Press release
Clinical Trials Market to Exceed USD 103.7 Billion by 2035, Accelerated by Decentralized Trial Models, AI-Driven Recruitment Platforms, and Precision Medicine Research
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global clinical trials market is valued at USD 57.2 billion in 2025 and is projected to reach approximately USD 103.7 billion by 2035, expanding at a CAGR of 6.1% during the forecast period (2025-2035). The market is experiencing sustained expansion as pharmaceutical companies, biotechnology firms, contract research organizations (CROs), and healthcare research institutions accelerate investment in decentralized clinical trial platforms, artificial intelligence-driven patient recruitment systems, digital clinical operations, and biomarker-based precision medicine studies.The increasing complexity of oncology, immunology, cardiovascular, and rare disease drug development programs is driving demand for scalable clinical research infrastructure, advanced analytics platforms, real-time patient monitoring technologies, and remote clinical trial management systems. Sponsors are increasingly adopting hybrid and decentralized trial models to improve patient accessibility, accelerate enrollment timelines, strengthen operational efficiency, and reduce development cycle delays.
Clinical trial ecosystems are rapidly evolving beyond conventional site-based research operations into digitally connected, AI-enabled clinical development environments integrating wearable technologies, electronic patient-reported outcomes (ePRO), predictive analytics, remote monitoring systems, cloud-native clinical data management, and virtual patient engagement platforms. As regulatory agencies and pharmaceutical sponsors prioritize patient diversity, operational scalability, and faster therapeutic commercialization, digital clinical research infrastructure is emerging as a strategic investment category across global drug development ecosystems.
Get Sample Copy of the Report: https://marketgenics.co/download-report-sample/clinical-trials-market-17657
==============================
MARKET OVERVIEW - KEY STATISTICS
Drivers - Drug Development Activity & Outsourcing Contract Research
Rising drug development activity and outsourcing to contract research organizations are driving the clinical trials market by enabling faster study execution, improved operational efficiency, and better management of complex global trials.
Pharmaceutical and biotechnology companies are increasingly expanding research pipelines across oncology, immunology, rare diseases, and precision medicine, creating strong demand for scalable clinical trial infrastructure and outsourced research capabilities. Sponsors are partnering with CROs to accelerate patient recruitment, optimize trial operations, improve regulatory compliance, and streamline global multi-site clinical development programs.
Key Fact: According to the Pharmaceutical Research and Manufacturers of America, more than 8,000 medicines are currently in clinical development worldwide.
Challenges - Patient Recruitment Challenges and High Trial Costs
Patient recruitment challenges and high trial costs are limiting the clinical trials market, as sponsors face difficulties in enrolling eligible participants, maintaining patient retention, and managing rising operational expenses associated with complex protocols, site management, and regulatory compliance.
Clinical trial sponsors continue to face operational challenges associated with strict eligibility criteria, geographic barriers, rising site management costs, and increasingly complex regulatory frameworks across multiple regions. High expenditures related to data management, patient engagement, compliance monitoring, and remote trial infrastructure continue to create financial pressure across small biotechnology firms and academic research institutions.
Regulatory Landscape
Regulatory oversight of the clinical trials market is governed by agencies such as the U.S. Food and Drug Administration, European Medicines Agency, and frameworks like ICH-GCP Guidelines that mandate strict standards for patient safety, data integrity, and ethical trial conduct.
Global regulatory authorities are increasingly prioritizing decentralized trial governance, electronic clinical data integrity, patient transparency, and real-world evidence generation to support more efficient and compliant clinical research ecosystems.
Impact: These regulations enhance transparency, patient protection, and data reliability while increasing compliance requirements and operational complexity for sponsors and research organizations.
