Press release
Prefillable Syringe Coating and Lubrication Systems Market - Competitive Landscape and Growth Forecast
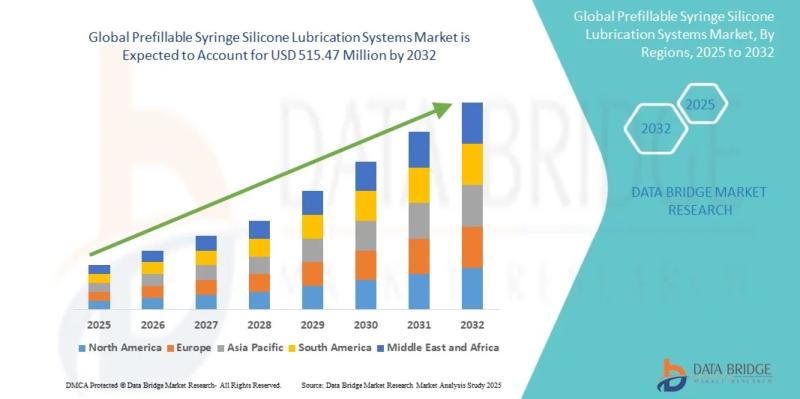
As per Data Bridge Market Research analysis, the Prefillable Syringe Silicone Lubrication Systems Market was estimated at USD 0.22 billion in 2025. The market is expected to grow from USD 0.25 billion in 2026 to USD 0.52 billion in 2032, at a CAGR of 12.92% during the forecast period with driven by the rising demand for biologics, injectable therapeutics, advanced drug delivery systems, and silicone oil-free syringe technologies.Get the full PDF sample copy of the report: https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-prefillable-syringe-silicone-lubrication-systems-market
The market is witnessing strong expansion due to the increasing adoption of prefilled syringes across vaccines, biologics, insulin therapies, and ophthalmic drugs. Pharmaceutical manufacturers are focusing on advanced silicone lubrication technologies to improve syringe glide force consistency, reduce protein aggregation risks, and enhance drug compatibility for sensitive biologics. Rising investments in injectable drug manufacturing infrastructure and increasing regulatory emphasis on particle contamination reduction are accelerating market adoption globally.
Growth is further supported by the rapid expansion of self-administration devices, including auto-injectors and wearable injectors, particularly in North America and Europe. Technological innovations such as baked-on silicone, cross-linked silicone coatings, and silicone oil-free syringe systems are reshaping the competitive landscape. Asia-Pacific is emerging as a strategic manufacturing hub due to increasing pharmaceutical production capacity, lower manufacturing costs, and expanding healthcare investments.
Market Size & Forecast
2025 Market Size: USD 0.22 Billion
2026 Projected Market Size: USD 0.25 Billion
2032 Projected Market Size: USD 0.52 Billion
CAGR (2026-2032): 12.92%
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Key Market Report Takeaways
North America accounted for the largest market share of approximately 38% due to advanced biologics manufacturing, strong pharmaceutical R&D spending, and high adoption of injectable drug delivery systems.
Asia-Pacific is projected to register the fastest CAGR during the forecast period owing to rapid pharmaceutical manufacturing expansion in China, India, and South Korea.
Silicone oil-based lubrication systems held the highest market share due to their widespread compatibility with conventional prefilled syringe manufacturing lines.
Silicone oil-free lubrication technologies are expected to witness the fastest growth because of increasing demand for low-particle and biologic-compatible delivery systems.
Glass prefillable syringes remain the dominant product segment owing to established pharmaceutical acceptance and superior barrier properties.
Biologics and vaccines represented the leading application segment due to rising injectable biologic approvals globally.
Pharmaceutical and biotechnology companies accounted for the largest end-use segment driven by increasing production of injectable therapies and self-administration devices.
Increasing adoption of auto-injectors and wearable drug delivery devices is accelerating demand for advanced low-friction lubrication technologies.
Details about the report and current availability can be viewed : https://www.databridgemarketresearch.com/reports/global-prefillable-syringe-silicone-lubrication-systems-market
Market Trends
Key Market Trends & Highlights
North America remains the leading regional market due to strong biologics manufacturing infrastructure, high adoption of advanced injectable therapies, and the presence of major pharmaceutical packaging companies.
