Press release
Virtual Clinical Trials Market to Reach USD 11.7 Billion by 2035 as Interventional Studies Capture 59% Share, North America Accounts for 45%-50% of Global Demand, and IQVIA, Medidata & ICON Accelerate Decentralized Clinical Research Infrastructure
Wilmington, DE, USA, 11th May 2026 - According to MarketGenics Global Research, the global virtual clinical trials market is valued at USD 6.6 billion in 2025 and is projected to reach USD 11.7 billion by 2035, expanding at a CAGR of 5.9% during the forecast period (2025-2035).The market is witnessing significant expansion due to rising adoption of decentralized and hybrid clinical trial models, increasing integration of AI-powered remote patient monitoring systems, growing use of wearable health monitoring devices, expansion of telemedicine and electronic health record infrastructure, and increasing demand for patient-centric clinical research methodologies.
Get the Detailed Industry Analysis (including the Table of Contents, List of Figures, and List of Tables): https://marketgenics.co/reports/virtual-clinical-trials-market-64545
==============================
MARKET OVERVIEW - KEY STATISTICS
==============================
Drivers - Rising Adoption of Decentralized and Hybrid Trial Models Accelerating Market Growth
The virtual clinical trials market is experiencing accelerated growth because pharmaceutical companies and clinical research organizations are increasingly deploying decentralized and hybrid trial designs to improve patient recruitment efficiency, expand geographic accessibility, reduce operational complexity, and accelerate clinical development timelines.
Key Fact
According to the Tufts Center for the Study of Drug Development, nearly 80% of clinical trials experience delays due to patient recruitment challenges, increasing adoption of decentralized clinical trial approaches.
Challenges - Data Privacy Concerns and Regulatory Complexity
The deployment of decentralized clinical trial systems continues facing barriers associated with patient data privacy concerns, cross-border regulatory complexity, integration challenges between digital trial platforms and existing clinical infrastructure, and compliance requirements surrounding remote patient data management.
Regulatory Landscape
The virtual clinical trials market is governed by frameworks established by the U.S. Food and Drug Administration, European Medicines Agency, GDPR regulations, and regional healthcare authorities emphasizing patient safety, digital consent management, data privacy, and compliance for decentralized and hybrid clinical studies.
==============================
EMERGING MARKET OPPORTUNITIES
==============================
Global Market Opportunity
~USD 5.1 Billion Total Forecast Opportunity by 2035
The global virtual clinical trials market is expected to create an incremental opportunity of nearly USD 5.1 billion by 2035, supported by expansion of decentralized trial platforms, wearable monitoring technologies, AI-powered recruitment analytics, telehealth-enabled clinical engagement systems, and cloud-based remote patient data management infrastructure.
Regional Opportunity Analysis
~North America Remains the Most Attractive Regional Opportunity During 2025-2035
North America continues dominating the global virtual clinical trials market due to advanced digital health infrastructure, strong pharmaceutical and biotechnology ecosystem presence, widespread adoption of telehealth and electronic health record systems, and increasing investments in decentralized clinical research technologies.
Get Sample Copy of the Report: https://marketgenics.co/download-report-sample/virtual-clinical-trials-market-64545
==============================
KEY FINDINGS OF THE VIRTUAL CLINICAL TRIALS MARKET REPORT
==============================
• Total forecast market opportunity expected to exceed USD 5.1 billion by 2035
• Interventional studies segment accounts for approximately 59% of the global virtual clinical trials market share in 2025
• North America contributes nearly 45%-50% of total global market demand
• Top five companies account for more than 50% of total market share globally
• AI-powered patient recruitment and wearable-enabled monitoring systems are transforming decentralized clinical research strategies
• Telemedicine integration, cloud-based trial management platforms, remote patient monitoring systems, and real-world evidence analytics remain high-growth implementation areas
==============================
ANALYST VIEWPOINT
==============================
The global virtual clinical trials industry is transitioning beyond traditional site-based research models toward digitally connected, patient-centric, decentralized clinical ecosystems optimized for remote participation, continuous patient monitoring, AI-driven recruitment analytics, real-time clinical data capture, and cloud-native trial management infrastructure.
