Press release
3D Bioprinting Moves Toward Clinical Reality: Advanced Biofabrication Systems Become Critical Infrastructure for Next-Generation Healthcare
NEWARK, DE / According to the latest analysis by Future Market Insights, the 3D Bioprinting Market is rapidly transitioning from an experimental capability to a clinically relevant and commercially scalable technology. As healthcare systems increasingly demand patient-specific solutions, regenerative therapies, and faster drug development pathways, 3D bioprinting is emerging as a foundational pillar in modern biomedical innovation.Once largely confined to research labs, 3D bioprinting is now being integrated into real-world medical workflows-from orthopedic implants and tissue engineering to pharmaceutical drug testing. This shift is driven by advancements in bioink formulations, multi-material printing capabilities, and improved precision, enabling the fabrication of complex, functional biological structures with vascularization potential.
In this evolving landscape, 3D bioprinting platforms-comprising hardware, bioinks, software, and services-are no longer optional tools but essential infrastructure supporting innovation across clinical, research, and pharmaceutical domains.
Read Full Report: https://www.futuremarketinsights.com/reports/3d-bio-printing-market
Quick Stats Snapshot
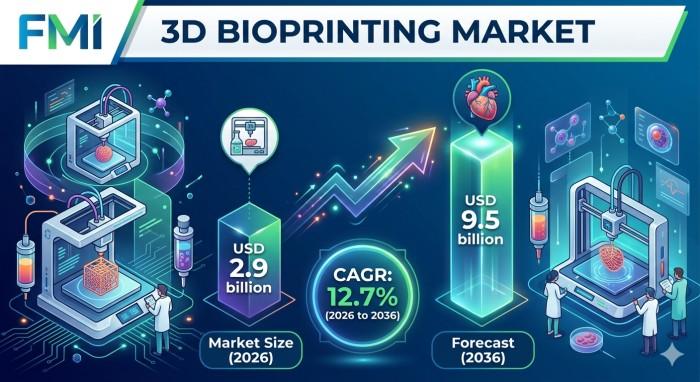
Market size (2026): USD 2.9 billion
Forecast (2036): USD 9.5 billion
CAGR (2026-2036): 12.7%
2025 baseline: USD 2.55 billion
Incremental opportunity: USD 6.63 billion
Leading application: Orthopedic implants (33.9% share)
Fastest-growing markets: Germany (14.9%), USA (14.1%)
Key end users: Hospitals, research institutions, pharma companies
Market Size and Forecast: From Experimental Innovation to Clinical Necessity
The 3D bioprinting market is entering a high-growth phase, projected to expand from USD 2.9 billion in 2026 to USD 9.5 billion by 2036. This growth reflects a structural transformation in healthcare delivery-where personalized medicine, regenerative therapies, and precision drug testing are becoming standard expectations.
As clinical validation improves and regulatory pathways mature, bioprinting is increasingly embedded across healthcare and life sciences workflows, driving sustained demand across multiple end-user segments.
Growth Drivers: Why Demand Is Accelerating
Advancements in Bioink and Multi-Material Printing: Innovations in bioink formulations and printing technologies are enabling the creation of complex tissue structures with enhanced biological functionality, accelerating the shift from research to clinical application.
Rising Demand for Patient-Specific Implants: Orthopedic and dental applications are driving adoption, as healthcare providers increasingly seek customized implants tailored to individual patient anatomy.
Pharmaceutical Industry Adoption: Bioprinted tissue models are transforming drug discovery by enabling more accurate toxicity testing and reducing reliance on animal models, thereby shortening development timelines.
Convergence of Technologies: The integration of additive manufacturing, biomaterials science, and regenerative medicine is unlocking new possibilities in tissue engineering and organ modeling.
Challenges: Where the Market Still Struggles
High Cost and Investment Requirements: Bioprinter hardware, bioinks, and regulatory compliance demand significant capital investment, limiting accessibility in cost-sensitive markets.
Regulatory Complexity: Approval pathways for bioprinted products remain evolving, requiring extensive clinical validation and documentation.
Technical Barriers: Developing functional, vascularized tissues requires multidisciplinary expertise, creating operational challenges for in-house adoption.
Opportunities: Where Strategic Value Is Emerging
Clinical Validation of Bioprinted Implants: Companies that can demonstrate successful clinical outcomes-especially in orthopedic and dental applications-are well positioned for commercialization.
Drug Discovery and Testing Platforms: Bioprinted tissue models offer scalable opportunities in pharmaceutical R&D, improving predictive accuracy and reducing costs.
Integrated Bioprinting Ecosystems: Solutions combining hardware, bioinks, and software into unified platforms are gaining traction among healthcare providers and research institutions.
Emerging Market Expansion: Countries with growing healthcare infrastructure and regulatory support present strong opportunities for adoption.
Segmentation Insights: How the Market Is Structured
By Technology: Extrusion-based and inkjet-based bioprinting dominate, with laser-assisted systems supporting high-resolution applications.
By Application: Orthopedic implants lead with 33.9% share, driven by demand for patient-specific solutions.
By End User: Hospitals and research institutions are primary users, with pharmaceutical companies and CROs showing increasing adoption.
By Product: Market includes bioprinters, bioinks, software, and services supporting end-to-end workflows.
Detailed market forecasts, competitive benchmarking, and service trends: https://www.futuremarketinsights.com/reports/sample/rep-gb-808
Regional Analysis: Where Growth Is Concentrated
Germany (14.9% CAGR): Strong biomedical infrastructure, advanced manufacturing capabilities, and supportive regulatory fr meworks position Germany as a global leader.
United States (14.1% CAGR): A mature research ecosystem, FDA advancements, and strong venture capital funding sustain market expansion.
China (13.9% CAGR): Rapid investment in healthcare innovation and research infrastructure is driving adoption.
India (13.0% CAGR): Growing healthcare demand and increasing focus on advanced medical technologies are supporting steady growth.
Japan (12.6% CAGR): A stable market emphasizing precision, quality, and long-term reliability in medical innovation.
Competitive Landscape: Innovation-Driven Positioning
The market is characterized by a mix of established players and emerging innovators competing on technology depth, product integration, and clinical validation.
Key participants include:
3D Systems, Inc
GE Healthcare (Concept Laser, Arcam AB)
EOS
Optomec
Renishaw plc.
ExOne
Organovo Holding, Inc
Strategic focus areas include product innovation, geographic expansion, and building long-term customer relationships through integrated solutions.
Strategic Implications for Decision-Makers
For Healthcare Providers: Bioprinting enables personalized treatment approaches, improving patient outcomes and surgical precision.
For Pharmaceutical Companies: Adoption of bioprinted models can significantly accelerate drug development and reduce costs.
For Investors: Companies with strong clinical validation and regulatory pathways are best positioned for long-term growth.
For Manufacturers: Integration of hardware, materials, and software into cohesive platforms will drive competitive advantage.
Future Outlook: Toward Scalable, Clinical-Grade Biofabrication
Over the next decade, the 3D bioprinting market is expected to evolve across three key dimensions:
Clinical Integration: Transition from experimental use to routine clinical application in implants and tissue engineering.
Regulatory Maturity: Development of standardized approval pathways to support commercialization.
Technology Convergence: Deeper integration of AI, biomaterials, and advanced manufacturing to enhance precision and scalability.
Unlock 360° insights for strategic decision making and investment planning: https://www.futuremarketinsights.com/checkout/808
Executive Takeaways
3D bioprinting is transitioning from research innovation to core healthcare infrastructure.
Market growth is driven by clinical demand, technological advancement, and pharmaceutical adoption.
Orthopedic implants remain the leading application segment.
Germany and the United States are key growth hubs, with strong global influence.
Competitive advantage lies in clinical validation, integrated platforms, and regulatory readiness.
For organizations navigating the future of healthcare innovation, the message is clear: the ability to fabricate living systems is no longer a distant vision-it is becoming a defining capability in modern medicine.
More Related Reports Form Future Market Insights (FMI)
Alcohol Use Disorder Treatment Market: https://www.futuremarketinsights.com/reports/alcohol-use-disorder-treatment-market
Coxsackievirus Infections Treatment Market: https://www.futuremarketinsights.com/reports/coxsackievirus-infections-treatment-market
Respiratory Pathogen Testing Kits Market: https://www.futuremarketinsights.com/reports/respiratory-pathogen-testing-market
DNA-RNA Extraction Market: https://www.futuremarketinsights.com/reports/dna-rna-extraction-market
Drug Eruptions Treatment Market: https://www.futuremarketinsights.com/reports/drug-eruptions-treatment-market
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware - 19713, USA
T: +1-347-918-3531
Website: https://www.futuremarketinsights.com
LinkedIn| Twitter| Blogs | YouTube
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release 3D Bioprinting Moves Toward Clinical Reality: Advanced Biofabrication Systems Become Critical Infrastructure for Next-Generation Healthcare here
News-ID: 4503337 • Views: …
More Releases from Future Market Insights
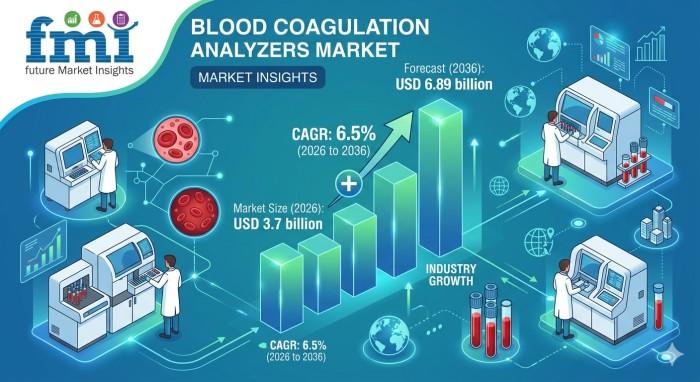
Blood Coagulation Testing Moves to the Clinical Frontline: Analyzer Systems Beco …
NEWARK, DE / According to the latest analysis by Future Market Insights, the Blood Coagulation Analyzers Market is transitioning from a routine laboratory function to a central pillar of clinical decision-making across hospital, laboratory, and point-of-care environments. As the global burden of cardiovascular diseases rises and surgical volumes increase, the need for rapid, accurate coagulation testing is intensifying.
Once confined to centralized laboratories, coagulation testing is now expanding into decentralized and…

AI Surgical Video Intelligence Moves to the Core of Operating Room Strategy: Str …
NEWARK, DE / According to the latest analysis by Future Market Insights, the AI Surgical Video Intelligence System Market is rapidly transitioning from a supplementary recording capability to a core component of digital operating room ecosystems. As hospitals and surgical centers shift toward data-driven care delivery, intraoperative video is no longer viewed as passive footage but as a structured, searchable, and analyzable clinical asset.
This transformation is being driven by the…
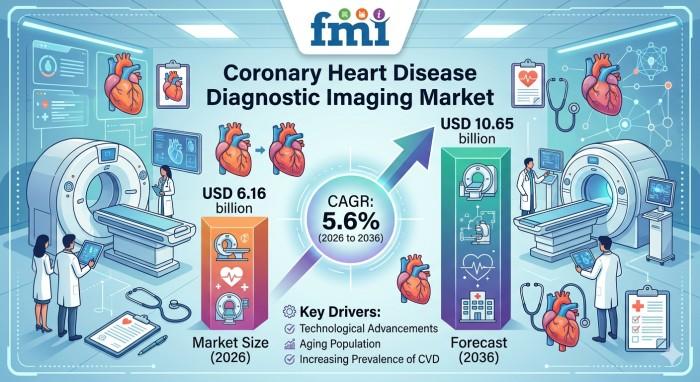
Coronary Heart Disease Diagnostic Imaging Market to Reach USD 10.65 Billion by 2 …
NEWARK, DE / - According to the latest analysis by Future Market Insights, the Coronary Heart Disease Diagnostic Imaging Market is entering a structurally evolving growth phase as diagnostic pathways shift toward earlier, imaging-led decision-making. The market, valued at USD 5.84 billion in 2025, is projected to reach USD 6.16 billion in 2026 and expand to USD 10.65 billion by 2036, reflecting a CAGR of 5.6%.
At the core of this…

Gene Editing Safety Moves to Center Stage: Off-Target Detection Systems Become C …
NEWARK, DE / According to the latest analysis by Future Market Insights, the Gene Editing Off-Target Effect Detection Test Systems Market is gaining strategic importance as gene editing moves beyond the research stage into clinical development and commercial application. A growing concern is the presence of unintended genetic modifications commonly referred to as off-target effects which are now emerging as a central consideration in development planning rather than a secondary…
More Releases for Clinical
Miami Clinical Research Sets the Standard for Clinical Trials
Miami Clinical Research, a frontrunner in the world of clinical trials and medical research, has emerged as the prime choice for global corporate pharmaceutical giants. With a deep understanding of the complexities of medical studies, the organization champions the crucial role of research in the evolution of transformative therapeutic interventions.
Miami, FL - Renowned as a first-rate center for professional medical exploration, Miami Clinical Research [https://miamiclinicalresearch.com] boasts state-of-the-art facilities, advanced technologies,…
E-Clinical Solutions Market: Revolutionizing Healthcare and Clinical Trials
Introduction
The e-Clinical solutions market has become a pivotal component of the healthcare and pharmaceutical industries. E-Clinical solutions refer to a set of software, tools, and platforms designed to streamline clinical trials and healthcare management. These solutions include electronic data capture (EDC), clinical trial management systems (CTMS), laboratory information management systems (LIMS), and other integrated tools that improve the efficiency, accuracy, and speed of clinical trials and healthcare services. The primary…
E-Clinical Solutions Market: Revolutionizing Clinical Trials
The e-clinical solutions market has experienced significant growth in recent years, driven by the increasing complexity of clinical trials and the need for efficient, accurate, and compliant data management. E-clinical solutions provide a comprehensive suite of tools and technologies to streamline clinical trial processes, accelerate drug development, and improve patient outcomes.
Market Size and Growth
The global e-clinical solutions market is estimated to be worth billions of dollars, with a significant portion…
Clinical Trials Management System Market Optimizing Clinical Trials: The Crucial …
Clinical Trials Management System Market to reach over USD 5.06 billion by the year 2031- Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical…
Clinical Research and Clinical Trials Summit
Clinical Research 2019 has been designed in an interdisciplinary manner with a multitude of tracks to choose from every segment and provides you with a unique opportunity to meet up with peers from both industry and academia and establish a scientific network between them. We cordially invite all concerned people to come join us at our event and make it successful by your participation.
This is the premier interdisciplinary forum for…
E-Clinical Trial Solutions Market To Accelerating Clinical Development Technolog …
The study of the "Global e-Clinical Trial Solutions Market" provides the market size information and market trends along with the factors and parameters impacting it in both short and long term. The study ensures a 360° view, bringing out the complete key insights of the industry.
The Global e-Clinical Trial Solutions Market Research Report Forecast 2017-2021 is a valuable source of insightful data for business strategists. It provides the e-Clinical…