market Value (2025): USD 1.9 Billion
Estimated Market Size (2026): USD 2.03 Billion
Forecast Market Value (2036): USD 3.96 Billion
CAGR (2026-2036): 6.9%
Absolute Dollar Opportunity: USD 1.93 Billion
Leading Technology Segment: PCR-Based Testing (65% Share in 2026)
Largest End User: Hospitals (50% Share in 2026)
Fastest-Growing Markets: India (7.8%), China (7.2%)
From Supplementary Testing to Primary Screening Infrastructure
The most important structural change in the HPV testing market is the transition from cytology-first screening toward HPV-first protocols. National cervical cancer prevention programs increasingly recognize molecular HPV testing as a more sensitive and scalable method for early detection than traditional Pap smear approaches.
This transition is fundamentally changing procurement patterns across hospitals, centralized laboratories, and public health systems. Countries adopting HPV-first screening protocols are creating predictable, recurring diagnostic demand driven by population-level testing mandates rather than episodic physician referrals.
According to Fact.MR analysis, the global HPV testing market is expected to grow from USD 2.03 billion in 2026 to approximately USD 3.96 billion by 2036, reflecting a compound annual growth rate (CAGR) of 6.9%. Over the forecast period, the market is projected to generate an absolute dollar opportunity of USD 1.93 billion.
The growth story is not purely epidemiological. It is increasingly policy-driven. Government-funded cervical cancer screening initiatives-particularly across low- and middle-income economies-are creating long-term procurement pipelines for molecular diagnostics companies.
Why Molecular Testing Is Becoming the Standard of Care
The strongest demand catalyst remains the global effort to reduce cervical cancer mortality through earlier intervention and broader screening participation. HPV infection remains the leading cause of cervical cancer, making early identification clinically and economically valuable.
Several forces are accelerating adoption:
Government-Funded Screening Programs Are Expanding
Public healthcare agencies are scaling cervical cancer prevention efforts, particularly across emerging markets where screening penetration has historically remained low. Institutional purchasing of HPV testing kits through public health tenders is creating stable demand for diagnostic manufacturers.
Healthcare systems increasingly view HPV testing not simply as a diagnostic expense, but as a preventive investment capable of reducing downstream oncology treatment costs.
Molecular Testing Is Replacing Cytology-Led Screening
The transition from cytology-first to HPV-first screening is reshaping laboratory workflows. Molecular tests provide higher sensitivity for detecting high-risk HPV strains, reducing false negatives and enabling more standardized screening.
This trend favors high-throughput automated systems capable of processing large testing volumes efficiently-particularly PCR-based diagnostic platforms.
Self-Collection Could Unlock Underserved Populations
Perhaps the most commercially significant opportunity lies in self-collected HPV testing.
Self-collection kits are emerging as a practical solution for populations facing cultural, logistical, or healthcare-access barriers that limit clinic attendance. Screening participation rates historically fall among rural populations, underserved communities, and women reluctant to undergo clinician-administered examinations.
Validated self-collection systems paired with centralized molecular analysis could dramatically expand the addressable screening population while reducing pressure on clinical infrastructure.
As Fact.MR notes, companies capable of integrating self-collection kits with laboratory workflows and digital result delivery systems are likely to secure disproportionate market share gains in the next decade.
Market Challenges: Infrastructure and Cost Still Matter
Despite strong momentum, market expansion remains uneven.
In many low-income regions, molecular testing infrastructure remains limited, forcing healthcare systems to continue relying on lower-cost but less accurate visual inspection or cytology-based approaches.
Cost sensitivity is another persistent challenge. PCR-based molecular diagnostics typically carry higher upfront costs than conventional alternatives, making procurement decisions especially complex in publicly funded healthcare systems operating under budget constraints.
Regulatory fragmentation further complicates market access. Screening recommendations, reimbursement models, and clinical protocols vary significantly across countries, forcing manufacturers to adapt commercialization strategies market by market.
For suppliers, winning share increasingly requires balancing technological sophistication with affordability, workflow simplicity, and regulatory adaptability.
Segmentation Insights: PCR Continues to Dominate
PCR Technology Leads the Market
PCR-based testing is projected to account for approximately 65% of technology share in 2026, making it the dominant testing platform.
Its leadership reflects strong clinical validation, broad institutional adoption, and compatibility with automated laboratory workflows. PCR testing also aligns closely with HPV-first screening guidelines increasingly adopted by national health agencies.