Competitive positioning within the industry is increasingly defined by:
• decentralized trial platform integration capability
• AI-driven patient recruitment expertise
• wearable and remote monitoring infrastructure
• cloud-native clinical data management capability
• telehealth and virtual visit ecosystem integration
• regulatory compliance and patient privacy specialization
==============================
SEGMENTAL INSIGHTS & GROWTH ANCHORS
==============================
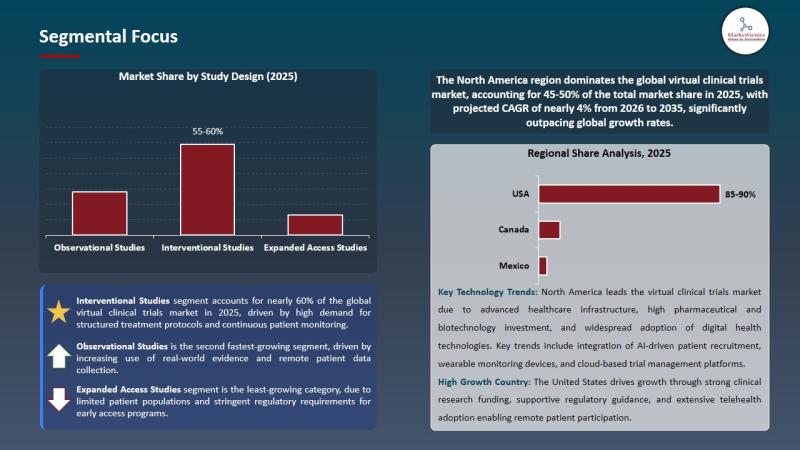
Market Share by Study Design (2025)
• Interventional Studies - ~59%
• Observational Studies
• Expanded Access Studies
The interventional studies segment dominates the global virtual clinical trials market due to increasing deployment of decentralized clinical trial technologies, structured treatment monitoring requirements, expansion of remote patient engagement systems, and growing adoption of telehealth-supported drug and biologics studies.
North America dominates the global virtual clinical trials market, accounting for approximately 45%-50% of total market share in 2025, supported by strong pharmaceutical R&D infrastructure, widespread digital health adoption, increasing telehealth penetration, and favorable regulatory support for decentralized clinical research methodologies.
Regional Share Analysis, 2025
• United States
• Canada
• Mexico
Key Technology Trends
North America continues leading the virtual clinical trials ecosystem due to increasing adoption of AI-powered patient recruitment platforms, wearable health monitoring technologies, cloud-based clinical management systems, and decentralized digital research infrastructure supporting remote clinical participation.
==============================
COMPETITIVE LANDSCAPE & KEY PLAYERS
==============================
Global Company Market Share (2025)
• IQVIA Holdings Inc.
• Oracle Corporation (Medidata Solutions)
• ICON plc
• Parexel International Corporation
• Veeva Systems Inc.
• Other Key Players
The global virtual clinical trials market remains highly consolidated with the top five manufacturers controlling more than 50% of total market share in 2025. IQVIA Holdings Inc. maintains strong leadership through its integrated decentralized clinical trial platforms, AI-powered analytics capabilities, end-to-end patient recruitment infrastructure, and scalable remote monitoring systems supporting global clinical research operations.
IQVIA Holdings Inc.
• Company Revenue: USD 15.4 Bn
• Headquarters: United States
• In 2025, IQVIA Holdings Inc. launched its IQVIA Nexus Decentralized Trial Suite featuring integrated remote patient monitoring, AI-driven recruitment analytics, and real-time clinical data capture capabilities, enabling pharmaceutical sponsors to streamline decentralized trial operations and improve patient engagement.
Oracle Corporation (Medidata Solutions)
• Company Revenue: USD 53 Bn
• Headquarters: United States
• Oracle Corporation (Medidata Solutions) partnered with Palantir Technologies to integrate advanced data analytics and real-time monitoring into the Medidata Clinical Cloud platform, combining scalable cloud infrastructure with AI-driven insights to optimize decentralized patient recruitment and remote clinical trial management.
Other Key Companies Operating in the Virtual Clinical Trials Market Include:
• Castor Research Inc.
• Clario (ERT Clinical)
• Clinical Ink, Inc.
• Curebase, Inc.
• CRF Health, Inc.
• Dassault Systèmes SE (Medidata Solutions)
• Veeva Systems Inc.
• ICON plc
• IQVIA Holdings Inc.
• Labcorp Holdings Inc.
• Lightship, Inc.
• Medable, Inc.
• ObvioHealth, Inc.
• Oracle Corporation
• Syneos Health, Inc.
• Parexel International Corporation
• THREAD Research, Inc.
• Science 37, Inc.
• Signant Health Holding Corp.
• Thermo Fisher Scientific Inc. (PPD, Inc.)
