Press release
Immunoassay Interference Blocker Market Sees North America & Asia-Pacific Regional Dominance | Bio-Rad, Thermo Fisher, Merck
The global immunoassay interference blocker market is entering a new phase of relevance within the broader in-vitro diagnostics ecosystem, driven less by headline-grabbing innovation and more by an increasingly urgent need for assay reliability. As healthcare systems expand immunoassay testing across clinical diagnostics, pharmaceutical development, and blood screening, laboratories are facing growing pressure to reduce false-positive and false-negative outcomes linked to assay interference.Get detailed market forecasts, competitive benchmarking, and pricing trends:
https://www.factmr.com/connectus/sample?flag=S&rep_id=9395
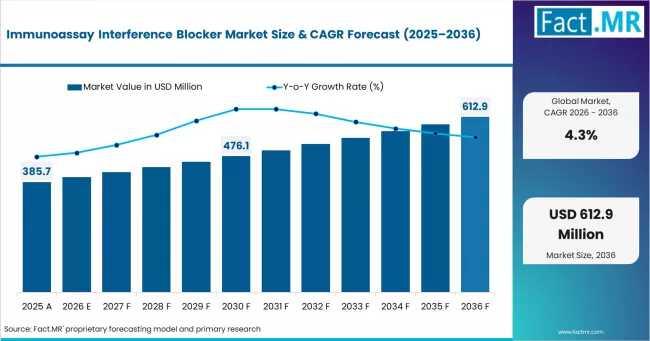
What was once considered a niche quality-control category is now becoming a structural requirement in modern laboratory workflows. According to Fact.MR analysis, the market is projected to grow from approximately USD 402.3 million in 2026 to USD 612.9 million by 2036, reflecting a steady CAGR of 4.3%. Growth is being reinforced by tightening regulatory oversight, rising awareness of heterophilic antibody and biotin-related interference, and expanding diagnostic infrastructure across emerging economies.
Quick Stats: Immunoassay Interference Blocker Market
Market Value (2025): USD 385.7 Million
Estimated Market Value (2026): USD 402.3 Million
Forecast Market Value (2036): USD 612.9 Million
CAGR (2026-2036): 4.3%
Absolute Dollar Opportunity: USD 210.6 Million
Leading Product Segment: Heterophile Antibody Blockers
Segment Share: 32.8%
Leading Product Form: Liquid Reagents
Segment Share: 36.7%
Fastest-Growing Markets: India and China
Key Companies: Bio-Rad Laboratories, Thermo Fisher Scientific, Merck KGaA (Sigma-Aldrich), Abcam plc, Agilent Technologies
Why Immunoassay Interference Has Become a Boardroom-Level Laboratory Issue
The commercial significance of interference blockers stems from a simple but consequential reality: diagnostic errors carry escalating financial, legal, and clinical risks.
Modern immunoassays are widely used for thyroid testing, cardiac biomarker analysis, infectious disease diagnostics, oncology screening, fertility assessment, and hormone analysis. Yet these assays remain vulnerable to interference from heterophilic antibodies, rheumatoid factors, biotin supplementation, autoantibodies, and other endogenous substances that can distort analytical results.
For hospitals and laboratory networks operating under tighter accreditation requirements, interference mitigation is increasingly shifting from optional best practice to mandatory quality assurance protocol.
This shift is particularly visible in high-volume testing environments where even a small percentage of erroneous results can create significant downstream costs through repeat testing, unnecessary treatment pathways, reimbursement disputes, or litigation exposure.
The growing integration of interference blocker reagents into routine workflows reflects a broader transformation in diagnostics: laboratories are moving from reactive troubleshooting toward preventive assay validation and systematic interference management.
Market Size and Long-Term Growth Outlook
The immunoassay interference blocker market remains relatively specialized in absolute value terms, but its strategic role within laboratory medicine is expanding steadily.
Fact.MR projects the market to increase from USD 385.7 million in 2025 to USD 612.9 million by 2036. While the projected 4.3% CAGR is moderate compared to high-growth diagnostics categories, the market benefits from unusually stable demand characteristics tied to regulatory compliance and essential laboratory operations.
Unlike discretionary laboratory upgrades, interference mitigation products are increasingly embedded within validation protocols, accreditation standards, and manufacturer quality requirements. That creates a more resilient procurement cycle, even during broader healthcare spending slowdowns.
Importantly, market growth is being fueled not only by higher testing volumes, but also by rising assay complexity. As immunoassays become more sensitive and multiplexed, the probability and clinical impact of interference-related anomalies increases correspondingly.
Heterophile Antibody Blockers Continue to Dominate Procurement Demand
Among product categories, heterophile antibody blockers are expected to account for 32.8% of the global market in 2026, maintaining leadership due to the persistent prevalence of heterophilic antibody interference across clinical immunoassays.
These blockers are widely integrated into sandwich immunoassay systems, where nonspecific antibody binding can generate misleading results with direct implications for patient diagnosis and treatment decisions.
The segment benefits from several structural advantages:
Established laboratory familiarity
Broad compatibility across assay platforms
Regulatory recognition of interference risks
Integration into standard validation workflows
Consistent demand across hospital and reference laboratories
The market's second major anchor is liquid reagent formulations, which are forecast to represent 36.7% of product form demand in 2026. Laboratories continue to favor liquid formats because they align more easily with automated workflows, reduce preparation variability, and simplify integration into high-throughput testing systems.
Biotin Interference Emerges as the Fastest-Growing Product Opportunity
One of the most important trends reshaping the market is the rapid emergence of biotin interference mitigation products.
The widespread consumer adoption of high-dose biotin supplements for hair, skin, and nail health has unintentionally created a major diagnostic challenge for streptavidin-biotin based immunoassays. Excess circulating biotin can distort results in thyroid function tests, cardiac biomarker assays, and hormone panels.
This issue has gained regulatory visibility in recent years, with health authorities issuing guidance regarding the clinical risks associated with biotin-related assay interference.
As a result, diagnostic laboratories are increasingly adopting:
Biotin-specific blocking reagents
Sample pre-treatment systems
Modified assay workflows
Interference-screening protocols
Redesigned assay platforms with reduced biotin sensitivity
The commercial opportunity here is particularly attractive because biotin interference mitigation often commands premium pricing relative to conventional blocking reagents, especially when integrated into automated laboratory systems.
Emerging Markets Are Reshaping the Global Demand Landscape
Although North America and Western Europe remain established markets, future expansion is increasingly concentrated in Asia Pacific and other emerging healthcare economies.
India is projected to lead global growth with a CAGR of 6.2% through 2036, supported by:
Expansion of diagnostic laboratory infrastructure
Rising immunoassay testing volumes
Tightening accreditation standards
Growth in private diagnostic chains
Increased adoption of automated laboratory platforms
China follows closely at 5.3%, driven by hospital modernization programs, rising diagnostic quality standards, and government emphasis on laboratory accuracy.
Brazil also represents an increasingly important regional growth market as private laboratory investment accelerates and automated immunoassay platforms become more widespread.
By contrast, mature markets such as the United States, Germany, Japan, and South Korea are expected to deliver steadier, replacement-driven growth supported by established laboratory quality frameworks.
North America Remains the Regulatory Benchmark
The United States continues to exert outsized influence on global market standards due to the regulatory role of the FDA and the country's highly developed laboratory quality infrastructure.
The U.S. market is projected to grow at a CAGR of 3.8% through 2036, supported by:
Stringent interference testing requirements
Broad immunoassay utilization
High laboratory accreditation penetration
Rising awareness of biotin interference
Mature procurement ecosystems
In practice, many international diagnostic manufacturers align product development and validation strategies with U.S. regulatory expectations, making North America a de facto benchmark market for interference mitigation technologies.
Competitive Landscape: Technical Validation and Laboratory Relationships Matter More Than Scale Alone
The competitive environment remains moderately fragmented, combining large life sciences companies with specialized reagent developers.
Major participants include:
Bio-Rad Laboratories
Thermo Fisher Scientific
Merck KGaA (Sigma-Aldrich)
Abcam plc
Agilent Technologies
Abnova Corporation
SeraCare Life Sciences
AMSBIO
Large players maintain advantages through:
Broad distribution reach
Existing laboratory relationships
Integrated quality-control portfolios
Regulatory documentation capabilities
Global technical support infrastructure
However, specialized manufacturers continue to compete effectively by focusing on application-specific solutions, assay optimization, and technical differentiation.
Recent developments illustrate how the market is evolving toward workflow integration and specificity enhancement:
Thermo Fisher Scientific introduced a biotin-specific sample pre-treatment solution optimized for high-volume clinical laboratories.
Bio-Rad Laboratories launched an updated heterophilic antibody blocking kit targeting false-result reduction in clinical immunoassays.
Merck KGaA introduced a polymer-based blocking reagent for pharmaceutical development applications requiring higher assay sensitivity.
Strategic Implications for Diagnostics Companies and Laboratory Operators
For diagnostic manufacturers, interference mitigation is increasingly becoming a competitive differentiator rather than merely a compliance obligation.
Assay platforms that demonstrate lower susceptibility to interference may gain advantages in:
Regulatory review processes
Hospital procurement decisions
Accreditation compliance
Clinical trust and adoption
Long-term platform standardization
For laboratory operators, the market signals a broader operational shift toward preventive quality assurance models. As testing volumes rise and staffing pressures intensify, laboratories are prioritizing workflow simplification and automation-compatible blocking systems that reduce operator variability.
This trend also creates opportunities for integrated reagent-service ecosystems that combine:
Blocking reagents
Validation protocols
QC software integration
Automated sample preparation
Training and accreditation support
Browse Full Report: https://www.factmr.com/report/immunoassay-interference-blocker-market
Future Outlook: A Quiet but Essential Diagnostics Category
The immunoassay interference blocker market is unlikely to become one of the healthcare sector's largest diagnostic categories by revenue. Yet its importance within modern laboratory medicine is steadily increasing.
The future trajectory of the market will be shaped by three structural forces:
Rising global immunoassay testing volumes
Expanding regulatory scrutiny around assay accuracy
Growing recognition of interference-related clinical risk
At the same time, the market is becoming more technically sophisticated. Biotin interference mitigation, assay-specific blocker formulations, automated workflow integration, and kit-based standardized solutions are transforming what was historically viewed as a narrow reagent category into a more strategically differentiated diagnostics segment.
For executives, investors, and procurement leaders, the message is increasingly clear: interference mitigation is no longer a peripheral laboratory function. It is becoming a foundational requirement for maintaining diagnostic reliability in an increasingly automated, high-volume, and regulation-driven testing environment.
Full Report: Unlock 360° insights for strategic decision making and investment planning:
https://www.factmr.com/checkout/9395
To View Related Report:
Immunoassay Market https://www.factmr.com/report/4766/immunoassay-market
Radioimmunoassays Market https://www.factmr.com/report/radioimmunoassays-market
Biomarker-based Immunoassays Market https://www.factmr.com/report/biomarker-based-immunoassays-market
Chemiluminescence Immunoassay (CLIA) Analyzers Market https://www.factmr.com/report/4734/chemiluminescence-immunoassay-clia-analyzers-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Immunoassay Interference Blocker Market Sees North America & Asia-Pacific Regional Dominance | Bio-Rad, Thermo Fisher, Merck here
News-ID: 4507293 • Views: …
More Releases from Fact MR
Dental Prosthetics Market Witnesses Strong Regional Dominance in Asia & North Am …
The global dental prosthetics market is entering a new phase of transformation as digital workflows, aesthetic dentistry, and demographic change redefine restorative care economics. Once dominated by conventional crowns, dentures, and laboratory-intensive fabrication methods, the sector is increasingly shifting toward digitally enabled prosthetic ecosystems built around CAD/CAM systems, zirconia materials, implant-supported restorations, and same-day dentistry.
Get detailed market forecasts, competitive benchmarking, and pricing trends:
https://www.factmr.com/connectus/sample?flag=S&rep_id=12058
According to Fact.MR analysis, the global dental prosthetics…
Endoscopic Spinal Surgery Market Expands with Asia-Pacific & North America Domin …
The global endoscopic spinal surgery market is entering a decisive growth phase as hospitals, ambulatory surgical centers, and spine specialists increasingly shift toward minimally invasive procedures designed to reduce recovery times, shorten hospital stays, and improve surgical precision. What was once considered a niche technique for early adopters is now evolving into a broader clinical movement supported by advancing surgical technologies, expanding physician training programs, and mounting pressure to reduce…
Endodontics and Orthodontics Market Witnesses Strong Regional Dominance in North …
The global endodontics and orthodontics market is undergoing a structural transformation shaped by two parallel shifts in modern dentistry: the rise of tooth preservation-focused restorative care and the mainstream adoption of aesthetic orthodontic treatments among adults. What was once largely procedure-driven is increasingly becoming technology-driven, with digital workflows, AI-assisted treatment planning, and advanced biomaterials reshaping how dental practices diagnose, plan, and deliver care.
Get detailed market forecasts, competitive benchmarking, and pricing…
Dual Oral Antivirals Market Witnesses East Asia & North America Regional Dominan …
The global dual oral antivirals market is entering a more structurally mature phase, shaped less by emergency-driven demand cycles and increasingly by long-term disease management, regulatory refinement, and institutional procurement discipline. As healthcare systems expand outpatient treatment capabilities and prioritize scalable antiviral strategies, combination oral therapies are becoming central to treatment frameworks across both developed and emerging markets.
Get detailed market forecasts, competitive benchmarking, and pricing trends:
https://www.factmr.com/connectus/sample?flag=S&rep_id=11215
What distinguishes the current market…
More Releases for Immunoassay
Prominent Chemiluminescence Immunoassay Analyzer Market Trend for 2025: Innovati …
What combination of drivers is leading to accelerated growth in the chemiluminescence immunoassay analyzer market?
The increasing incidence of chronic illnesses is predicted to spur the expansion of the chemiluminescence immunoassay analyzer market. Factors like aging populations, unhealthy lifestyles, and advancements in healthcare which prolong the lifespan of individuals with severe conditions, contribute to the rise in chronic diseases. Chemiluminescence immunoassay analyzers play a crucial role in managing chronic illnesses by…
Global Bench-top Automated Immunoassay Analyzer Market Size - By Product Type(Ch …
Bench-top Automated Immunoassay Analyzer Market Insights: Trends, Drivers, and Outlook 2024 - 2031
Bench-top Automated Immunoassay Analyzer Market Scope: Unveiling Today's Trends
A Bench-top Automated Immunoassay Analyzer is a compact device used for conducting immunoassays in laboratory settings, enhancing efficiency and accuracy. The market for these analyzers is experiencing significant growth driven by rising demand for rapid diagnostic testing and advancements in technology. Increased prevalence of conditions such as infectious diseases and…
Immunoassay Market - Advancing Patient Care through Immunoassay Solutions
Newark, New Castle, USA: The "Immunoassay Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors.
Immunoassay Market: https://www.growthplusreports.com/report/immunoassay-market/8429
This latest report researches the industry structure, sales, revenue, price and…
Bahrain Immunoassay Analyzer Market
Transparent healthcare framework & strict procurement system enables the authorities in gaining the trust of the end-users leading to the growth of the medical devices industry in Bahrain: Ken Research
Heavy Government involvement in the industry: Bahrain has been maintaining a basic framework for a transparent, rules-based government procurement system. Bahrain hospitals and clinics, as well as developing specialist treatments, are continuously growing. This has improved the installation of BGA, POC…
Immunoassay Market Competitive Dynamics 2027
According to the new research report published by The Insight Partners, titled "Immunoassay Market- Global Analysis and Forecast to 2027".
The global immunoassay market is US$ 21,800.0 Mn in 2018 and is predicted to grow at a CAGR of 6.5% during the forecast period 2019 - 2027, to reach US$ 37,987.8 Mn by 2027.
Immunoassay Market Report presents a competitive scenario of key Immunoassay Industry players with their market share, sales, revenue…
Immunoassay Market: Opportunities and Challenges
The Immunoassay Market is expected to reach 27.15 billion by 2023 from USD 20.19 billion in 2018, at a CAGR of 6.1% during the forecast period.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=436
Will the development of condition-specific biomarkers and tests create opportunities for growth for players in the immunoassay market?
Biomarkers have gained significant clinical value and interest in the medical industry, as they are very useful in the screening, diagnosis, and treatment of diseases.…