Press release
United States Bioprocess Validation Market to Grow at CAGR 11.20% Through 2033, Driven by Rising Biopharmaceutical Production and Increasing Regulatory Compliance Requirements
The Bioprocess Validation Market was growing at a CAGR of 11.20% during the forecast period 2026-2033. Growth is driven by increasing demand for biopharmaceuticals, vaccines, and personalized medicines worldwide. Rising regulatory requirements for product quality, safety, and compliance are accelerating the adoption of bioprocess validation solutions. Expanding investments in biotechnology and pharmaceutical manufacturing facilities are further supporting market expansion. Additionally, advancements in biologics production technologies and process automation are enhancing validation efficiency and reliability. Growing focus on reducing production risks and ensuring consistent product performance is also contributing to steady market growth.Download PDF Sample Report:
https://www.datamintelligence.com/download-sample/bioprocess-validation-market?prasad
Bioprocess Validation Market Major Players:
Cobetter Filtration Equipment Co., Ltd.
Cobetter Filtration Equipment is a specialized life sciences and filtration technology company with strong relevance in the bioprocess validation market through its single-use systems and biopharmaceutical filtration solutions. In 2025, the company reported revenue of USD 0.6 billion, driven by rising demand in biopharma manufacturing and biologics production. In the bioprocess validation market, Cobetter plays a key role by providing filtration validation services, sterile filtration systems, and single-use bioprocess components that ensure regulatory compliance, sterility assurance, and process consistency in biologics manufacturing. Its solutions are widely used in vaccine production, cell culture processing, and biopharmaceutical purification workflows.
SGS S.A.
SGS is a global leader in testing, inspection, and certification services, with strong involvement in bioprocess validation through its pharmaceutical and life sciences assurance services. In 2025, the company reported revenue of USD 7.2 billion, driven by industrial services, certification, and quality assurance segments. In the bioprocess validation market, SGS plays a major role by offering validation services for cleanrooms, equipment qualification (IQ/OQ/PQ), process validation, and regulatory compliance testing. Its expertise supports pharmaceutical and biotech companies in ensuring product safety, process reliability, and adherence to global regulatory standards.
DOC S.r.l.
DOC S.r.l. is a specialized life sciences engineering company focused on bioprocess validation, cleanroom qualification, and pharmaceutical compliance services. In 2025, the company reported revenue of USD 0.1 billion, reflecting its niche focus in biopharma validation services. In the bioprocess validation market, DOC S.r.l. plays a key role by providing validation protocols, process engineering support, and GMP-compliant documentation services for biopharmaceutical manufacturing facilities. Its services are widely used in biologics production, vaccine manufacturing, and sterile drug development environments where regulatory validation is critical.
Thermo Fisher Scientific Inc.
Thermo Fisher Scientific is a global leader in life sciences, laboratory equipment, and bioprocessing solutions, with a strong presence in the bioprocess validation market. In 2025, the company reported revenue of USD 45.0 billion, driven by its bioproduction, analytical instruments, and laboratory products segments. In the bioprocess validation market, Thermo Fisher plays a major role by offering single-use bioprocess systems, validation services, analytical tools, and GMP-compliant solutions that support end-to-end biopharmaceutical manufacturing. Its technologies are widely used in biologics, vaccine production, and cell and gene therapy manufacturing where process validation and regulatory compliance are essential.
Find the Right Partner to Accelerate Your Growth Today with Our Latest Report:
https://www.datamintelligence.com/partner-identification-enquiry/bioprocess-validation-market?prasad
United States: Recent Developments
✅ In April 2026, Thermo Fisher Scientific expanded its bioprocess validation solutions with advanced automated process monitoring systems for biologics manufacturing. The upgrades improve batch consistency and regulatory compliance in large-scale production. Thermo Fisher continues strengthening biopharma process reliability.
✅ In March 2026, Sartorius Stedim North America enhanced its bioprocess validation platforms with real-time analytics tools for upstream and downstream process verification in cell and gene therapy production. The development improves manufacturing precision. Sartorius continues advancing bioprocessing technologies.
✅ In February 2026, Danaher Corporation (Cytiva) improved its bioprocess validation workflows with integrated digital twin modeling for biologics scale-up and quality assurance. The innovation reduces production variability and accelerates validation cycles. Danaher continues expanding life sciences manufacturing solutions.