• Other Key Players
==============================
RECENT DEVELOPMENTS & INDUSTRY NEWS
==============================
• In August 2025, Veeva established a strategic partnership with Amgen to utilize the Veeva Clinical Platform for developing advanced hybrid and decentralized clinical trial methodologies and improving operational efficiency across clinical research workflows.
• In March 2024, Medidata Solutions launched new decentralized trial capabilities for its Medidata Platform integrating wearable device data, remote consent management, and virtual patient visit functionality for decentralized research studies.
• In January 2024, Medable partnered with Google Cloud to strengthen decentralized clinical trial functionality through cloud-based analytics and artificial intelligence systems supporting remote patient data management.
• In October 2023, Science 37 and Medidata Solutions collaborated to integrate decentralized trial technologies with traditional clinical research systems enabling remote patient visits and continuous digital monitoring capabilities.
The industry is rapidly evolving toward AI-enabled decentralized research ecosystems, wearable-integrated patient monitoring systems, cloud-native trial management infrastructure, telehealth-supported clinical engagement models, and real-world evidence analytics platforms supporting next-generation patient-centric clinical research.
==============================
VIRTUAL CLINICAL TRIALS MARKET SEGMENTATION
==============================
By Trial Type
• Fully Decentralized Trials
• Hybrid Trials
• Site-Based Trials with Virtual Components
By Study Design
• Interventional Studies
• Observational Studies
• Expanded Access Studies
By Phase
• Phase I
• Phase II
• Phase III
• Phase IV
By Component
• Software Platforms
• Clinical Trial Management Systems (CTMS)
• Electronic Data Capture (EDC) Systems
• Electronic Clinical Outcome Assessment (eCOA) Platforms
• Electronic Patient-Reported Outcome (ePRO) Systems
• eConsent Platforms
• Telemedicine and Virtual Visit Platforms
• Remote Patient Monitoring (RPM) Software
• Patient Recruitment and Engagement Platforms
• Randomization and Trial Supply Management (RTSM) Systems
• Clinical Data Management Systems (CDMS)
• Digital Biomarker Analytics Platforms
• AI-Based Clinical Trial Analytics Platforms
• Data Integration and Interoperability Platforms
• Regulatory Compliance and Documentation Software
• Others
By Services
• Decentralized Clinical Trial (DCT) Design and Consulting
• Virtual Clinical Trial Implementation Services
• Patient Recruitment and Retention Services
• Remote Monitoring and Site Management Services
• Telehealth and Virtual Visit Support Services
• Home Healthcare and Mobile Nursing Services
• Data Management and Biostatistics Services
• Regulatory Affairs and Compliance Services
• Logistics and Drug Supply Management
• Technology Integration and Platform Support
• Training and Technical Support Services
• Others
By Hardware and Connected Devices
• Wearable Health Monitoring Devices
• Biosensors and Digital Biomarker Devices
• Smart Medical Devices (Connected Devices)
• Remote Diagnostic Devices
• Mobile Devices (Smartphones and Tablets)
• Patient Monitoring Equipment
• Home-Based Clinical Trial Kits
• Smart Medication Adherence Devices
• Telehealth Communication Devices
• Portable Diagnostic and Imaging Devices
• Others
By Technology
• Telemedicine Platforms
• Electronic Data Capture (EDC) Systems
• eConsent Platforms
• Mobile Health (mHealth) Applications
• Wearable Devices and Biosensors
• Remote Patient Monitoring Platforms
• Clinical Trial Management Systems (CTMS)
• eClinical Platforms
• Others
By Therapeutic Area
• Oncology
• Cardiovascular Diseases
• Central Nervous System (CNS) Disorders
• Infectious Diseases
• Metabolic and Endocrine Disorders
• Respiratory Diseases
• Immunology and Autoimmune Diseases
• Rare Diseases
• Others
By Patient Engagement Model
• Direct-to-Patient Trials
• Home Healthcare Supported Trials
• Hybrid Patient Engagement Models
By Data Collection Method
• Electronic Patient-Reported Outcomes (ePRO)
• Sensor and Wearable-Based Monitoring
• Mobile App-Based Data Collection
• Teleconsultation and Virtual Visits
• Others
By End User
• Pharmaceutical Companies
• Biotechnology Companies
• Contract Research Organizations (CROs)
• Medical Device Companies
• Academic and Research Institutes
• Others
==============================
FUTURE OUTLOOK & STRATEGIC PERSPECTIVE
==============================
The global virtual clinical trials market is positioned for substantial long-term growth as pharmaceutical companies, biotechnology firms, contract research organizations, and digital health providers continue accelerating investments in decentralized trial technologies, AI-powered patient recruitment systems, wearable monitoring platforms, telehealth-enabled clinical engagement infrastructure, and cloud-native clinical data ecosystems.