Asia-Pacific is the fastest-growing region supported by expanding fill-finish manufacturing facilities, increasing healthcare investments, and growing biosimilar production in China and India.
Silicone oil-free syringe systems are gaining significant traction for ophthalmic drugs, gene therapies, and biologics due to reduced particle contamination and improved drug stability.
Rising demand for self-administration devices, including auto-injectors and wearable injectors, is driving innovation in low-friction and precision lubrication technologies.
Advanced coating technologies such as baked-on silicone and fluoropolymer-coated plungers are reshaping syringe compatibility for high-viscosity biologics.
Regulatory focus on extractables, leachables, and particulate contamination is encouraging pharmaceutical manufacturers to adopt premium lubrication systems and high-performance syringe platforms.
Market Dynamics
Market Drivers
Rising Demand for Biologics and Injectable Therapies
The increasing global use of biologics, monoclonal antibodies, vaccines, and gene therapies is significantly driving demand for advanced prefillable syringe lubrication systems. These therapies require precise dosing, high drug stability, and minimal particulate contamination. North America and Europe continue to lead biologics adoption due to robust pharmaceutical innovation pipelines. The growing prevalence of chronic diseases such as diabetes, autoimmune disorders, and cancer is further expanding injectable drug demand globally.
Expansion of Self-Administration Drug Delivery Devices
The rapid adoption of auto-injectors, pen injectors, and wearable injectors is accelerating market growth. Pharmaceutical companies are increasingly developing patient-centric drug delivery systems to support home healthcare and self-administration trends. Advanced silicone lubrication technologies improve injection smoothness, reduce break-loose force variability, and enhance patient comfort. This trend is particularly strong in the U.S., Germany, Japan, and other developed healthcare markets.
Technological Advancements in Silicone-Free and Low-Particle Systems
Growing concerns regarding silicone-induced protein aggregation and particle contamination are driving innovation in silicone oil-free syringe technologies. Manufacturers are investing in fluoropolymer coatings, baked-on silicone technologies, and advanced surface treatments to improve biologic compatibility. These technologies are increasingly adopted in ophthalmic drugs, mRNA therapeutics, and sensitive biologic formulations. Innovation-driven competition is strengthening product differentiation across global suppliers.
Growth in Pharmaceutical Fill-Finish Manufacturing Capacity
Global pharmaceutical companies are expanding sterile fill-finish production capabilities to meet rising injectable drug demand. Increased investments in manufacturing facilities across North America, Europe, China, and India are creating sustained demand for prefillable syringe components and lubrication systems. Contract development and manufacturing organizations (CDMOs) are also scaling injectable production capacity. The expansion of biologics manufacturing ecosystems continues to support long-term market growth.
Regulatory Emphasis on Drug Safety and Product Integrity
Regulatory agencies are increasingly emphasizing container closure integrity, extractables and leachables testing, and particulate reduction in injectable drug delivery systems. This is encouraging pharmaceutical manufacturers to adopt premium lubrication systems with higher consistency and lower contamination risks. Compliance with FDA, EMA, and USP standards is driving demand for technologically advanced syringe platforms. Companies are prioritizing quality-focused lubrication solutions to accelerate regulatory approvals.
Increasing Adoption of Ready-to-Fill Syringe Platforms
Ready-to-fill (RTF) syringe systems are witnessing growing demand due to their operational efficiency, sterility assurance, and reduced manufacturing complexity. Silicone lubrication technologies play a critical role in maintaining syringe functionality and fill-finish performance. Pharmaceutical companies are increasingly adopting pre-sterilized syringe systems to reduce production downtime and contamination risks. This trend is especially prominent in biologics and vaccine manufacturing applications.
Market Restraints
High Cost of Advanced Silicone-Free Technologies
Silicone oil-free syringe systems and advanced coating technologies involve higher manufacturing and validation costs compared to traditional silicone lubrication systems. Specialized coatings, precision engineering, and cleanroom production environments increase overall product pricing. Small and mid-sized pharmaceutical companies may face budget constraints in adopting premium syringe systems. Cost sensitivity remains particularly significant in emerging markets.
Stringent Regulatory Validation Requirements
Advanced lubrication systems require extensive compatibility testing, extractables and leachables studies, and long-term stability validation before commercialization. Regulatory approval timelines can be lengthy and resource-intensive, particularly for biologics applications. Compliance with evolving FDA and EMA standards increases operational complexity for manufacturers. These regulatory barriers can delay product launches and increase development expenditures.