==============================
EMERGING MARKET OPPORTUNITIES
Global Market Opportunity
~USD 46.5 Billion Total Forecast Opportunity by 2035
The global clinical trials market is expected to create an estimated incremental opportunity of over USD 46.5 billion by 2035, driven by increasing adoption of decentralized clinical trial technologies, artificial intelligence-enabled trial optimization systems, digital patient engagement platforms, and biomarker-based precision medicine studies. Expanding use of predictive analytics, virtual trial management systems, remote patient monitoring technologies, and electronic data capture platforms is transforming global clinical research infrastructure.
The market is further benefiting from rising pharmaceutical R&D investments, growing adoption of personalized medicine, increasing demand for faster drug commercialization, and expanding use of advanced analytics to improve patient matching and operational efficiency. Sponsors are increasingly prioritizing AI-powered trial intelligence platforms, decentralized research ecosystems, and scalable clinical data management systems capable of accelerating global study execution.
Regional Opportunity Analysis
~USD 24 Billion Opportunity in North America During 2026-2035
North America represents the most commercially attractive regional market within the global clinical trials industry, accounting for an estimated opportunity of nearly USD 24 billion during the forecast period (2026-2035). The region continues to dominate due to strong pharmaceutical and biotechnology research activity, a well-established regulatory framework, and the presence of leading research institutions and contract research organizations.
High research and development investments, advanced healthcare infrastructure, and rapid adoption of decentralized and digital trial technologies further strengthen the region's leadership. Additionally, supportive regulatory pathways and extensive patient registries enable faster trial initiation, recruitment, and data collection across large multi-site clinical research programs.
At the same time, Asia Pacific, Latin America, and Middle East healthcare markets are emerging as high-growth clinical research expansion regions as governments, CROs, and pharmaceutical sponsors increase investment in digital trial infrastructure, precision medicine studies, and AI-powered clinical development systems. Growing availability of diverse patient populations and lower operational costs are expected to create substantial commercialization opportunities across emerging clinical research markets.
==============================
KEY FINDINGS OF THE CLINICAL TRIALS MARKET REPORT
• Total market opportunity of approximately USD 46.5 billion by 2035
• Phase III segment accounts for approximately 50%-55% of the global market share in 2025
• North America accounts for nearly 50%-55% of the global market share
• Top five players control nearly 40% of total market revenue
• Decentralized trial technologies and AI-powered recruitment systems are among the fastest-growing solution categories
• Oncology, immunology, and precision medicine studies remain high-growth therapeutic segments
• Contract research organizations (CROs) continue to represent the dominant operational ecosystem globally
==============================
ANALYST VIEWPOINT
The clinical trials market is transitioning from traditional site-centric research operations into digitally connected, intelligence-driven clinical development ecosystems supporting decentralized participation, predictive recruitment analytics, real-time monitoring, and precision medicine research. The competitive landscape is increasingly shaped by the convergence of artificial intelligence, decentralized trial technologies, wearable devices, cloud-native clinical data systems, and remote patient engagement infrastructure.
As pharmaceutical companies and contract research organizations globally continue to prioritize operational scalability, faster study execution, patient diversity, and regulatory compliance, enterprise adoption is increasingly shifting toward integrated digital clinical research ecosystems capable of delivering real-time trial intelligence, predictive enrollment optimization, remote patient participation, and advanced data analytics.
Competitive positioning within the industry is increasingly defined by:
• decentralized clinical trial infrastructure
• AI-powered patient recruitment and matching
• real-time clinical data analytics
• cloud-native clinical trial management systems
• wearable-enabled remote monitoring technologies
• electronic patient-reported outcome platforms
• biomarker-driven precision medicine capabilities
• regulatory compliance and global study execution expertise
Organizations capable of combining scalable research infrastructure, advanced analytics capabilities, decentralized trial ecosystems, and regulatory execution efficiency are expected to establish long-term competitive advantage within the global clinical trials market.
==============================
SEGMENTAL INSIGHTS & GROWTH ANCHORS
Segmental Focus
Market Share by Phase (2025)
• Phase I
• Phase III (50-55%)
• Phase II
• Phase IV (Post-Marketing Surveillance)
• Combined Phases (Phase I/II, Phase II/III)
Phase III Segment
Phase III segment accounts for over 50% of the global clinical trials market in 2025, driven by its large-scale evaluation of efficacy and safety required for regulatory approval.