North America is expected to remain the dominant regional growth engine due to strong digital healthcare infrastructure, high pharmaceutical R&D investments, supportive regulatory frameworks, and increasing adoption of decentralized patient-centric clinical research methodologies.
BUY NOW: https://marketgenics.co/buy/virtual-clinical-trials-market-64545
Contact:
Mr. Debashish Roy
MarketGenics Global Research
800 N King Street, Suite 304 #4208, Wilmington, DE 19801, United States
USA: +1 (302) 303-2617
Email: sales@marketgenics.co
Website: https://marketgenics.co
About MarketGenics
MarketGenics is a global market research and business advisory firm empowering decision-makers across startups, Fortune 500 companies, non-profit organizations, universities, and government institutions. The company delivers comprehensive market intelligence, industry analysis, and strategic insights across diverse sectors.
MarketGenics publishes detailed industry research reports combining granular quantitative analysis with expert insights on market trends, competitive landscapes, and emerging opportunities. These reports help organizations make informed strategic decisions, identify growth opportunities, and support sustainable business development.
In addition to research publications, MarketGenics supports organizations with strategic insights on product development, application modeling, market expansion strategies, and identifying niche growth opportunities.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Virtual Clinical Trials Market to Reach USD 11.7 Billion by 2035 as Interventional Studies Capture 59% Share, North America Accounts for 45%-50% of Global Demand, and IQVIA, Medidata & ICON Accelerate Decentralized Clinical Research Infrastructure here
News-ID: 4508553 • Views: …
More Releases from MarketGenics Global Research
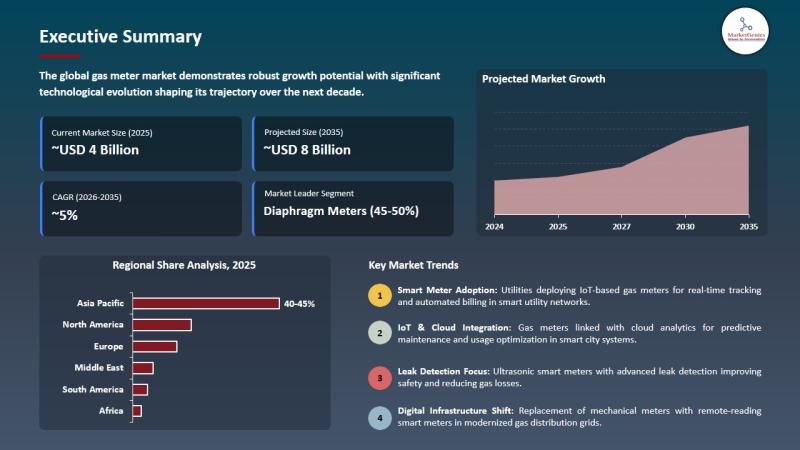
Gas Meter Market to Reach USD 8 Billion by 2035 as Diaphragm Meters Hold 46% Sha …
Wilmington, DE, USA, 11th May 2026 - According to MarketGenics Global Research, the global gas meter market is valued at USD 4.3 billion in 2025 and is projected to reach USD 7.9 billion by 2035, expanding at a CAGR of 5.3% during the forecast period (2026-2035).