Risk of Drug Interaction and Protein Aggregation
Traditional silicone oil lubrication systems may interact with sensitive biologics, potentially leading to protein aggregation or particulate formation. Such risks can impact drug efficacy, patient safety, and regulatory approval outcomes. Pharmaceutical companies remain cautious regarding syringe-drug compatibility for advanced biologics. This challenge is driving transition costs toward alternative lubrication technologies.
Supply Chain Volatility in Pharmaceutical Packaging Materials
The market remains vulnerable to disruptions in the supply chain for pharmaceutical-grade glass, elastomers, specialty coatings, and polymer materials. Geopolitical tensions, logistics disruptions, and raw material shortages can affect manufacturing continuity. Supply constraints may increase lead times and production costs across the syringe ecosystem. Europe and North America experienced intermittent supply challenges following pandemic-driven pharmaceutical demand surges.
Competitive Pricing Pressure from Regional Manufacturers
The presence of low-cost regional suppliers, particularly in Asia-Pacific, is intensifying pricing competition across standard silicone lubrication products. Global manufacturers face pressure to maintain margins while investing heavily in innovation and compliance. Price competition is especially intense in conventional syringe systems serving generic injectable drugs. This may limit profitability for premium solution providers in cost-sensitive markets.
Market Opportunities
Expansion of Silicone Oil-Free Syringe Platforms
The increasing demand for biologics, ophthalmic therapies, and gene therapies creates significant opportunities for silicone oil-free syringe technologies. Pharmaceutical companies are prioritizing low-particle delivery systems to improve biologic stability and reduce immunogenicity risks. Advanced coatings and polymer-based syringe systems are expected to generate substantial revenue opportunities through 2032. Innovation-led differentiation will remain a key competitive advantage.
Growth of Biologics and Biosimilars in Emerging Markets
Asia-Pacific and Latin America are experiencing strong growth in biologics manufacturing and biosimilar commercialization. Expanding pharmaceutical production facilities in China, India, and Brazil are increasing demand for advanced syringe lubrication technologies. Government support for domestic biologics production is further accelerating market penetration. Suppliers with localized manufacturing strategies are positioned to benefit significantly.
Rising Demand for Wearable Injectors and High-Volume Drug Delivery
The transition toward large-volume subcutaneous biologic delivery is creating demand for advanced syringe and cartridge lubrication systems. Wearable injectors require highly consistent glide force and compatibility with viscous drug formulations. Manufacturers are developing specialized coatings and low-friction technologies to support these applications. This segment is expected to become a high-value growth area for premium suppliers.
Strategic Partnerships Between Pharma and Packaging Companies
Collaborations between pharmaceutical companies, CDMOs, and syringe component manufacturers are increasing to accelerate development timelines and ensure compatibility for complex injectable drugs. Joint innovation programs are supporting customized lubrication solutions for specific biologic formulations. Strategic partnerships also improve supply chain reliability and long-term commercial contracts. These collaborations are strengthening market consolidation trends.
Increasing Investments in Injectable Drug Manufacturing Infrastructure
Governments and pharmaceutical companies are investing heavily in sterile injectable manufacturing capacity across North America, Europe, and Asia-Pacific. The expansion of vaccine manufacturing, biologics production, and fill-finish operations is creating strong demand for advanced syringe systems. New facilities increasingly prioritize automation-compatible and contamination-controlled lubrication technologies. Long-term injectable drug demand will continue supporting market expansion.
Market Challenges
Complex Compatibility Requirements for Sensitive Biologics
Biologics and advanced therapeutics exhibit high sensitivity to syringe materials, coatings, and lubrication agents. Ensuring long-term compatibility without aggregation or degradation remains technically challenging. Manufacturers must conduct extensive formulation-specific testing, increasing development complexity and commercialization timelines. This challenge is particularly critical in ophthalmic biologics and mRNA therapies.
Operational Complexity in Precision Coating Technologies
Producing consistent silicone coating thickness and lubrication performance across high-volume syringe manufacturing lines requires sophisticated automation and process controls. Variability in coating application can impact syringe functionality and drug safety. Manufacturers must invest heavily in advanced quality assurance systems and precision engineering. Maintaining production consistency at scale remains a significant operational challenge.