Phase I Segment
Phase I is the second fastest-growing segment, driven by increasing early-stage drug development and seamless transition between phases for faster clinical advancement.
Phase IV (Post-Marketing Surveillance) Segment
Phase IV (Post-Marketing Surveillance) segment is the least-growing category, due to its limited scope and smaller patient populations compared to pre-approval trials.
Regional Market Insights
The North America region dominates the global clinical trials market, accounting for 50-55% of the total market share in 2025, with projected CAGR of over 4% from 2026 to 2035, significantly outpacing global growth rates.
Regional Share Analysis, 2025
• USA: 85-90%
• Canada
• Mexico
Key Technology Trends
North America leads the clinical trials market due to strong pharmaceutical and biotechnology research activity and high adoption of advanced clinical research technologies. Key trends include the expansion of decentralized trials, artificial intelligence-driven patient recruitment, and increasing use of digital health tools for remote monitoring.
High Growth Country
The United States drives growth through substantial pharmaceutical research and development investments, a large patient pool for diverse studies, and supportive regulatory frameworks.
==============================
COMPETITIVE LANDSCAPE & KEY PLAYERS
Global Company Market Share (2025)
• IQVIA Holdings Inc.
• Laboratory Corporation of America Holdings
• ICON plc
• Pharmaceutical Product Development, LLC (PPD)
• Syneos Health, Inc.
• Other Key Players
The global clinical trials market remains moderately consolidated with the top 5 manufacturers controlling nearly 40% of the market share in 2025. IQVIA Holdings Inc. maintains its leadership in the global clinical trials market through comprehensive end-to-end solutions, advanced data analytics, and global contract research services. Regional players are increasingly gaining ground in emerging markets, especially across North America, where local firms are expanding access.
==============================
IQVIA HOLDINGS INC.
Market Leader
Company Revenue
USD 15.4 Bn
Headquarters
United States
Innovation Highlights
In 2025, IQVIA Holdings Inc. introduced its IQVIA AI-Powered Clinical Insights Platform, incorporating machine learning algorithms, predictive analytics, and electronic patient-reported outcomes, enabling sponsors and contract research organizations to improve patient matching, monitor trial progress in real time, ensure regulatory compliance, and optimize operational efficiency.
==============================
LABCORP HOLDINGS INC.
Innovation Leader
Company Revenue
USD 13.01 Bn
Headquarters
United States
Innovation Highlights
Labcorp Holdings Inc. partnered with Medable to integrate decentralized trial technologies and real-time patient monitoring into its clinical research operations, combining Labcorp's laboratory and data infrastructure with advanced digital health tools to deliver secure, efficient, and patient-centric solutions for pharmaceutical sponsors, biotechnology companies, and global multi-site clinical trials.
==============================
OTHER KEY COMPANIES OPERATING IN THE CLINICAL TRIALS MARKET INCLUDE:
• Charles River Laboratories International, Inc.
• CROMSOURCE S.r.l.
• Ergomed plc
• Eurofins Scientific SE
• Fortrea Holdings Inc.
• WuXi AppTec Co., Ltd.
• Medpace Holdings, Inc.
• Novotech Pty Ltd
• PSI CRO AG
• Parexel International Corporation
• SGS SA
• Precision for Medicine, Inc.
• Thermo Fisher Scientific Inc.
• Syngene International Limited
• Worldwide Clinical Trials Holdings, Inc.
• Other Key Players
==============================
RECENT DEVELOPMENTS AND STRATEGIC DIRECTION
In October 2024, IQVIA established its One Home for Sites platform to improve clinical trial site operations through its digital platform which combines all essential workflows from regulatory document management to patient recruiting and financial control. The solution decreases research site administrative requirements while improving sponsor-investigator collaboration and shortening global clinical trial study initiation processes.