The market is witnessing accelerated growth due to rapid deployment of smart gas metering infrastructure, increasing government-backed utility modernization programs, rising adoption of IoT-enabled metering…
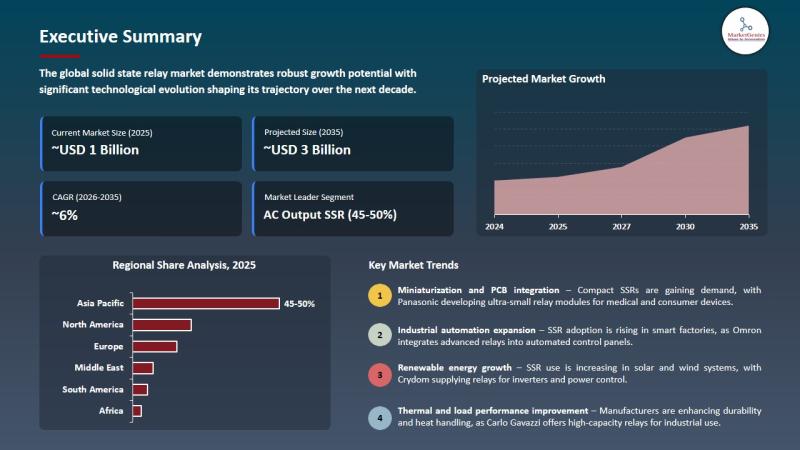
Solid State Relay Market to Reach USD 2.5 Billion by 2035, Driven by Industrial …
Wilmington, DE, USA, 11th May 2026 - According to MarketGenics, the global solid state relay market is valued at USD 1.4 billion in 2025 and is projected to reach approximately USD 2.5 billion by 2035, expanding at a CAGR of 6.1% during the forecast period. Growth is primarily driven by rising industrial automation, Industry 4.0 adoption, electric vehicle expansion, renewable energy infrastructure, and increasing demand for energy-efficient, maintenance-free switching solutions…

Heat Pump Compressors Market to Reach USD 38 Billion by 2035 as Scroll Compresso …
Wilmington, DE, USA, 11th May 2026 - According to MarketGenics Global Research, the global heat pump compressors market is valued at USD 15 billion in 2025 and is projected to reach USD 38 billion by 2035, expanding at a CAGR of 9.7% during the forecast period (2026-2035).
The market is witnessing strong expansion due to increasing electrification of heating systems, growing adoption of inverter-driven compressor technologies, rising demand for energy-efficient HVAC…
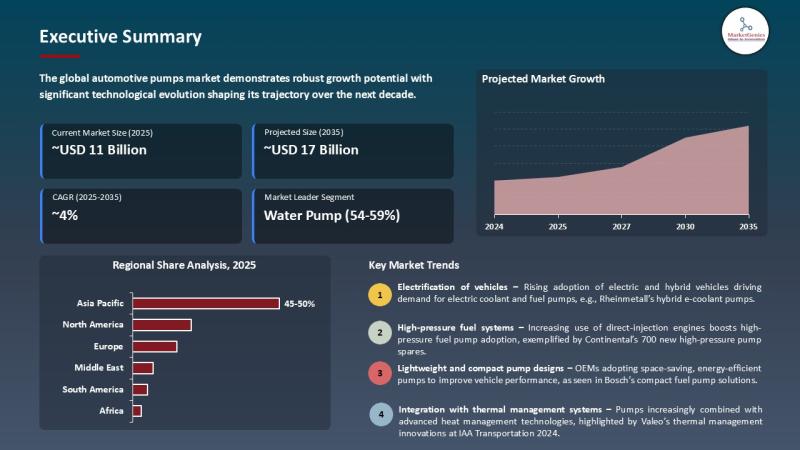
Automotive Pumps Market to Reach USD 17.1 Billion by 2035 Driven by EV Thermal M …
Wilmington, DE, USA, 11th May 2026 - According to MarketGenics, the global automotive pumps market is valued at USD 11.3 billion in 2025 and is projected to reach approximately USD 17.1 billion by 2035, expanding at a CAGR of 4.2% during the forecast period. Growth is primarily driven by increasing demand for fuel-efficient and emission-compliant vehicles, rapid electrification of automotive systems, and rising integration of electric pumps in hybrid and…
More Releases for Trial
Clinical Trial Investigative Site Network Market Clinical Trial Investigative Si …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trial Investigative Site Network Market - (By Therapeutic Areas (Oncology, Cardiology, CNS, Pain Management, Endocrine, Others), By Phase (Phase I, Phase II, Phase III, Phase IV), By End-use (Sponsor, CRO)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic, the Global Clinical Trial Investigative Site Network Market…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Clinical Trial Management System
According to a new market report published by Persistence Market Research “Global Market Study on Clinical Trial Management System: Asia to Witness Highest Growth by 2019” the global clinical trial management system market was valued at USD 844.0 million in 2013 and is expected to grow at a CAGR of 14% from 2014 to 2019, to reach an estimated value of USD 1,848.5 million in 2019.
Request Report TOC @ https://www.persistencemarketresearch.com/methodology/3017
…
Clinical Trial Logistics
Clinical Trial Logistics
16th to 17th May 2011, Marriott Regents Park, London, United Kingdom.
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical trials…
Clinical Trial Logistics
Announcing SMi's 5th annual…
Clinical Trial Logistics conference
16th and 17th May 2011, Central London, UK
www.smi-online.co.uk/2011logistics-london6.asp
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical…