Press release
DNA-Based Non-Viral Technologies Market Size to Surpass USD 27.85 Billion by 2035 as Safer Gene Delivery Platforms Accelerate Global Adoption
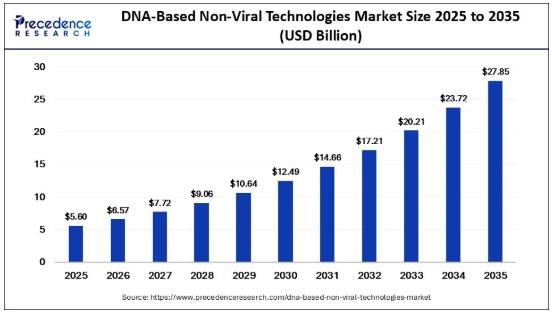
According to Precedence Research, the global DNA-based non-viral technologies market size is projected to rise from USD 6.57 billion in 2026 to approximately USD 27.85 billion by 2035, expanding at a robust CAGR of 17.40% during the forecast period. The market size was valued at USD 5.60 billion in 2025The rapid shift toward non-viral gene delivery systems is being driven by the growing need for lower immunogenicity, repeat dosing capability, flexible manufacturing, and cost-efficient therapeutic development. Technologies such as lipid nanoparticles, electroporation, polymer-based delivery, and nanoparticle-mediated systems are gaining momentum as viable alternatives to conventional viral vectors.
The market is also benefiting from increasing investments in gene therapy pipelines, rising prevalence of rare genetic disorders, and the growing commercialization of personalized medicine platforms across global healthcare systems.
Where Data Meets Strategic Clarity 📥 View Sample Pages of the Complete Report 👉 https://www.precedenceresearch.com/sample/8374
DNA-Based Non-Viral Technologies Market Size and Forecasts
🔹 Market size in 2025: USD 5.60 Billion
🔹 Market size in 2026: USD 6.57 Billion
🔹 Market size by 2035: USD 27.85 Billion
🔹 CAGR: 17.40% (2026-2035)
🔹 Forecast period: 2026-2035
🔹 Base year: 2025
How is Artificial Intelligence Transforming DNA-Based Non-Viral Technologies?
Artificial intelligence is becoming a strategic enabler across the DNA-based non-viral technologies ecosystem. AI-powered algorithms are increasingly being used to optimize delivery vector design, predict gene expression outcomes, and improve transfection efficiency across different cell types.
Machine learning models are also helping researchers identify ideal nanoparticle formulations, reduce toxicity risks, and shorten therapeutic development timelines. This is particularly valuable for advanced genome editing applications where delivery precision directly impacts treatment success.
AI further supports large-scale genomic data analysis, enabling biotechnology companies to accelerate discovery workflows and develop more targeted DNA constructs for next-generation therapies. As therapeutic complexity increases, AI integration is expected to become a core competitive differentiator within the non-viral delivery landscape.
🔗 What's Fueling the Next Wave of Growth? 👉 https://www.precedenceresearch.com/dna-based-non-viral-technologies-market
DNA-Based Non-Viral Technologies Market Key Growth Drivers
🔹Rising Demand for Safer Alternatives to Viral Vectors: One of the strongest growth drivers is the increasing preference for safer gene delivery systems. Non-viral technologies provide lower immunogenicity and reduced toxicity while supporting repeat administration, making them highly attractive for advanced therapeutic applications.
🔹Expanding Gene Therapy and Cell Therapy Pipelines: The growing number of clinical trials in gene therapy, CAR-T therapy, stem cell engineering, and genome editing is accelerating market demand. Pharmaceutical and biotechnology companies are investing aggressively in scalable DNA delivery technologies to support commercialization.
🔹Increasing Investments in Biotechnology Research: Governments, venture capital firms, and healthcare organizations are increasing funding for biotechnology innovation, precision medicine, and advanced therapeutic research. This trend is significantly boosting the development and adoption of DNA-based non-viral delivery platforms.
🔹Growth in Personalized and Precision Medicine: The rising focus on individualized treatments is creating substantial demand for flexible and customizable DNA delivery systems capable of supporting patient-specific therapeutic approaches.