In May 2024, Medidata Solutions introduced new AI-enabled capabilities within its Medidata Clinical Data Studio platform to enhance clinical data quality and streamline data reviewing tasks. The platform provides a combination of advanced analytics and centralized monitoring tools which enable sponsors and contract research organizations to detect risks at earlier stages while achieving better data accuracy and improved operational effectiveness throughout the clinical trial process.
Pharmaceutical companies and contract research organizations are increasingly prioritizing decentralized clinical trial infrastructure, AI-driven recruitment platforms, precision medicine research ecosystems, and digital patient engagement technologies capable of accelerating study execution, improving patient participation, and optimizing global trial performance.
==============================
CLINICAL TRIALS MARKET TRENDS & INNOVATION LANDSCAPE
The clinical trials market is witnessing accelerated innovation driven by increasing integration of decentralized trial technologies, artificial intelligence, predictive analytics, wearable devices, telemedicine platforms, and cloud-native clinical research systems. Pharmaceutical companies and CROs are increasingly deploying digitally connected trial ecosystems capable of supporting remote participation, real-time monitoring, predictive recruitment, and scalable global clinical development operations.
At the same time, biomarker-driven precision medicine studies, AI-powered protocol optimization, electronic patient-reported outcome systems, and decentralized patient engagement platforms are transforming clinical research models globally. Digital clinical infrastructure and remote trial management ecosystems are becoming strategic priorities across pharmaceutical and biotechnology R&D initiatives.
==============================
CLINICAL TRIALS MARKET SEGMENTATION
Global Clinical Trials Market Analysis, by Phase
• Phase I
• Phase II
• Phase III
• Phase IV (Post-Marketing Surveillance)
• Combined Phases (Phase I/II, Phase II/III)
Global Clinical Trials Market Analysis, by Study Design
• Interventional Studies
• Observational Studies
• Expanded Access Studies
• Randomized Trials
• Non-Randomized Trials
Global Clinical Trials Market Analysis, by Sponsor Type
• Pharmaceutical & Biotechnology Companies
• Medical Device Companies
• Academic & Research Institutes
• Government Organizations
• Non-Profit Organizations
• Others
Global Clinical Trials Market Analysis, by Therapeutic Area
• Oncology
• Cardiovascular Diseases
• Infectious Diseases
• Neurology Disorders
• Metabolic Disorders (Diabetes, Obesity)
• Respiratory Diseases
• Immunology & Autoimmune Diseases
• Rare Diseases
• Others
Global Clinical Trials Market Analysis, by Indication Type
• Chronic Diseases
• Acute Diseases
• Rare Diseases
• Infectious Diseases
• Genetic Disorders
• Others
Global Clinical Trials Market Analysis, by Service Type
• Clinical Trial Management Services
• Site Management Services
• Patient Recruitment & Retention Services
• Data Management & Biostatistics Services
• Medical Writing Services
• Regulatory & Compliance Services
• Pharmacovigilance Services
• Laboratory Services
• Others
Global Clinical Trials Market Analysis, by Delivery Model
• In-house Clinical Trials
• Outsourced Clinical Trials (CROs)
• Hybrid Model
Global Clinical Trials Market Analysis, by End User
• Pharmaceutical & Biopharmaceutical Companies
• Medical Device Companies
• Academic Institutes
• Contract Research Organizations (CROs)
• Hospitals & Clinical Research Centers
• Others
Global Clinical Trials Market, by Region
• North America
• Europe
• Asia Pacific
• Middle East
• Africa
• South America
==============================
FUTURE OUTLOOK & STRATEGIC PERSPECTIVE
The clinical trials market is positioned for substantial long-term transformation as pharmaceutical sponsors, biotechnology firms, and CROs globally accelerate adoption of decentralized clinical development, AI-enabled trial intelligence, precision medicine research, and digital patient engagement ecosystems. The integration of predictive analytics, remote monitoring technologies, biomarker-driven studies, and cloud-native clinical trial management systems is expected to redefine global clinical development over the next decade.