Regulatory Scrutiny on Extractables and Particulates
Regulators are increasingly focusing on particulate contamination, extractables, and leachables associated with syringe lubrication systems. Compliance requires extensive analytical testing and continuous quality monitoring. Evolving global standards create uncertainty for manufacturers operating across multiple regulatory jurisdictions. Compliance-related costs continue to rise across the industry.
Global Supply Chain and Raw Material Constraints
The market depends on reliable access to pharmaceutical-grade glass, specialty polymers, silicone materials, and elastomer components. Supply chain disruptions, geopolitical instability, and inflationary pressures can significantly impact production costs and delivery timelines. Shortages in pharmaceutical packaging materials may affect capacity expansion strategies. Regional supply imbalances remain a persistent challenge.
Intense Innovation and Competitive Pressure
The market is becoming increasingly competitive as global leaders and regional manufacturers introduce differentiated syringe technologies. Companies must continuously invest in R&D to maintain technological leadership and regulatory compliance. Innovation cycles are shortening, increasing pressure on profitability and commercialization speed. Emerging players offering cost-effective alternatives are intensifying market fragmentation.
Unlock Your Offer :https://www.databridgemarketresearch.com/checkout/buy/global-prefillable-syringe-silicone-lubrication-systems-market/compare-licence
Market Segmentation & Analysis
By Product Type
Silicone Oil-Based Lubrication Systems
Silicone oil-based systems represent the largest market segment due to their widespread use in conventional glass prefillable syringes. These systems provide reliable lubrication performance and compatibility with existing fill-finish infrastructure. The segment accounted for over 60% of market revenue in 2025 owing to broad pharmaceutical acceptance. Demand remains strong in vaccines, insulin delivery, and standard injectable therapies. However, growth is gradually moderating due to increasing concerns over particle contamination and protein aggregation.
Silicone Oil-Free Lubrication Systems
Silicone oil-free systems are projected to register the fastest CAGR through 2032 due to growing adoption in biologics and ophthalmic applications. These systems minimize particulate formation and improve compatibility with sensitive formulations. Advanced fluoropolymer coatings and baked-on lubrication technologies are supporting rapid adoption. Pharmaceutical companies increasingly prefer silicone-free platforms for gene therapies, mRNA drugs, and high-value biologics. North America and Europe remain key adoption centers for this segment.
By Material Type
Glass Prefillable Syringes
Glass syringes dominate the market due to superior barrier properties, established regulatory acceptance, and compatibility with a broad range of injectable drugs. The segment continues to hold the largest market share globally. Pharmaceutical manufacturers prefer glass syringes for vaccines, biologics, and insulin therapies. Advanced lubrication technologies are increasingly integrated into premium glass syringe platforms to improve functionality and drug stability.
Polymer Prefillable Syringes
Polymer syringes are expected to witness the fastest growth due to break resistance, lightweight characteristics, and compatibility with advanced biologics. Cyclic olefin polymer (COP) and cyclic olefin copolymer (COC) syringes are increasingly used in mRNA vaccines and ophthalmic drugs. These syringes require advanced low-friction lubrication technologies to ensure functionality. Demand is rising rapidly in wearable injectors and home healthcare applications.
By Application
Biologics and Biosimilars
Biologics remain the dominant application segment due to increasing approvals of injectable monoclonal antibodies and protein therapeutics. Advanced lubrication systems help minimize aggregation risks and ensure precise dosing. Pharmaceutical companies prioritize high-performance syringe systems for biologics commercialization. The segment is expected to maintain strong double-digit growth throughout the forecast period.
Vaccines
Vaccines represent a significant application segment driven by global immunization programs and pandemic preparedness initiatives. Prefillable syringes offer operational efficiency, reduced contamination risk, and accurate dosing for vaccine administration. Demand remains particularly strong in North America and Europe. Advanced lubrication technologies improve syringe functionality during high-volume manufacturing.
Ophthalmic Drugs
The ophthalmic segment is emerging rapidly due to increasing adoption of silicone oil-free syringe technologies. Sensitive ophthalmic biologics require ultra-low particle systems and precise micro-volume dosing capabilities. Manufacturers are developing specialized lubrication solutions for intravitreal injections. The segment is expected to register one of the highest CAGRs through 2032.