DNA-Based Non-Viral Technologies Market Opportunities
🔹Expanding Applications in Genome Editing and Cancer Therapy: The increasing use of CRISPR technologies, oncology therapeutics, and regenerative medicine is creating major growth opportunities for non-viral delivery systems.
🔹Growth Potential in Emerging Markets: Asia Pacific, Latin America, and parts of the Middle East are witnessing rising healthcare investments, biotechnology infrastructure expansion, and supportive government initiatives, opening new avenues for market growth.
🔹Advancements in In Vivo Delivery Technologies: In vivo delivery technologies are rapidly evolving, enabling more efficient gene therapies without complex cell extraction procedures. This advancement is expected to create substantial long-term opportunities.
➡️ Become a valued research partner with us https://www.precedenceresearch.com/schedule-meeting
DNA-Based Non-Viral Technologies Market Trends
🔹Rapid Shift Toward Non-Viral Gene Delivery Systems: The healthcare and biotechnology sectors are rapidly transitioning from viral vectors to non-viral delivery technologies due to improved safety profiles, lower immune response risks, and better scalability. Non-viral systems are increasingly preferred for repeat dosing applications and personalized therapeutics, making them highly suitable for long-term treatment strategies.
🔹Advancements in Lipid Nanoparticles and Nanotechnology: Lipid nanoparticle (LNP) technologies continue to transform the DNA-based non-viral technologies market. Advances in nanoparticle engineering are improving cellular uptake, targeting precision, gene expression efficiency, and therapeutic stability. Companies are investing heavily in next-generation nanoparticle-mediated delivery platforms to support gene therapy and mRNA applications.
🔹Growing Demand for Personalized Medicine and Genome Editing: The rise of precision medicine and CRISPR-based genome editing technologies is significantly increasing demand for customizable DNA delivery systems. Non-viral technologies enable flexible therapeutic development, faster production timelines, and targeted disease correction approaches for oncology, rare diseases, and regenerative medicine.
🔹Artificial Intelligence Enhancing Gene Therapy Development: Artificial intelligence is becoming a transformative force in the DNA-based non-viral technologies market. AI-driven analytics are helping researchers optimize DNA constructs, predict gene expression outcomes, improve delivery vector design, and accelerate therapeutic discovery. AI integration is expected to reduce development timelines while enhancing delivery precision and therapeutic effectiveness.
🔹Increasing Strategic Collaborations Across the Industry: Biotechnology companies, academic institutions, CROs, and CDMOs are increasingly entering strategic partnerships to accelerate innovation in non-viral gene delivery systems. Collaborative R&D efforts are improving commercialization speed, manufacturing scalability, and clinical development success rates.
🔓 Instant Access. Zero Waiting. 📥 Buy the Premium Market Research Report Now 👉 https://www.precedenceresearch.com/checkout/8374
DNA-Based Non-Viral Technologies Market Regional Analysis
North America dominated the global DNA-based non-viral technologies market with a 45% market share in 2025. The region benefits from a highly developed biotechnology ecosystem, strong funding for gene therapy research, advanced clinical trial infrastructure, and favorable regulatory pathways.
The United States remains the primary growth engine within North America due to increasing investments in precision medicine, expanding gene therapy pipelines, and strong collaboration between biotech companies and research institutions.
Asia Pacific is projected to register the fastest CAGR of 21.5% between 2026 and 2035. Rapid growth in the region is driven by expanding clinical research activity, improving healthcare infrastructure, government support for biotechnology innovation, and rising demand for advanced therapies.
China and India are emerging as key contributors due to large-scale biotech investments, growing manufacturing capabilities, skilled scientific talent, and increasing adoption of precision medicine technologies.
Note: This report is readily available for immediate delivery. We can review it with you in a meeting to ensure data reliability and quality for decision-making.
Try Before You Buy - Get the Sample Report@ https://www.precedenceresearch.com/sample/8374
DNA-Based Non-Viral Technologies Market Segment Analysis
🔸Technology Type Analysis
The lipid-based transfection segment led the DNA-based non-viral technologies market in 2025 with a 30% share. Its dominance is driven by high transfection efficiency, low toxicity, and broad compatibility with multiple cell types. It is widely used in both research and therapeutic applications due to its ease of use and reliability in gene delivery.