North America will continue to lead commercialization and large-scale study execution, while Asia Pacific is expected to emerge as a high-growth clinical research region driven by expanding pharmaceutical manufacturing, diverse patient populations, and accelerating investment in digital research infrastructure. As market competition intensifies, leadership will increasingly depend on decentralized trial execution capabilities, AI-powered operational intelligence, global regulatory expertise, patient recruitment efficiency, and scalable clinical analytics infrastructure.
Access the Full Report and Strategic Intelligence Insights: https://marketgenics.co/reports/clinical-trials-market-17657
==============================
RECOMMENDED REPORTS:
Patient Engagement Solutions Market: https://marketgenics.co/reports/patient-engagement-solutions-market-21970
Healthcare Interoperability Solutions Market: https://marketgenics.co/reports/healthcare-interoperability-solutions-market-85413
Contact:
Mr. Debashish Roy
MarketGenics Global Research
800 N King Street, Suite 304 #4208, Wilmington, DE 19801, United States
USA: +1 (302) 303-2617
Email: sales@marketgenics.co
Website: https://marketgenics.co
About MarketGenics
MarketGenics is a global market research and business advisory firm empowering decision-makers across startups, Fortune 500 companies, non-profit organizations, universities, and government institutions. The company delivers comprehensive market intelligence, industry analysis, and strategic insights across diverse sectors.
MarketGenics publishes detailed industry research reports combining granular quantitative analysis with expert insights on market trends, competitive landscapes, and emerging opportunities. These reports help organizations make informed strategic decisions, identify growth opportunities, and support sustainable business development.
In addition to research publications, MarketGenics supports organizations with strategic insights on product development, application modeling, market expansion strategies, and identifying niche growth opportunities.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Clinical Trials Market to Exceed USD 103.7 Billion by 2035, Accelerated by Decentralized Trial Models, AI-Driven Recruitment Platforms, and Precision Medicine Research here
News-ID: 4506240 • Views: …
More Releases from MarketGenics Global Research
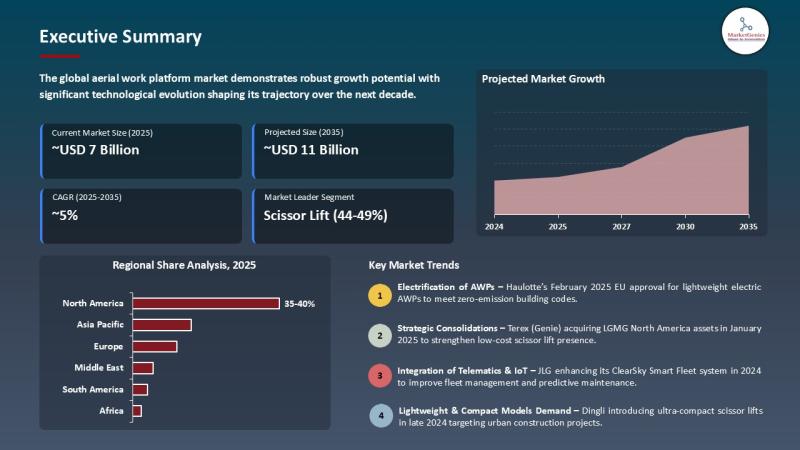