While hybrid capture and next-generation sequencing technologies continue to expand in specialized settings, PCR maintains the strongest balance between scalability, accuracy, and cost-effectiveness.
Hospitals Remain the Largest End User
Hospitals are expected to account for roughly 50% of end-user demand in 2026.
Their dominance stems from centralized procurement capabilities, diagnostic infrastructure, and integration with oncology, pathology, and women's health programs. However, diagnostic laboratories are emerging as increasingly influential stakeholders as testing volumes shift toward centralized processing models.
Cervical Cancer Screening Drives Core Demand
Cervical cancer screening remains the market's anchor application, representing approximately 35.8% of demand in 2026.
The Next Big Shift: Radiology Meets Diagnostics Through Precision Screening
A major trend shaping the future market is integration between molecular diagnostics, digital pathology, and AI-assisted interpretation.
Diagnostic providers are increasingly investing in platforms that combine automated HPV detection with standardized result interpretation and digital patient tracking systems. Such capabilities improve throughput, reduce interpretation variability, and support national-scale screening efficiency.
The broader move toward precision medicine also opens opportunities for biomarker-linked testing pathways and risk-based patient stratification.
In practical terms, the future of HPV testing may be less about selling standalone diagnostic kits and more about delivering integrated screening ecosystems.
Asia Pacific Emerges as the Growth Engine
While North America continues to lead global demand due to strong institutional purchasing and established screening frameworks, Asia Pacific is becoming the fastest-growing regional market.
India: A Fast-Rising Opportunity
India is projected to record the strongest growth at 7.8% CAGR through 2036.
Expansion of healthcare infrastructure, increasing awareness of cervical cancer prevention, and public-sector screening initiatives are improving market fundamentals. The country also presents strong opportunities for scalable, cost-efficient testing platforms capable of serving rural and underserved populations.
China: Policy Support Accelerates Adoption
China follows closely with projected growth of 7.2%.
Government healthcare modernization programs, stronger clinical adoption, and growing domestic diagnostics capabilities are contributing to rising testing volumes. Urban-rural healthcare integration initiatives are expected to further expand demand.
Mature Markets Continue to Upgrade
Markets such as the United States, Germany, and France remain steady growth centers.
In these regions, demand is increasingly tied to protocol upgrades, replacement cycles, automation investments, and integration of advanced molecular screening workflows.
Competition Is Shifting Toward Workflow Ownership
The competitive landscape remains moderately fragmented, but leadership increasingly depends on platform integration rather than standalone product performance.
Major players such as Roche Diagnostics AG, Hologic Inc., and QIAGEN N.V. maintain strong positions through broad portfolios, established clinical credibility, and global distribution reach.
Additional competition comes from Abbott Laboratories, Cepheid Inc., and Thermo Fisher Scientific Inc., alongside regional and niche diagnostic specialists.
What increasingly differentiates market leaders is the ability to combine testing hardware, validated assays, automation, service support, digital integration, and regulatory readiness into cohesive solutions.
In other words, buyers are increasingly selecting workflow ecosystems-not just diagnostic kits.
Strategic Implications for Industry Stakeholders
For diagnostic manufacturers, the next decade will reward companies that think beyond instrumentation and reagent sales.
Three priorities stand out:
Invest in self-collection infrastructure: Expanding access beyond hospitals and clinics could unlock substantial incremental testing populations.
Build scalable automation capabilities: High-throughput molecular systems are increasingly essential for public screening programs.
Strengthen digital and AI-enabled workflows: Faster interpretation, centralized reporting, and patient engagement tools improve program efficiency and customer retention.
Healthcare systems, meanwhile, face a parallel challenge: balancing screening expansion with affordability and infrastructure readiness.
Markets able to combine HPV-first screening, digital pathology, and accessible testing formats are likely to see the strongest gains in participation and early diagnosis.
Future Outlook: From Diagnostics Market to Prevention Infrastructure
The human papillomavirus testing market is steadily evolving from a diagnostic niche into a foundational component of population-scale cancer prevention.
The next phase of growth will not be driven solely by disease incidence. Instead, it will be shaped by public policy, screening accessibility, molecular automation, and participation rates.
By 2036, the winners in this market are unlikely to be companies selling only tests. They will be organizations enabling complete prevention ecosystems-combining diagnostics, digital workflows, self-collection access, and scalable screening infrastructure.
