Press release
POCT Chemiluminescence Immunoassay (CLIA) Analyzer Market Share Driven by Rapid Diagnostic Demand and Healthcare Automation Expansion | Valuates Reports
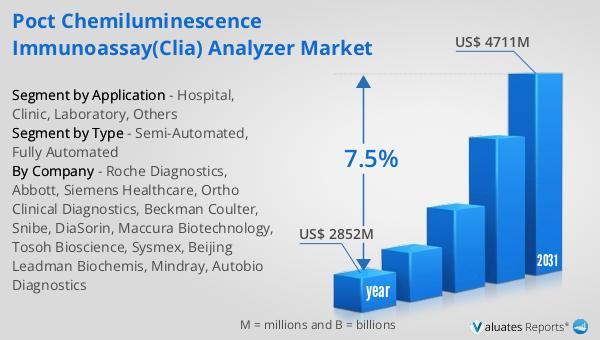
POCT Chemiluminescence Immunoassay(CLIA) Analyzer Market SizeThe global market for POCT Chemiluminescence Immunoassay(CLIA) Analyzer was valued at US$ 2852 million in the year 2024 and is projected to reach a revised size of US$ 4711 million by 2031, growing at a CAGR of 7.5% during the forecast period.
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-13R16278/Global_POCT_Chemiluminescence_Immunoassay_CLIA_Analyzer_Market_Research_Report_2023
The global POCT Chemiluminescence Immunoassay (CLIA) Analyzer market is witnessing strong market growth as healthcare providers increasingly adopt rapid, accurate, and decentralized diagnostic technologies to improve patient outcomes and clinical efficiency. Point-of-care testing chemiluminescence immunoassay analyzers are advanced diagnostic systems designed to detect biomarkers, infectious diseases, hormones, cardiac markers, and various clinical conditions through highly sensitive immunoassay techniques that provide fast and reliable results near the patient setting. Rising demand for rapid diagnostics, increasing prevalence of chronic and infectious diseases, and expanding healthcare infrastructure modernization are significantly influencing market size and long-term market forecast trends. One of the most important market trends shaping the industry is the growing shift toward decentralized healthcare delivery models that prioritize faster clinical decision-making, reduced laboratory turnaround time, and improved accessibility of diagnostic services. Hospitals, clinics, and diagnostic laboratories are increasingly integrating POCT CLIA analyzers into routine clinical workflows to support emergency diagnostics, disease monitoring, preventive healthcare, and personalized medicine initiatives. The increasing focus on healthcare automation, digital diagnostics, and integrated laboratory information systems is further strengthening market growth across the global in-vitro diagnostics industry. In addition, rising demand for high-sensitivity testing technologies and expanding use of immunoassay diagnostics in infectious disease detection, oncology, endocrinology, and cardiology are accelerating adoption of advanced CLIA analyzer systems worldwide. Manufacturers are heavily investing in miniaturization technologies, automated sample processing, artificial intelligence-assisted diagnostics, and multiplex testing capabilities to strengthen their market share and support evolving healthcare requirements.
Based on type segmentation, the Fully Automated segment currently holds the largest market share because healthcare providers increasingly prioritize high-throughput diagnostic systems capable of delivering rapid, accurate, and standardized testing with minimal manual intervention. Fully automated POCT CLIA analyzers are widely utilized in hospitals, centralized diagnostic facilities, and high-volume healthcare environments because they improve workflow efficiency, reduce human error, and enhance laboratory productivity. The growing demand for fast diagnostic turnaround and continuous expansion of automated healthcare infrastructure continue driving strong market growth within this segment. Healthcare institutions are increasingly investing in integrated automated platforms that support barcode tracking, digital reporting, quality control management, and seamless connectivity with hospital information systems. At the same time, the Semi-Automated segment continues maintaining an important market presence because these systems offer cost-effective diagnostic capabilities suitable for smaller healthcare facilities, independent clinics, and resource-constrained medical environments. Semi-automated analyzers are particularly valued in emerging healthcare markets where affordability, operational flexibility, and simplified testing procedures remain essential. Technological advancements in compact analyzer design, assay sensitivity, and user-friendly interfaces are significantly influencing broader market trends across both fully automated and semi-automated diagnostic systems. As healthcare providers continue prioritizing efficiency, diagnostic accuracy, and decentralized testing capabilities, the overall market forecast for POCT CLIA analyzer technologies remains highly favorable.