By End User
Pharmaceutical & Biotechnology Companies
Pharmaceutical and biotechnology companies account for the largest market share due to increasing injectable drug development activities. These companies demand high-quality lubrication systems to ensure product stability, compliance, and manufacturing efficiency. Growing biologics pipelines are driving sustained adoption globally. North America remains the leading regional market for this segment.
Contract Development and Manufacturing Organizations (CDMOs)
CDMOs are witnessing rapid growth due to increasing outsourcing of injectable drug manufacturing and fill-finish operations. These organizations require scalable, automation-compatible syringe systems for multi-client operations. Demand is particularly strong in Asia-Pacific and Europe. Strategic investments in sterile manufacturing capacity are accelerating segment growth.
Regional Analysis
By geography, the market is categorized into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America represents the largest regional market, accounting for approximately 38% of global revenue in 2025. The region benefits from strong biologics manufacturing infrastructure, high injectable drug adoption, and advanced pharmaceutical packaging technologies. The U.S. remains the dominant contributor due to the presence of leading pharmaceutical companies and syringe technology providers. Strong FDA regulatory standards and extensive R&D investments continue supporting market leadership. Expansion of self-injection therapies and biologics manufacturing further strengthens regional demand.
Europe
Europe is a mature and steadily growing market supported by strong pharmaceutical innovation and regulatory compliance standards. Germany, the U.K., and France remain key regional contributors due to advanced healthcare infrastructure and biologics production capabilities. European pharmaceutical manufacturers are increasingly investing in silicone oil-free syringe technologies to address regulatory and patient safety concerns. Strong emphasis on sustainability and premium drug delivery systems supports long-term growth. The region also benefits from the presence of major syringe packaging manufacturers.
Asia Pacific
Asia Pacific is projected to register the fastest CAGR during the forecast period due to rapid pharmaceutical manufacturing expansion and increasing healthcare investments. China, India, and Japan are major growth markets driven by biologics production, biosimilar development, and expanding injectable drug demand. Government incentives supporting domestic pharmaceutical manufacturing are accelerating market growth. Lower manufacturing costs and expanding CDMO activities are attracting global investments. Increasing healthcare access and chronic disease prevalence further support regional expansion.
Latin America
Latin America is an emerging market with gradual adoption of advanced injectable drug delivery technologies. Brazil and Mexico remain the leading contributors due to improving healthcare infrastructure and increasing pharmaceutical investments. Market growth is supported by rising vaccine production and biosimilar adoption. However, economic volatility and limited high-end manufacturing infrastructure continue to constrain rapid expansion. Regional demand for cost-effective syringe systems remains strong.
Middle East & Africa
The Middle East & Africa market is experiencing steady but slower growth compared to other regions. Government healthcare modernization initiatives and increasing pharmaceutical imports are supporting market development. Gulf countries are investing in healthcare infrastructure and localized pharmaceutical manufacturing. However, limited advanced manufacturing ecosystems and regulatory inconsistencies continue to challenge market penetration. Long-term growth opportunities remain linked to healthcare investment and injectable drug adoption.
Key Insights:
Largest Region: North America
Fastest Growing Region: Asia Pacific
Competitive Landscape
Market Structure Overview
The prefillable syringe silicone lubrication systems market is moderately consolidated, with competition led by a mix of global pharmaceutical packaging companies, specialized component manufacturers, and regional suppliers. Leading companies compete based on technological innovation, product quality, regulatory compliance, and global manufacturing capabilities. The market is witnessing increasing investment in silicone oil-free technologies, biologics-compatible systems, and advanced coating platforms. Competitive landscape analysis helps stakeholders understand technological positioning, innovation intensity, and strategic expansion initiatives across the industry.
Key Industry Players
Major market participants focus on expanding advanced syringe platforms, strengthening biologics compatibility, and improving low-particle delivery technologies. Global leaders maintain competitive advantage through broad product portfolios, integrated manufacturing capabilities, and strong pharmaceutical partnerships. Companies are increasingly investing in automation, cleanroom manufacturing, and next-generation coating technologies to strengthen differentiation.
List of Key Industry Players
West Pharmaceutical Services, Inc.
Gerresheimer AG
SCHOTT Pharma
Datwyler Holding Inc.
Becton, Dickinson and Company (BD)
Stevanato Group
Nipro Corporation
AptarGroup, Inc.
Terumo Corporation
Taisei Medical Co., Ltd.