Aerial Work Platform Market to Reach USD 10.8 Billion by 2035, Driven by Smart C …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics, the global aerial work platform market is valued at USD 6.7 billion in 2025 and is projected to reach approximately USD 10.8 billion by 2035, expanding at a CAGR of 4.9% during the forecast period. Growth is primarily driven by rising urban infrastructure projects, increasing focus on worker safety, rapid adoption of electric aerial work platforms, and growing demand for…
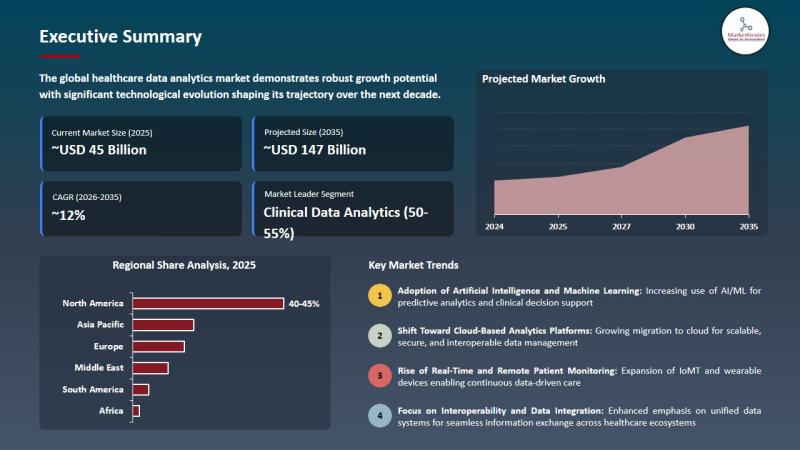
Healthcare Data Analytics Market to Reach USD 146.9 Billion by 2035, Fueled by A …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global healthcare data analytics market is valued at USD 45.6 billion in 2025 and is projected to reach approximately USD 146.9 billion by 2035, expanding at a CAGR of 12.4% during the forecast period (2025-2035). The market is experiencing accelerated growth as hospitals, healthcare providers, payers, pharmaceutical companies, and healthcare IT organizations increase investment in artificial intelligence-driven…
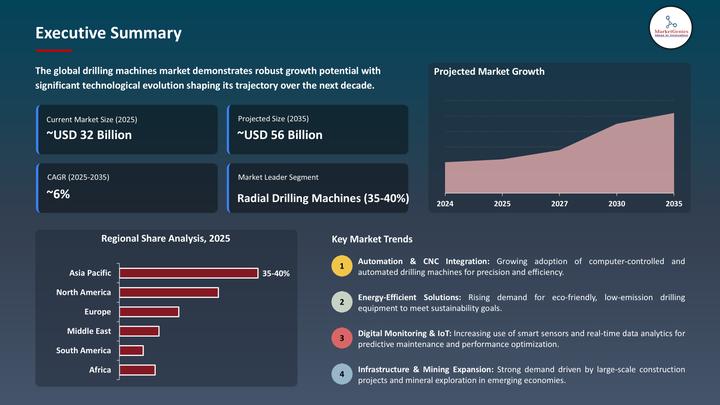
Global Drilling Machines Market to Reach USD 55.7 Billion by 2035, Driven by Aut …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics, the global drilling machines market is valued at USD 31.7 billion in 2025 and is projected to reach approximately USD 55.7 billion by 2035, expanding at a CAGR of 5.8% during the forecast period. Growth is primarily driven by rapid industrialization, increasing infrastructure development, rising mining activities, and growing adoption of CNC-enabled and automated drilling technologies across manufacturing, construction, aerospace,…
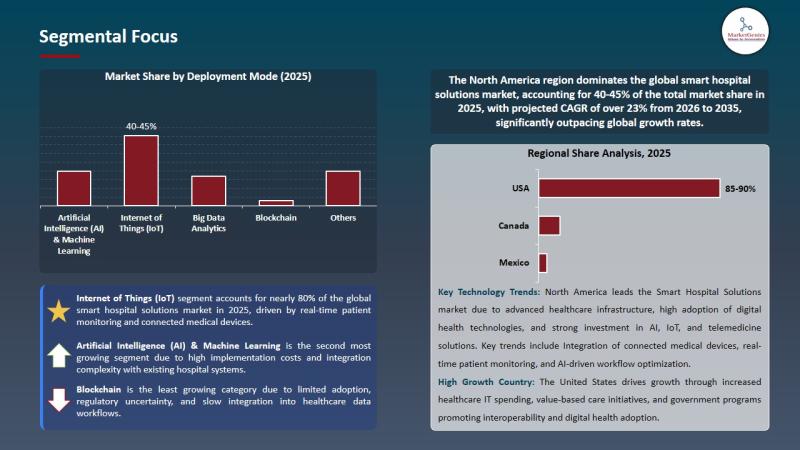
Smart Hospital Solutions Market to Surpass USD 34.3 Billion by 2035, Driven by A …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global smart hospital solutions market is valued at USD 2.8 billion in 2025 and is projected to reach approximately USD 34.3 billion by 2035, expanding at a CAGR of 28.4% during the forecast period (2026-2035). The market is witnessing accelerated transformation as hospitals, healthcare systems, digital health providers, and enterprise healthcare technology companies increase investment in AI-powered…
More Releases for Phase
The Quiet Phase Is Almost Over: BlazPay's Phase 8 Presale Is 90% Sold Out
There's a moment in every successful crypto project that most people miss. It's not the launch. It's not the exchange listing. It's the final stretch of the presale - when the numbers are real, the momentum is visible, and the entry price is about to change forever. BlazPay is at that moment right now.