From an application perspective, the Hospital segment currently accounts for the largest market share due to the extensive use of POCT CLIA analyzers in emergency departments, intensive care units, outpatient services, and specialized diagnostic centers. Hospitals increasingly rely on rapid immunoassay testing technologies to support timely diagnosis, treatment planning, patient monitoring, and critical care decision-making across a wide range of medical conditions. The growing burden of chronic diseases, infectious disease outbreaks, and increasing patient demand for rapid healthcare services are significantly contributing to market growth within this segment. Healthcare providers are integrating advanced point-of-care testing systems into clinical workflows to improve operational efficiency, reduce patient waiting times, and optimize resource utilization. The Laboratory segment also represents a major application category because diagnostic laboratories increasingly utilize CLIA analyzers for high-sensitivity biomarker testing, hormone analysis, and infectious disease diagnostics. The increasing expansion of laboratory automation and precision diagnostics is strengthening broader market trends within this segment. Clinics are experiencing rapid market growth as outpatient healthcare facilities increasingly adopt compact and efficient diagnostic platforms to improve patient convenience and support decentralized testing services. The Others category includes community healthcare centers, mobile diagnostic units, specialty medical facilities, and research institutions utilizing advanced immunoassay technologies for clinical and analytical applications. As healthcare systems continue emphasizing early disease detection, rapid diagnostics, and personalized treatment approaches, the market forecast for POCT CLIA analyzer applications remains highly optimistic.
The competitive landscape of the POCT Chemiluminescence Immunoassay Analyzer market is characterized by continuous technological innovation, strategic healthcare partnerships, and increasing investments in automated diagnostic platforms. Roche Diagnostics remains one of the companies with the largest market share due to its strong global presence in in-vitro diagnostics, advanced immunoassay technologies, and comprehensive clinical testing portfolio supporting hospitals and laboratories worldwide. The company continues strengthening its position through highly automated diagnostic systems, integrated digital healthcare platforms, and broad assay development capabilities. Abbott also maintains a significant market presence because of its expertise in point-of-care diagnostics, rapid testing technologies, and automated healthcare solutions supporting decentralized medical services. Siemens Healthcare continues contributing strongly to market growth through advanced immunoassay analyzers, integrated laboratory automation systems, and digital healthcare technologies designed for high-throughput diagnostic environments. Beckman Coulter is recognized for its strong position in laboratory diagnostics and automated testing systems supporting clinical chemistry and immunoassay applications. Ortho Clinical Diagnostics and DiaSorin continue expanding their market share through specialized immunodiagnostic technologies and advanced assay development supporting infectious disease and specialty testing applications. Sysmex maintains an important market presence through integrated diagnostic platforms and expanding healthcare automation capabilities. Snibe, Mindray, Maccura Biotechnology, Autobio Diagnostics, Beijing Leadman Biochemis, and Tosoh Bioscience are increasingly gaining market attention through cost-effective analyzer systems, expanding product portfolios, and growing international distribution networks supporting emerging healthcare markets. Industry competition is expected to intensify further as companies focus on artificial intelligence-assisted diagnostics, multiplex testing technologies, miniaturized analyzer systems, cloud-based healthcare integration, and next-generation biomarker detection capabilities supporting precision medicine and decentralized healthcare delivery.
Regionally, North America currently dominates the POCT Chemiluminescence Immunoassay Analyzer market in terms of market share due to advanced healthcare infrastructure, strong adoption of diagnostic automation technologies, and increasing investments in rapid testing capabilities across the United States and Canada. The region benefits from extensive healthcare spending, strong laboratory networks, and growing emphasis on early disease detection and personalized medicine initiatives. The United States remains one of the largest contributors to market growth because of its highly developed diagnostic industry, large hospital networks, and rapid integration of point-of-care testing technologies into clinical practice. Europe also represents a major market supported by strong public healthcare systems, advanced laboratory infrastructure, and increasing demand for automated diagnostics across Germany, France, the United Kingdom, Italy, and Russia. Asia-Pacific is emerging as the fastest-growing regional market due to expanding healthcare infrastructure, rising healthcare awareness, increasing chronic disease prevalence, and rapid modernization of diagnostic services across China, Japan, South Korea, and Taiwan. China continues strengthening its market position through substantial healthcare investments, expanding diagnostic manufacturing capabilities, and increasing adoption of advanced medical technologies. Southeast Asia, particularly India, is witnessing rapid market growth because of expanding healthcare access, rising diagnostic demand, and increasing investments in laboratory infrastructure modernization. Latin America is also gradually increasing market participation through healthcare expansion initiatives and growing adoption of advanced diagnostic technologies across Mexico and Brazil. The overall market forecast for the POCT Chemiluminescence Immunoassay Analyzer market remains highly positive as healthcare systems continue prioritizing rapid diagnostics, decentralized healthcare delivery, and automated testing solutions. Future market trends are expected to focus on portable diagnostic platforms, artificial intelligence-enabled clinical decision support, multi-analyte testing systems, digital healthcare integration, and broader accessibility of point-of-care diagnostics that will continue driving long-term market growth globally.