Browse Full Report - https://www.factmr.com/report/human-papillomavirus-testing-market
Executive-Level Takeaways
The HPV testing market is forecast to grow from USD 2.03 billion in 2026 to USD 3.96 billion by 2036, creating USD 1.93 billion in incremental opportunity.
PCR-based molecular diagnostics will remain the dominant technology, supported by HPV-first screening protocols.
Self-collection testing represents the most disruptive growth opportunity, particularly in underserved populations.
India and China are emerging as high-growth markets driven by healthcare expansion and public screening investments.
Competitive advantage is increasingly tied to workflow integration, automation, and digital screening ecosystems, rather than standalone testing capability.
Full Report: Unlock 360° insights for strategic decision making and investment planning-
https://www.factmr.com/checkout/9339
To View Related Report:
Human Platelet Lysate Market https://www.factmr.com/report/human-platelet-lysate-market
Human Growth Hormone (HGH) Market https://www.factmr.com/report/1389/human-growth-hormone-market
Humanized Mice Model Market https://www.factmr.com/report/humanized-mice-model-market
Human Umbilical Vein Endothelial Cells (HUVEC) Market https://www.factmr.com/report/human-umbilical-vein-endothelial-cells-huvec-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Human Papillomavirus Testing Market Witnesses Asia-Pacific & North America Growth | Roche, Qiagen, Hologic here
News-ID: 4509676 • Views: …
More Releases from Fact MR
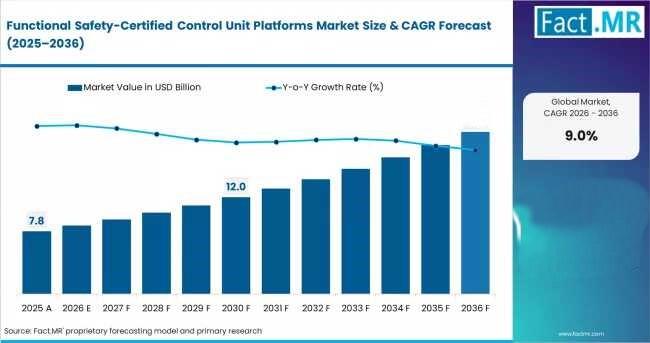
Functional Safety Certified Control Unit Platforms Market Expands Across Europe …
The global market for functional safety-certified control unit platforms is entering a new phase of industrial relevance. Once viewed primarily as a niche requirement for highly regulated environments, these platforms are increasingly becoming foundational infrastructure across automotive electronics, industrial automation, energy systems, and intelligent machinery. As electrification, software-defined systems, and autonomous functionality move deeper into mainstream manufacturing, organizations are prioritizing platforms that simplify compliance, reduce certification risk, and accelerate deployment…
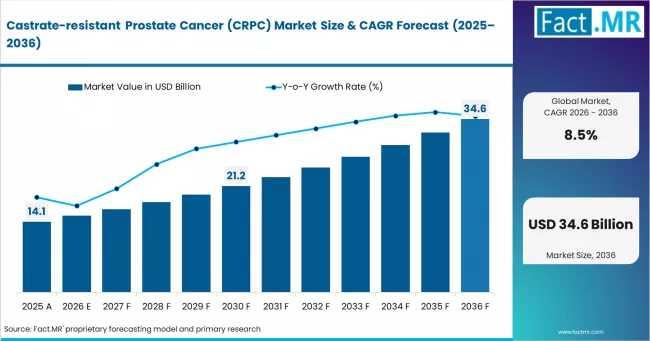
Castrate-Resistant Prostate Cancer (CRPC) Market Leads in North America & Europe …
The global market for castrate-resistant prostate cancer (CRPC) therapies is entering a new phase-one shaped less by broad-spectrum oncology and more by precision medicine, targeted radiopharmaceuticals, and biomarker-led treatment sequencing. For pharmaceutical companies, oncology providers, investors, and healthcare procurement leaders, the commercial conversation around CRPC is no longer centered solely on patient volume growth. Increasingly, it revolves around treatment complexity, companion diagnostics, and the growing role of personalized interventions in…
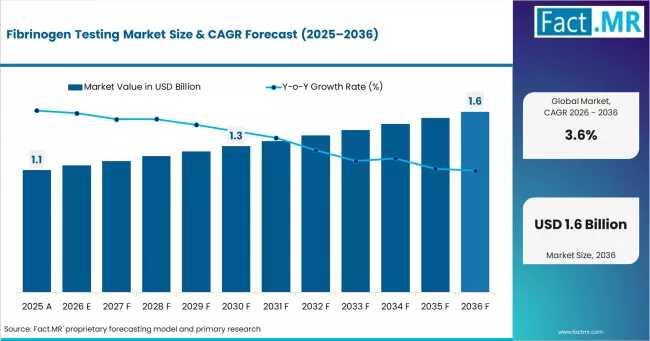
Fibrinogen Testing Market Sees Strong Regional Dominance in Europe & North Ameri …
As healthcare systems intensify their focus on surgical precision, trauma care, and bleeding-risk management, fibrinogen testing is quietly becoming a more important component of clinical decision-making. Once viewed primarily as a routine coagulation laboratory function, fibrinogen measurement is increasingly being integrated into perioperative medicine, emergency care, and transfusion protocols. According to Fact.MR analysis, the global fibrinogen testing market is transitioning from a stable diagnostic category into a strategically important support…
Ocular Implants Market Sees North America & Europe Regional Dominance | Alcon, B …