The electroporation-based delivery segment held a 25% share and is expected to grow at a 16% CAGR through 2035. Its strength lies in efficient gene transfer without chemical carriers, making it especially useful for hard-to-transfect cells such as immune and primary cells. Meanwhile, nanoparticle-mediated delivery accounted for 20% share and is projected to grow at the fastest CAGR of 21.5%, supported by advancements in nanotechnology enabling targeted and precise gene delivery.
🔸Application Analysis
Gene therapy development dominated applications with a 30% share in 2025, supported by rising clinical trials and increasing investment in genetic disorder treatments. Strong pipeline activity continues to reinforce its leading position.
Cell therapy held a 25% share and is expected to grow at 20.5% CAGR, driven by CAR-T and other engineered cell therapies. Genome editing accounted for a 20% share but is projected to grow fastest at 21% CAGR, fueled by CRISPR advancements and precision medicine applications. Genetic research & functional genomics held 15% share, supported by continued demand for disease mechanism studies and biomarker discovery.
🔸Nucleic Acid Type Analysis
Plasmid DNA dominated with a 50% share in 2025 due to its widespread use in gene expression, vaccine development, and research applications. Its stability and ease of production make it the most preferred nucleic acid type.
Synthetic DNA constructs held a 30% share and are expected to grow at the fastest CAGR of 20%, driven by advancements in DNA synthesis and rising demand for customized genetic sequences.
🔸End-Use Analysis
Pharmaceutical and biotechnology companies led with a 45% share in 2025, driven by strong investments in drug discovery and gene therapy pipelines. Their dominance reflects extensive adoption of DNA-based non-viral technologies for therapeutic development.
Academic and research institutes accounted for 35% share and are growing steadily due to increased focus on genetic research. CROs/CDMOs held 20% share but are expected to grow fastest at 18.5% CAGR, supported by rising outsourcing trends and demand for scalable, compliant manufacturing solutions.
🔸Delivery Mode Analysis
Ex vivo delivery dominated the market with a 60% share in 2025 due to its high efficiency in modifying cells outside the body, especially in cell therapy applications.
In vivo delivery held a 40% share and is expected to grow at a 20.5% CAGR. Growth is driven by increasing preference for direct patient administration, simplified treatment processes, and improving delivery technologies.
✚ Related Topics You May Find Useful:
➡️ Viral and Non-Viral Vectors Market Size, Share and Trends 2026 to 2035 👉 https://www.precedenceresearch.com/viral-and-non-viral-vectors-market
➡️ Plasmid DNA Manufacturing Market Size, Share and Trends 2026 to 2035 👉 https://www.precedenceresearch.com/plasmid-dna-manufacturing-market
➡️ Viral Vectors-Based Gene Therapy for Non-Human Primates Market Size, Share and Trends 2026 to 2035 👉 https://www.precedenceresearch.com/viral-vectors-based-gene-therapy-for-non-human-primates-market
➡️ Viral Vectors and Plasmid DNA Manufacturing Market Size, Share and Trends 2026 to 2035 👉 https://www.precedenceresearch.com/viral-vectors-and-plasmid-dna-manufacturing-market
Top Companies in the DNA-Based Non-Viral Technologies Market and Their Offerings
➢Thermo Fisher Scientific Inc. offers InvitrogenTM VivofectamineTM Delivery Solutions, a lipid nanoparticle (LNP) technology for non-viral nucleic acid delivery, including DNA, optimized for in vivo efficiency, specificity, and safety in genetic medicine.
➢Lonza Group AG provides Nucleofector® Technology, an electroporation-based system for non-viral genetic modifications using DNA, supporting CRISPR and transposon applications in cell and gene therapies.
➢Bio-Rad Laboratories, Inc. supports RNA therapeutics development with Droplet Digital PCR (ddPCR) for nucleic acid quantification, aiding non-integrating nucleic acid approaches, though primarily focused on analysis rather than direct delivery.
➢Promega Corporation specializes in genomic DNA extraction kits for high-quality DNA isolation from various samples, essential for preparing DNA used in non-viral transfection workflows.