Phase 8 of the BlazPay presale is 90.4% sold. Over 267 million of 295 million BLAZ tokens…
Single-Phase and Three-Phase Amplifiers: An Innovative Solution to Overcome Sing …
I. Rural Power Grid Upgrades: Solving Power Supply Challenges in Remote AreasChallenges:In remote rural areas, long supply radii and scattered users, combined with single-phase power supply, lead to unstable end-point voltages and insufficient power, making it difficult to meet the electricity demands of modern home appliances and agricultural machinery.Benefits:Improved Voltage Quality: By converting single-phase power into balanced three-phase output, line losses are reduced, resolving the issue of low end-point voltages.Expanded…
Clinical Trials by Phase (Phase I, Phase II, Phase III, Phase IV) Market Forecas …
A clinical trial is a research study, where a group of people is given a test or treatment. Clinical trials study the safety and efficacy of tests and treatments. If the test or treatment is safe and meets regulatory requirements, then it is approved as a standard of care.
Download Sample Copy at https://www.theinsightpartners.com/sample/TIPRE00006203/?utm_source=OpenPR&utm_medium=10379
Key Players Analysis:
IQVIA
Parexel International Corporation
Charles River Laboratories
ICON plc
SGS SA
Chiltern International Ltd
Syneos Health
PRA Health Sciences
Wuxi AppTec Inc
Pharmaceutical Product Development,…
Digital Phase Shifters Market
Digital Phase Shifters Market A recently identified vacuum in the literature about the creation of digital phase shifters for modern communication systems is attempted to be filled in the book Design of Digital Phase Shifters for Multipurpose Communication Systems. By significantly reducing RF power consumption and improving noise immunity, directed beams enhance the development of new-generation mobile communication systems. In this regard, digital phase shifters in particular, which are part…
COVID-19 - Pipeline Analysis 2020 for Global Market | Emphasis on Products cover …
COVID-19 (also known as Anderson COVID-19)?is a viral disease caused by RNA virus, SARS-CoV-2 or commonly known as corona virus. These viruses can cause respiratory, enteric, hepatic, and neurologic diseases. At the end of 2019, a new coronavirus was identified as the cause of a cluster of pneumonia cases in Wuhan, China. It rapidly spread, resulting in an epidemic throughout China, followed by an increasing number of cases in other…
HIV Vaccines Market Perceive Aggrandized Growth at a CAGR of 11.48% Till 2023 | …
HIV Vaccines Market Report Added on MarketResearchFuture.com with Overall Analysis. Key developments and Strategies Cover in this Report. The Market for Expected to Grow Globally Over the CAGR of 5 % During the Period 2018 to 2027 from USD 2,702.3 Billion in 2027.
HIV Vaccines Market - Segmentation
The global HIV vaccines market has been segmented on the basis of basis of antibiotics, type, and lastly, region.
Antibiotics have been segmented into dicloxacillin,…