The global ocular implants market is entering a more technologically differentiated phase, moving beyond its historical dependence on cataract surgery volumes toward premium intraocular lenses (IOLs), minimally invasive glaucoma devices, and advanced retinal implants. As ophthalmology shifts toward personalized visual outcomes and lower-burden disease management, implant makers are increasingly competing on precision, patient experience, and long-term treatment efficiency rather than procedural volume alone.
Get detailed market forecasts, competitive benchmarking, and pricing…
More Releases for HPV
Human Papillomavirus (HPV) Vaccine: Core Growth Enabler in the Rising Prevalence …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
What Will the Human Papillomavirus (HPV) Vaccine Industry Market Size Be by 2025?
The market size for the Human Papillomavirus (HPV) vaccine has seen a swift expansion in the past few years. The market is projected to inflate from $5.8 billion in 2024 to $6.57 billion in 2025, marking…
Global Human Papillomavirus (HPV) Vaccine Market Report 2025: Insights, Drivers …
The Global Human Papillomavirus (HPV) Vaccine Market size reached US$ 3,043.19 Million in 2024 and is expected to reach US$ 12,361.15 Million by 2033, growing at a CAGR of 16.9% during the forecast period 2025-2033.
The Human Papillomavirus (HPV) Vaccines Market Report by DataM Intelligence offers comprehensive insights into the latest market trends, key growth drivers, and emerging challenges. Crafted to support smarter and faster decision-making, our reports blend in-depth data…
Rising Prevalence Of HPV-Related Diseases Sparks Surge In Human Papillomavirus ( …
The Human Papillomavirus (HPV) Vaccine Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Expected Human Papillomavirus (HPV) Vaccine Market Size During the Forecast Period?
The market size for the human papillomavirus (HPV) vaccine has seen swift growth in the past few years. The…
HPV Vaccines Market 2020-2026 - COVID19 Impact On HPV Vaccines Industry | Top Ke …
HPV Vaccines Market Size, Share & Industry Analysis, By Type (Bivalent and Polyvalent), By Disease Indication (HPV Associated Cancer and Genital Warts), By Distribution Channel (Hospital & Retail Pharmacies, Government Suppliers, and Others) and Geography Forecast, 2019-2026.
Get Sample Copy Of Report To Know The Impact of Covid19 on this Industry: https://www.fortunebusinessinsights.com/enquiry/request-sample-pdf/human-papillomavirus-hpv-vaccines-market-101962
Top Key Players: GSK, Merck.
An Overview of the Impact of COVID-19 on HPV Vaccines Market:
The emergence of COVID-19 has brought…
HPV Testing & PAP Test Market by Test Type (HPV Testing, Follow-up HPV Testing, …
Global HPV Testing & PAP Test Market was valued at USD XX billion in the year 2017. Global HPV Testing & PAP Test market is further estimated to grow at a CAGR of XX % from 2018 to reach USD XX billion by the year 2023.
Get Free Access to Sample Report @ https://genesismarketinsights.com/Sample_Request/GENRE385
In the Global HPV Testing & PAP Test Market, by region, North America holds the highest market share…
Cytology and HPV Testing Market: Disparate Levels of Awareness among Patients Re …
Over the years, the level of awareness among the global population regarding the various types of cancer has increased tremendously. The significance of early detection and diagnosis is not lost on them, resulting in the increased adoption of screening tests such as cytology and human papillomavirus (HPV) testing.
The global market for cytology and HPV testing is presented with a host of sustained growth opportunities arising mainly from several emerging economies,…