Competitive Strategies
Companies are actively pursuing product innovation, silicone-free technologies, and strategic collaborations with pharmaceutical manufacturers to strengthen market position. Partnerships with biologics developers and CDMOs are becoming increasingly important for customized syringe platform development. Mergers, acquisitions, and regional manufacturing expansions are supporting supply chain resilience and market penetration. Firms are also investing heavily in automation and analytics-driven manufacturing to improve operational efficiency and product consistency. Geographic expansion into Asia-Pacific remains a major strategic priority.
Emerging Players & Market Dynamics
Emerging players and niche innovators are intensifying competition by offering specialized coating technologies, polymer syringe platforms, and cost-effective lubrication solutions. Startups focused on biologics-compatible delivery systems are disrupting traditional syringe manufacturing approaches. Increasing funding activity in drug delivery technologies is accelerating innovation across the market. Digital manufacturing and smart quality-control systems are becoming critical competitive differentiators. The market is expected to witness continued technological transformation through 2032.
Latest Developments
September 2024 - Gerresheimer AG: Introduced innovative silicone oil-free prefillable syringe systems for ophthalmic drugs at CPHI Worldwide 2024. The systems reduce particle load and improve compatibility for sensitive biologics, strengthening the company's position in advanced injectable delivery solutions.
March 2024 - SCHOTT Pharma: Announced a USD 371 million investment in a new U.S. prefillable syringe manufacturing facility in North Carolina. The facility will support rising demand for GLP-1 therapies, biologics, and mRNA-related injectable delivery systems.
October 2024 - Datwyler Holding Inc.: Expanded its NeoFlex coated plunger portfolio for large-volume biologics and wearable injectors. The development supports silicone-free functionality and improved compatibility with high-viscosity biologic formulations.
October 2024 - West Pharmaceutical Services: Presented advanced prefillable syringe component technologies and large-volume delivery solutions at the PDA Universe of Pre-Filled Syringes Conference. The company highlighted innovations addressing low-temperature performance and combination product compatibility.
January 2024 - Gerresheimer AG: Launched silicone oil-free prefillable syringe systems specifically designed for sensitive ophthalmic molecules, RNAi therapies, and gene therapies. The development reflects increasing industry focus on low-particle injectable delivery technologies.
2024 - Pharmaceutical Industry Expansion: Multiple pharmaceutical packaging companies increased investments in ready-to-fill syringe manufacturing capacity to support growing demand for biologics and self-administration therapies globally. Expansion activities were particularly strong across North America and Asia-Pacific.
2024 - Industry-Wide Technology Shift: Manufacturers accelerated development of baked-on silicone and fluoropolymer-coated syringe technologies to reduce extractables, improve glide performance, and enhance biologic compatibility. This trend is reshaping competitive differentiation across premium syringe systems.
check out more related studies published by data bridge market research:
https://www.databridgemarketresearch.com/reports/global-telecom-power-system-market
https://www.databridgemarketresearch.com/reports/global-aws-managed-services-market
https://www.databridgemarketresearch.com/reports/north-america-aws-managed-services-market
https://www.databridgemarketresearch.com/reports/europe-aws-managed-services-market
https://www.databridgemarketresearch.com/reports/middle-east-africa-aws-managed-services-market
https://www.databridgemarketresearch.com/reports/global-managed-file-transfer-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
Data Bridge Market Research deliver objective and actionable insights, crafted to meet the unique needs of our clients. Our database features thousands of statistics and in-depth analyses on 200 plus industries and more than 5000 markets in 75 major countries globally. Additionally, DBMR has served for more than 40% of Fortune 500 firms internationally and has a more than 3000 client's network.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Prefillable Syringe Coating and Lubrication Systems Market - Competitive Landscape and Growth Forecast here
News-ID: 4505708 • Views: …
More Releases from Data Bridge Market Research
Soy Protein Isolate Industry Forecast 2026-2032: Market to Witness 9.10% CAGR Gr …
Market Summary
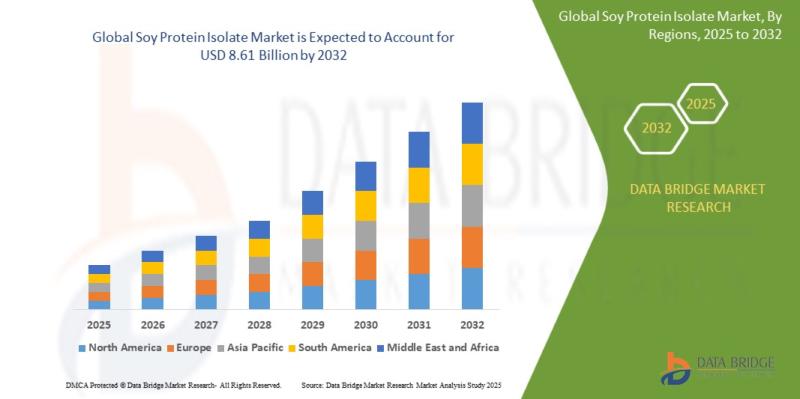
As per Data Bridge Market Research analysis, the Soy Protein Isolate Market was estimated at USD 4.68 billion in 2025. The market is expected to grow from USD 5.10 billion in 2026 to USD 8.61 billion in 2032, at a CAGR of 9.10% during the forecast period with driven by the rising demand for plant-based protein ingredients, increasing adoption of functional foods and beverages, growing vegan and flexitarian consumer…
Cloud Security and Zero-Trust Adoption Accelerate Big Data Security Market at 12 …
Market Summary
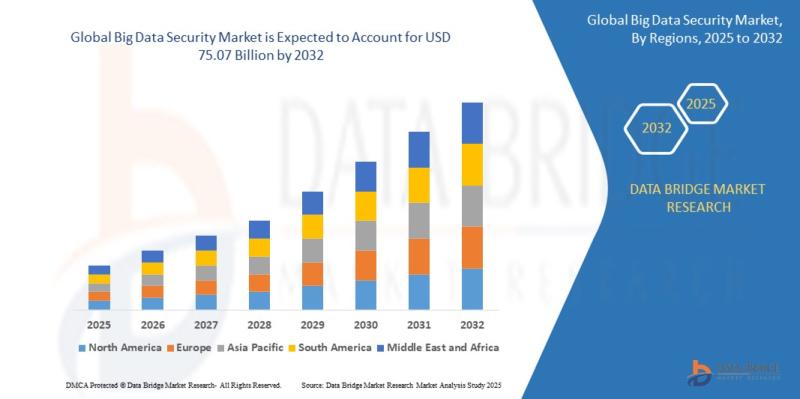
"As per Data Bridge Market Research analysis, the Global Big Data Security Market was estimated at USD 32.10 billion in 2025. The market is expected to grow from USD 36.24 billion in 2026 to USD 75.07 billion in 2032, at a CAGR of 12.90% during the forecast period with driven by the rising demand for advanced cybersecurity solutions, increasing enterprise adoption of cloud-based big data platforms, growing frequency of…
Sustainable and Premium Packaging Trends Propel Skincare Packaging Market at 6.2 …
Market Summary
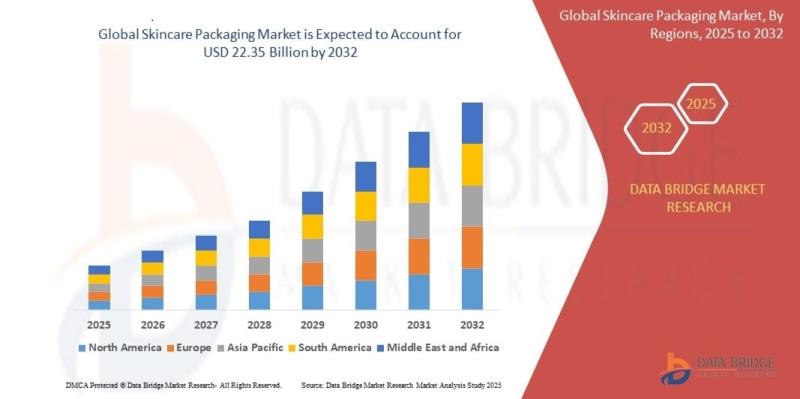
"As per Data Bridge Market Research analysis, the Global Skincare Packaging Market was estimated at USD 15.08 billion in 2025. The market is expected to grow from USD 16.01 billion in 2026 to USD 22.35 billion in 2032, at a CAGR of 6.2% during the forecast period with driven by the rising demand for sustainable and premium skincare packaging solutions, increasing adoption of recyclable and refillable materials, rapid expansion…
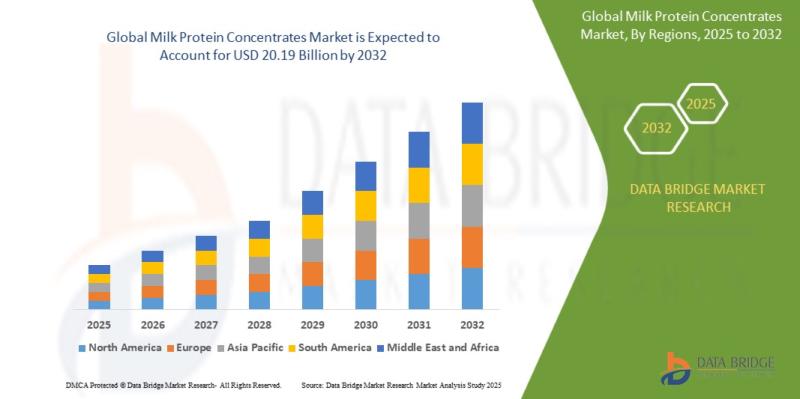
Milk Protein Concentrates Market Size to Reach USD 20.19 Billion by 2032 | Growi …
As per Data Bridge Market Research analysis, the Milk Protein Concentrates Market was estimated at USD 14.35 billion in 2025. The market is expected to grow from USD 15.07 billion in 2026 to USD 20.19 billion in 2032, at a CAGR of 5.0% during the forecast period with driven by the rising demand for high-protein functional foods, expanding sports and clinical nutrition applications, increasing health-conscious consumer preferences, and advancements in…