Segment by Type
• Semi-Automated
• Fully Automated
Segment by Application
• Hospital
• Clinic
• Laboratory
• Others
By Company
Roche Diagnostics, Abbott, Siemens Healthcare, Ortho Clinical Diagnostics, Beckman Coulter, Snibe, DiaSorin, Maccura Biotechnology, Tosoh Bioscience, Sysmex, Beijing Leadman Biochemis, Mindray, Autobio Diagnostics
View full report
https://reports.valuates.com/market-reports/QYRE-Auto-13R16278/global-poct-chemiluminescence-immunoassay-clia-analyzer
Valuates,
4th Floor,
Balaraj's Arcade,
Whitefield Main road,
Bangalore 560066,
Valuates offers an extensive collection of market research reports that helps companies to take intelligent strategical decisions based on current and forecasted Market trends.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release POCT Chemiluminescence Immunoassay (CLIA) Analyzer Market Share Driven by Rapid Diagnostic Demand and Healthcare Automation Expansion | Valuates Reports here
News-ID: 4506346 • Views: …
More Releases from Valuates Reports

800V Silicon Carbide Inverters for EV Market Share Driven by Fast-Charging Elect …
800V Silicon Carbide Inverters for EV Market Size
The global market for 800V Silicon Carbide Inverters for EV was valued at US$ 184 million in the year 2024 and is projected to reach a revised size of US$ 923 million by 2031, growing at a CAGR of 22.3% during the forecast period.
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-14P19290/Global_800V_Silicon_Carbide_Inverters_for_EV_Market_Research_Report_2025
The global 800V Silicon Carbide Inverters for EV market is experiencing rapid market growth as automotive manufacturers accelerate…
Automotive Testing Equipment Market Share Driven by Vehicle Electrification and …
Automotive Testing Equipment Market Size
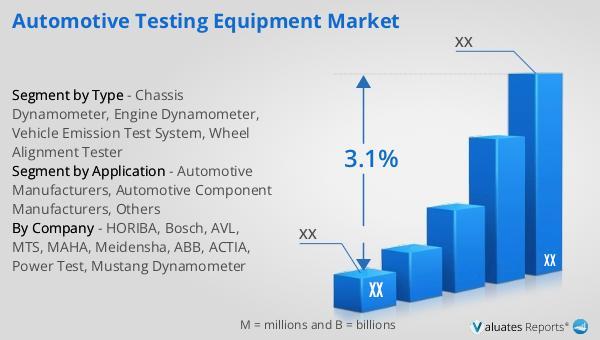
In 2024, the global market size of Automotive Testing Equipment was estimated to be worth US$ 2283 million and is forecast to reach approximately US$ 2818 million by 2031 with a CAGR of 3.1% during the forecast period 2025-2031.
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-30O5123/Global_Automotive_Testing_Equipment_Market_Size_Manufacturers_Supply_Chain_Sales_Channel_and_Clients_2021_2027
The global Automotive Testing Equipment market is witnessing substantial market growth as automotive manufacturers and component suppliers increasingly invest in advanced vehicle validation, performance optimization, and…

Semiconductor Process Parts Market Share Driven by Advanced Chip Manufacturing a …
Semiconductor Process Parts Market Size
The global market for Semiconductor Process Parts was valued at US$ 25487 million in the year 2024 and is projected to reach a revised size of US$ 39484 million by 2031, growing at a CAGR of 6.2% during the forecast period.
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-6C19994/Global_Semiconductor_Process_Parts_Market_Research_Report_2025
The global Semiconductor Process Parts market is witnessing substantial market growth as semiconductor manufacturers accelerate investments in advanced fabrication technologies, high-performance chip production, and…
Maritime Software Market Share Driven by Digital Fleet Management and Smart Ship …
Maritime Software Market Size
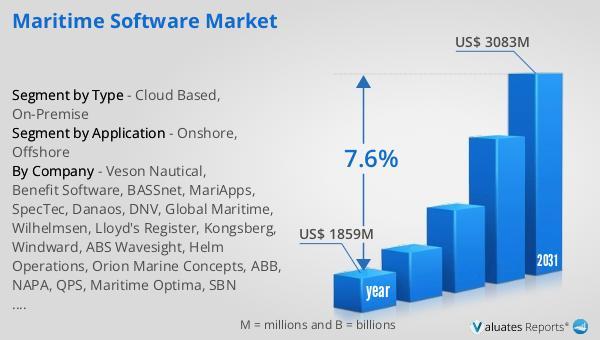
The global market for Maritime Software was valued at US$ 1859 million in the year 2024 and is projected to reach a revised size of US$ 3083 million by 2031, growing at a CAGR of 7.6% during the forecast period.
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-6T6169/Global_Maritime_Software_Market_Research_Report_2022
The global Maritime Software market is witnessing strong market growth as shipping companies, port operators, offshore service providers, and maritime logistics organizations increasingly adopt digital platforms…
More Releases for POCT
POCT Product and Devices Market Key Players, Share and Forecast Outlook
"The global Point-of-Care Testing (POCT) market is valued at approximately $35 billion in 2024, driven by technological advancements and a growing emphasis on decentralizing healthcare. By 2034, the market is projected to reach around $60 billion, demonstrating significant growth opportunities. This trajectory implies a robust Compound Annual Growth Rate (CAGR) of about 5.5% from 2025 to 2034."
Exactitude Consultancy., Ltd. released a research report titled "POCT Product and Devices Market". This…
Point-of-Care Testing (POCT) Market Size, Opportunities, Trends And Scope 2032
Point-of-Care Testing (POCT) Market Outlook & Investment Analysis
The Point-of-Care Testing (POCT) market is experiencing rapid growth due to increasing demand for rapid diagnostic solutions, technological advancements, and rising healthcare expenditure. The market is driven by the need for decentralized testing, particularly in remote and resource-limited areas. The growing prevalence of chronic diseases, such as diabetes and cardiovascular disorders, further accelerates adoption. North America dominates the market due to its well-established…
Molecular POCT Diagnostics Research:CAGR of 9.7% during the forecast period
Molecular POCT Diagnostics Market Summary
Molecular POCT (Point-of-Care Testing) Diagnostics refers to a diagnostic method that enables the detection of molecular markers-such as nucleic acids (DNA, RNA) or proteins-in biological samples (e.g., blood, saliva, or nasal swabs) directly at or near the site of patient care. Unlike traditional laboratory-based tests, which require sending samples to a central lab for analysis, molecular POCT diagnostics allow for rapid and decentralized testing, often providing…
India POCT Market Outlook to 2027F: Ken Research
India ranks at number 7 amongst the 20 wellness tourism markets with millions of trips made to India, medical value tourism generated multi-billion in revenue in 2022.
One of the nations where pricey medical procedures can be afforded is India. The cost of treatment in India is thought to be only 10% to 20% of what comparable procedures would cost in the West and other nations. India has a sizable number of hospitals with Joint Commission International…
Point-of-Care Testing (POCT) Market Current Scenario Forecast to 2027
The Global “Point-of-Care Testing (POCT)” Market Report 2021 to 2027 could be a fundamental examination of the global analysis. Trending innovation, advertise drivers, sectional declination, analysis measurements, advertise forecasts, manufacturers, and hardware merchants are all included within the substance. The report incorporates a point-by-point examination of the Point-of-Care Testing (POCT) Market as well as empowering advances, current patterns, openings, and obstruction, as well as a self-governing point of view, arrangement…
What Does POCT Mean?
POCT, which means point-of-care testing, is defined as medical diagnostic testing at or near the point of care. In these years, POCT received more and more attention, and we’d like to give a simple introduction in order to have a general idea about point-of-care.
What is POCT test?
POCT is medical abbreviation of Point-of-care testing, also known as bedside testing. It’s a form of testing which is designed to be used at…