✅ In January 2026, Merck Millipore strengthened its bioprocess validation services with enhanced viral clearance and filtration validation technologies for monoclonal antibody production. The upgrade ensures higher regulatory compliance standards. Merck continues supporting biologics manufacturing excellence.
Japan: Recent Developments
✅ In April 2026, Takeda Pharmaceutical advanced its internal bioprocess validation systems with AI-driven process monitoring for biologics and vaccine production. The focus is on improving yield consistency and regulatory compliance. Takeda continues enhancing biomanufacturing efficiency.
✅ In March 2026, FUJIFILM Diosynth Biotechnologies Japan expanded its bioprocess validation capabilities with advanced cell culture monitoring and scale-up verification technologies. The improvement supports global biologics supply expansion. Fujifilm continues strengthening contract manufacturing leadership.
✅ In February 2026, Ajinomoto Bio-Pharma Services Japan upgraded its bioprocess validation systems with enhanced quality-by-design frameworks for peptide and biologics production. The development improves manufacturing robustness. Ajinomoto continues advancing biopharmaceutical production technologies.
✅ In January 2026, Shimadzu Corporation enhanced its analytical bioprocess validation tools with improved chromatography and real-time monitoring systems for pharmaceutical manufacturing. The upgrade improves process traceability and compliance. Shimadzu continues supporting precision bioprocess analytics.
Segment Covered in the Bioprocess Validation Market:
By Test Type: Extractables & Leachables Testing Services
Extractables and leachables (E&L) testing services dominate the bioprocess validation market due to their critical role in ensuring the safety, quality, and regulatory compliance of biopharmaceutical products. These tests are essential for evaluating chemical interactions between single-use systems, packaging materials, and biologics during production and storage.
This segment accounts for approximately 33% of the total market. Based on a global bioprocess validation market size assumption of USD 5.4 billion in 2025, this translates to an estimated segment value of USD 1.8 billion. Growth is driven by increasing biologics production, stricter regulatory requirements, and rising adoption of single-use bioprocessing systems.
By Process: Media Containers and Bags
Media containers and bags dominate the process segment due to their widespread use in biopharmaceutical manufacturing for storage, transport, and handling of sterile media and buffers. These single-use systems require rigorous validation to ensure product safety and process integrity.
This segment accounts for approximately 38% of the total market. Based on the USD 5.4 billion market size in 2025, this corresponds to an estimated value of USD 2.1 billion. Growth is driven by expanding biologics production, increasing adoption of disposable technologies, and rising demand for contamination-free manufacturing systems.
By End-User: Pharmaceutical Companies
Pharmaceutical companies dominate the end-user segment due to their extensive use of validated bioprocess systems in drug development, production, and quality assurance processes. These companies require strict compliance with regulatory standards such as FDA and EMA guidelines.
This segment accounts for approximately 57% of the total market. Based on the USD 5.4 billion global market size in 2025, this corresponds to an estimated value of USD 3.1 billion. Growth is driven by increasing biologics pipelines, rising investment in biomanufacturing, and stringent regulatory compliance requirements.
Market Segmentation
The Bioprocess Validation Market is segmented by test type into extractables/leachables testing services, microbiological testing services, integrity testing services, physicochemical testing services, compatibility testing services, and others; by process into media containers and bags, freezing and thawing process bags, filter elements, and others; by end-user into pharmaceutical companies, biotechnology companies, and others; and by region into North America, Latin America, and Europe.
Get Customization in the report as per your requirements:
https://www.datamintelligence.com/customize/bioprocess-validation-market?prasad
Regional Analysis
North America
North America is the leading market due to strong biopharmaceutical manufacturing capacity, advanced regulatory frameworks, and high adoption of single-use technologies. The region is led by the United States.
The North American market is estimated at approximately USD 2.4 billion in 2025, accounting for about 44% of the global market. Growth is driven by expanding biologics production, strong presence of contract manufacturing organizations, and increasing regulatory scrutiny for bioprocess validation.
Europe
Europe is a significant market supported by strong pharmaceutical manufacturing infrastructure, increasing biologics production, and stringent quality standards enforced by regulatory authorities. Key contributors include Germany, France, and the United Kingdom.
The European market is valued at approximately USD 1.9 billion in 2025, representing about 35% of the global market. Growth is driven by rising adoption of single-use technologies, expanding biomanufacturing facilities, and strong regulatory compliance requirements.
Latin America
Latin America is an emerging market driven by increasing investments in pharmaceutical manufacturing, growing biotechnology sector development, and improving regulatory frameworks, with Brazil and Mexico as key contributors.
The market is estimated at approximately USD 1.1 billion in 2025, accounting for about 21% of the global market. Growth is supported by expansion of local biopharma production capabilities and rising adoption of quality assurance standards.
Data-Driven Research Methodology for the Bioprocess Validation Market:
Our research process for the Bioprocess Validation Market at DataM Intelligence combines rigorous primary and secondary research to ensure accurate and reliable insights. We engage with industry experts, key stakeholders, and validated data sources to capture real-time market dynamics. Advanced analytical tools and proprietary models are applied to forecast trends, competitive landscapes, and growth opportunities. Each report undergoes multi-level validation to deliver actionable intelligence that supports strategic decision-making.
Unlock Full 360° Strategic Report:
https://www.datamintelligence.com/buy-now-page?report=bioprocess-validation-market?prasad
✅ Competitive Landscape
✅ Technology Roadmap Analysis
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Consumer Behavior & Demand Analysis
✅ Import-Export Data Monitoring
✅ Live Market & Pricing Trends
Contact Us-
Fabian
DataM Intelligence 4market Research LLP
6th Floor, M2 Tech Hub, DataM Intelligence 4market Research LLP, Lalitha Nagar, Habsiguda, Secunderabad, Hyderabad, Telangana 500039
USA: +1 877-441-4866
UK: +44 161-870-5507
Email: fabian@datamintelligence.com
About Us
DataM Intelligence is a market research and consulting firm that delivers comprehensive end-to-end business solutions, covering everything from in-depth research to strategic consulting. The company leverages key industry trends, insights, and developments to provide fast, reliable, and actionable solutions tailored to diverse client requirements.
It offers both syndicated and customized research reports supported by a strong and robust methodology. With an extensive database comprising 9000+ reports across 40+ industry domains, DataM Intelligence serves over 200 companies in more than 50 countries, helping organizations access critical business intelligence that drives informed decision-making and sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release United States Bioprocess Validation Market to Grow at CAGR 11.20% Through 2033, Driven by Rising Biopharmaceutical Production and Increasing Regulatory Compliance Requirements here
News-ID: 4503414 • Views: …
More Releases from DataM intelligence 4 Market Research LLP
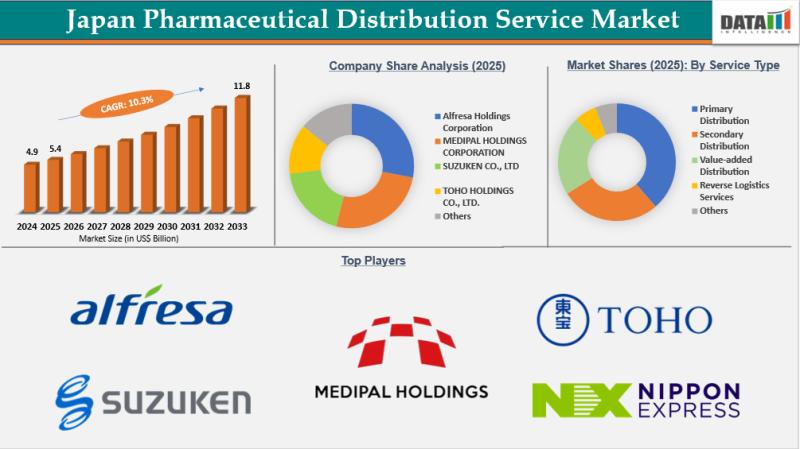
Japan Pharmaceutical Distribution Service Market Set to Record US$5.96 Billion i …
Japan pharmaceutical distribution service market reached US$5.40 Billion in 2025, rising to US$5.96 Billion in 2026 and is expected to reach US$11.83 Billion by 2033, growing at a CAGR of 10.3% from 2026 to 2033.
Download Free Sample Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/download-sample/japan-pharmaceutical-distribution-service-market?kb
Japan: Recent Industry Developments
✅ March 2026: Alfresa Holdings Corporation expanded its nationwide pharmaceutical distribution network, strengthening cold-chain infrastructure for temperature-sensitive drugs.