More Releases for America
Stabilit America Highlights Applications of Fiberglass Roof Panels with Stabilit …
Roofing materials are very important in the realm of modern construction, as they should be long lasting, economical and attractive. Fiberglass roof panels are a few of the numerous choices among several alternatives that have received a reputation of being versatile, long life, and adaptable in various sectors. They are favored by the architects, contractors, and property developers due to their lightweight construction, resistance to weather factors, and the ease…
Deodorants Market Report by Region (North America, EMEA, Latin America, Asia)
2025 - Pristine Market Insights, a leading market research firm, announced the release of its latest and comprehensive market research report on Deodorants market. The report spans over 500 pages and delivers 10-year market forecast in US dollars (or custom currencies upon request). It provides in-depth analysis of market dynamics (drivers, opportunities, restraints), PESTLE insights, latest industry trends, and demand factors. The report includes segmented market value, share (%), compound…
Sequestrant Market Report by Region (North America, EMEA, Latin America, Asia)
2025 - Pristine Market Insights, a leading market research firm, announced the release of its latest and comprehensive market research report on Sequestrant market. The report spans over 500 pages and delivers 10-year market forecast in US dollars (or custom currencies upon request). It provides in-depth analysis of market dynamics (drivers, opportunities, restraints), PESTLE insights, latest industry trends, and demand factors. The report includes segmented market value, share (%), compound…
Buttermilk Market Study by Region (North America, Latin America, Europe, Asia, M …
2025 - Pristine Market Insights, a leading market research firm, announced the release of its latest and comprehensive market research report on Buttermilk market. The report spans over 500 pages and delivers 10-year market forecast in US dollars (or custom currencies upon request). It provides in-depth analysis of market dynamics (drivers, opportunities, restraints), PESTLE insights, latest industry trends, and demand factors. The report includes segmented market value, share (%),…
Textiles Market Analysis Report, Regional Outlook - Europe, North America, South …
Adroit Market Research has announced the addition of the “Global Textiles Market Size Status and Forecast 2025”, The report classifies the global Textiles in a precise manner to offer detailed insights about the aspects responsible for augmenting as well as restraining market growth.
This report studies the global Textiles Speaker market, analyzes and researches the Textiles Speaker development status and forecast in Europe, North America, Central America, South America, Asia Pacific…
Global Gaucher Disease Market 2018 Covering North America, South America, Europe
Gaucher Disease Market
Summary
The Global Gaucher Disease Market is defined by the presence of some of the leading competitors operating in the market, including the well-established players and new entrants, and the suppliers, vendors, and distributors. The key players are continuously focusing on expanding their geographic reach and broadening their customer base, in order to expand their product portfolio and come up with new advancements.
Gaucher Disease market size to maintain the average annual growth…