➢Qiagen N.V. supplies large-scale purification technologies for DNA-based genetic drugs and vaccines, enabling production of high-purity plasmid DNA for non-viral applications.
➢Polyplus-transfection SA (Sartorius) develops jetPRIME® transfection reagents for efficient, low-toxicity DNA and siRNA delivery into adherent cells, now integrated into Sartorius for gene therapy plasmid transfection.
➢MaxCyte, Inc. delivers the ExPERT GTx Flow Electroporation platform for non-viral DNA delivery in CAR-T manufacturing, achieving high efficiency and cell viability with nanoplasmid DNA and CRISPR/Cas9.
➢Mirus Bio LLC (Part of Gamma Biosciences) pioneered TransIT-VirusGEN® transfection reagents for high-efficiency plasmid DNA delivery with low toxicity, optimized for virus production and genome editing.
➢Takara Bio Inc. offers gene expression systems and custom reagents for gene therapy, including Tet-based inducible systems and services supporting non-viral DNA applications like immunotherapy.
➢Agilent Technologies, Inc. provides imaging tools like Cytation systems with Gen5 software to optimize and analyze transfection efficiency for DNA introduction into cells.
➢STEMCELL Technologies Inc. focuses on primary cell solutions but lacks specific DNA-based non-viral delivery products; supports cell culture for transfection workflows.
➢Altogen Biosystems offers AltoFectTM and cell-specific transfection reagents for DNA/siRNA delivery into primary and hard-to-transfect cells, plus in vivo tissue-targeted systems.
➢VectorBuilder Inc. specializes in custom plasmid DNA vector design, cloning, and production (including MuteFreeTM AAV plasmids), foundational for non-viral gene delivery.
➢OriGene Technologies, Inc. provides Turbofectin 8.0 reagents and protocols for transient DNA transfection into adherent and suspension cells.
➢Creative Biogene produces non-GMP plasmids for preclinical studies and supports nucleic acid services like RNA design, aiding non-viral vector preparation.
Latest Industry Updates
⚡In April 2026, Corning Life Sciences introduced advanced high-density cell culture and automation systems designed to improve plasmid DNA and non-viral vector manufacturing efficiency and scalability.
⚡PlasmidFactory strengthened its GMP-grade DNA manufacturing capabilities in April 2026 to support growing demand for high-quality plasmid DNA used in advanced therapeutic applications.
⚡On April 08, 2026, Aldevron highlighted rising industry confidence in next-generation non-viral and in vivo gene delivery technologies aimed at improving scalability and reducing manufacturing limitations.
Thank you for reading. You can also get individual chapter-wise sections or region-wise report versions, such as North America, Europe, or Asia Pacific.
📥 Instant Report Delivery Available | 💳 Buy Now 👉 https://www.precedenceresearch.com/checkout/8374
Segments Covered in the Report
🔸By Technology Type
Electroporation-based Delivery
Lipid-based Transfection (Lipofection)
Polymer-based Delivery Systems
Nanoparticle-mediated Delivery (LNPs, Gold Nanoparticles)
Physical Methods (Microinjection, Gene Gun)
🔸By Application
Gene Therapy Development
Cell Therapy (CAR-T, Stem Cell Engineering)
DNA Vaccines
Genetic Research & Functional Genomics
Genome Editing (CRISPR/Cas Systems)
🔸By Nucleic Acid Type
Plasmid DNA
Synthetic DNA Constructs
Oligonucleotides
🔸By End-Use
Pharmaceutical & Biotechnology Companies
Academic & Research Institutes
Contract Research & Manufacturing Organizations (CROs/CDMOs)
🔸By Delivery Mode
In Vivo Delivery
Ex Vivo Delivery
🔸By Region
North America
Latin America
Europe
Asia-pacific
Middle and East Africa
Connect With Us
📞 USA: +1 804 441 9344
📞 APAC: +61 485 981 310 or +91 87933 22019 | +6531051271
📞 Europe: +44 7383 092 044
📩 Email: sales@precedenceresearch.com
Precedence Research is a worldwide market research and consulting organization. We give an unmatched nature of offering to our customers present all around the globe across industry verticals. Precedence Research has expertise in giving deep-dive market insight along with market intelligence to our customers spread crosswise over various undertakings. We are obliged to serve our different client base present over the enterprises of medicinal services, healthcare, innovation, next-gen technologies, semi-conductors, chemicals, automotive, and aerospace & defense, among different ventures present globally.
🌐 Web: https://www.precedenceresearch.com
Our Trusted Data Partners:
🔹https://www.towardshealthcare.com
🔹https://www.towardspackaging.com
🔹https://www.towardschemandmaterials.com
🔹https://www.towardsfnb.com
🔹https://www.marketstatsinsight.com
Get Recent News:
https://www.precedenceresearch.com/news
For the Latest Update, Follow Us:
🔹https://www.linkedin.com/company/precedence-research
🔹https://x.com/Precedence_R
🔹https://www.facebook.com/precedenceresearch
🔹https://precedence-research.medium.com/
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release DNA-Based Non-Viral Technologies Market Size to Surpass USD 27.85 Billion by 2035 as Safer Gene Delivery Platforms Accelerate Global Adoption here
News-ID: 4502929 • Views: …
More Releases from Precedence Research
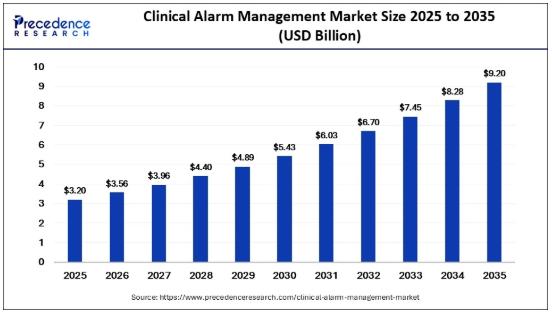
Clinical Alarm Management Market Poised for 11.20% CAGR Growth Reaching USD 9.20 …
According to Precedence Research, the global clinical alarm management market size was estimated at USD 3.20 billion in 2025 and is projected to surge from USD 3.56 billion in 2026 to approximately USD 9.20 billion by 2035, registering a robust CAGR of 11.20% from 2026 to 2035.
The surge in demand is primarily driven by the growing adoption of healthcare IT infrastructure, increasing prevalence of chronic diseases, and a significant rise…
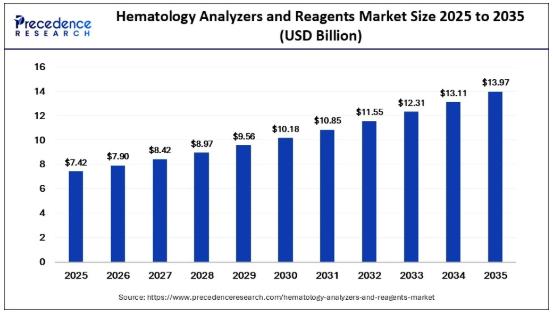
Hematology Analyzers and Reagents Market Size Forecasted to Reach USD 13.97 Bill …
The global hematology analyzers and reagents market is entering a transformative growth phase, fueled by increasing diagnostic testing volumes, rising chronic disease prevalence, and rapid adoption of automation technologies.
According to Precedence Research, the global hematology analyzers and reagents market size was estimated at USD 7.42 billion in 2025 and is projected to grow from USD 7.90 billion in 2026 to USD 13.97 billion by 2035, expanding at a steady…
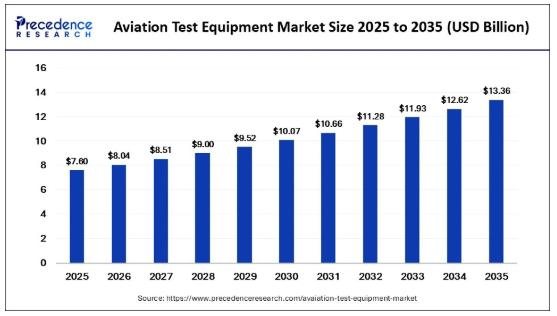
Aviation Test Equipment Market Surges Toward USD 13.36 Billion by 2035 Amid Risi …
According to Precedence Research, the global aviation test equipment market size was valued at USD 7.60 billion in 2025 and is forecasted to reach around USD 13.36 billion by 2035, growing at a steady CAGR of 5.80% from 2026 to 2035. The surge is primarily driven by the rapid expansion of global aircraft fleets, increasing safety regulations, and the growing complexity of avionics and propulsion systems.
The aviation industry's shift toward…
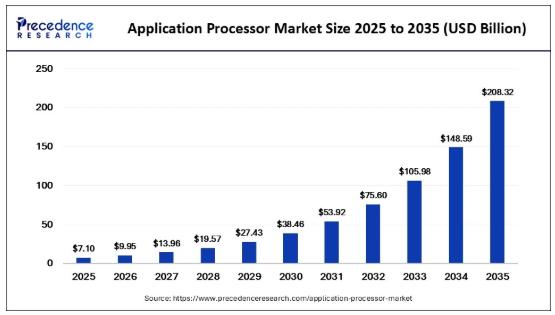
Application Processor Market Size to Surge to USD 208.32 Billion by 2035 Driven …
According to Precedence Research, the global application processor market size will grow from USD 7.10 billion in 2025 to nearly USD 208.32 billion by 2035, expanding at a CAGR of 7.10% from 2026 to 2035.
As industries shift toward smarter, faster, and more connected systems, application processors are becoming the backbone of modern digital infrastructure. Increasing adoption of octa-core processors, 5G-enabled devices, and AI-integrated chipsets is further fueling market expansion globally.
Why…
More Releases for DNA
High-Quality Plasmid DNA Fuels Growth in Global DNA Plasmid Manufacturing Market
🌍 Market Overview
The DNA Plasmid Manufacturing Market is experiencing robust growth as advancements in cell & gene therapy, DNA vaccines, and genetic engineering continue to expand globally. Plasmid DNA plays a critical role as a raw material in the development of advanced therapies, fueling demand across biopharmaceutical research and production.
Key factors driving the market include:
Increasing adoption of gene and cell therapies
Rising prevalence of chronic and rare genetic disorders
Expansion of DNA-based…
DNA Synthesis Market Increasing Demand for Synthetic Genes and DNA Sequences
As demonstrated by Precision Business Insights (PBI), the latest report, the global DNA synthesis market was valued at USD 3,702.0 million in 2023 and is expected to reach USD 10,289.5 million by 2029, growing at a CAGR of 18.6% during the forecast period 2024-2030. The key drivers for the growth of the global DNA synthesis market include increasing demand for synthetic genes and DNA sequences, growing applications in the agriculture…
Wealth DNA Code Review Legit Price? (Wealth Manifestation DNA Code Audio Frequen …
Wealth DNA Code Wealth DNA Code is a digital program with seven minutes of soundtracks that manifest and listen to daily to activate the "Wealth DNA," which is part of your DNA to help you attract wealth by making money a part of your mentality and making your dreams to come true.
https://bit.ly/Visit-The-Official-Website-Here-To-Order-Wealth-DNA-Code
Making money, creating assets as well as increasing wealth are the primary objectives that every human being has to…
DNA Paternity Testing Market Size [2022-2029] -DNA Diagnostics Center, EasyDNA, …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global DNA Paternity Testing. On the basis of historic growth analysis and current scenario of DNA Paternity Testing place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
DNA Paternity Testing Market Trends 2020 | Growth by Top Companies: DNA Diagnost …
The report begins with the overview of the DNA Paternity Testing Market and offers throughout development. It presents a comprehensive analysis of all the regional and major player segments that gives closer insights upon present market conditions and future market opportunities along with drivers, trending segments, consumer behaviour, pricing factors and market performance and estimation. The forecast market information, SWOT analysis, DNA Paternity Testing market scenario, and feasibility study are…
DNA Paternity Testing Market Rapidly Growing in Healthcare, Competitor Analysis …
The exclusive research report on the Global DNA Paternity Testing Market 2020 examines the market in detail along with focusing on significant market dynamics for the key players operating in the market. Global DNA Paternity Testing Industry research report offers granulated yet in-depth analysis of revenue share, market segments, revenue estimates and various regions across the globe.
Overview of Global DNA Paternity Testing Market:
This report studies the Global DNA Paternity Testing…