✅ February 2026: Suzuken Co.,…

United States Gut Health Supplements Market Set to Reach USD 6.72 Billion by 203 …
The global gut health supplements market reached US$ 13,931.6 million in 2025 and is expected to reach US$ 22,407.2 million by 2033, growing at a CAGR of 6.12% during the forecast period 2026-2033. Growth is driven by increasing consumer awareness regarding digestive health and overall wellness. Rising prevalence of gastrointestinal disorders and lifestyle-related health issues is boosting demand for gut health supplements. Growing interest in probiotics, prebiotics, and functional nutrition…

United States Carbonated Probiotic Drink Market Set to Reach USD 1.33 Billion by …
The Global Carbonated Probiotic Drink Market reached US$ 3.03 billion in 2025 and is expected to reach US$ 4.44 billion by 2033, growing with a CAGR of 5.6% during the forecast period 2026-2033. Growth is driven by increasing consumer awareness regarding gut health and digestive wellness. Rising demand for functional beverages with probiotic benefits is accelerating market adoption worldwide. Growing preference for healthy alternatives to sugary carbonated drinks is further…

United States Mycorrhiza-Based Biofertilizer Market Set to Reach USD 930 Million …
The Mycorrhiza-Based Biofertilizer Market was valued at USD 1.24 billion in 2025 and is expected to reach USD 3.11 billion by 2033, growing at a CAGR of 12.16% during the forecast period 2026-2033. Growth is driven by increasing demand for sustainable and organic farming practices worldwide. Rising concerns regarding soil health degradation and excessive chemical fertilizer usage are accelerating the adoption of biofertilizers. The ability of mycorrhiza-based products to enhance…
More Releases for USD
Bone Cement Market Outlook USD 1,871.10M-USD 3,512.31M
How Is the Bone Cement Market Supporting the Rise of Modern Orthopedic Surgery?
The Bone Cement Market plays a critical role in modern orthopedic and spinal procedures, acting as a foundational material for joint replacement, fracture fixation, and vertebral stabilization. Bone cement is widely used to anchor implants, restore bone structure, and improve patient mobility-making it an essential component of musculoskeletal care.
In 2025, the global bone cement market was valued at…
Autologous Cell Therapy Market Outlook USD 9.31B-USD 54.83B
How Is the Autologous Cell Therapy Market Redefining the Future of Precision Medicine?
The Autologous Cell Therapy Market is rapidly emerging as one of the most transformative areas in modern healthcare, offering highly personalized treatment options for complex and chronic diseases. By using a patient's own cells to repair, replace, or regenerate damaged tissues, autologous cell therapy minimizes immune rejection risks while maximizing therapeutic effectiveness.
In 2025, the global autologous cell therapy…
US Ostomy Care and Accessories Market USD 4.03B-USD 6.75B
How Is the United States Ostomy Care and Accessories Market Evolving to Meet the Needs of a Growing Patient Population?
The United States Ostomy Care and Accessories Market plays a critical role in improving the quality of life for millions of patients who undergo life-altering surgical procedures involving the digestive or urinary systems. Ostomy care products are essential medical devices designed to manage bodily waste safely and discreetly following surgeries such…
PACS Market USD 5.59B in 2025, USD 9.73B by 2035
Picture Archiving and Communication System (PACS) Market Expands as Digital Imaging Transforms Global Healthcare
Introduction: PACS at the Core of Modern Medical Imaging
The healthcare industry is undergoing a rapid digital transformation, with medical imaging playing a critical role in diagnosis, treatment planning, and patient monitoring. At the heart of this transformation lies the Picture Archiving and Communication System (PACS)-a technology that enables the storage, retrieval, management, and sharing of medical images…
Global HEOR Market USD 1.70B-USD 6.03B
Health Economics and Outcomes Research (HEOR) Market Accelerates as Value-Based Healthcare Redefines Global Decision-Making
Introduction: The Growing Importance of HEOR in Modern Healthcare
The global healthcare industry is undergoing a profound transformation, shifting from volume-driven care models to value-based healthcare systems that prioritize patient outcomes, cost efficiency, and real-world effectiveness. At the center of this transformation lies Health Economics and Outcomes Research (HEOR)-a discipline that evaluates the economic value, clinical outcomes, and…
Foam Tape Market Outlook 2035: Industry Growth from USD USD 4.89 Billion (2025) …
The Foam Tape Market plays a vital role in modern industrial and manufacturing ecosystems. Foam tapes are pressure-sensitive adhesive products manufactured using materials such as polyurethane, polyethylene, PVC, and acrylic foam. These tapes are widely used for bonding, sealing, insulation, cushioning, vibration damping, and noise reduction across multiple industries. Their ability to replace traditional mechanical fasteners like screws, bolts, and rivets has positioned foam tapes as a preferred solution in…
