Press release
Viral Vector and Plasmid DNA Manufacturing Market to Expand Rapidly as Precision Medicine, Advanced Diagnostics and AI-Enabled Care Models Accelerate Global Demand
Austin, Texas, April 29, 2026: DataM Intelligence has released its latest analysis on the Viral Vector and Plasmid DNA Manufacturing Market, highlighting how precision medicine, advanced diagnostics, and AI-enabled R&D are driving sustained demand across the biotech, medtech and healthcare ecosystem.Download PDF Sample Report:
https://www.datamintelligence.com/download-sample/viral-vector-and-plasmid-dna-manufacturing-market?prasad
According to the report, the global Viral Vector and Plasmid DNA Manufacturing Market is witnessing strong growth over the forecast period, supported by increasing adoption of gene therapies, cell-based treatments and nucleic acid-based vaccines. The market is projected to expand at a robust CAGR during the forecast period, with rising investments in advanced therapies and omics-based research strengthening long-term demand. Growth is further underpinned by the expanding pipeline of biologics and increasing reliance on viral vectors and plasmid DNA as core components in therapeutic development and manufacturing workflows.
The market's momentum is closely tied to the convergence of AI-enabled drug discovery platforms, precision medicine frameworks and diagnostics-driven care pathways. Organizations are scaling investments in viral vector and plasmid DNA manufacturing to meet stringent regulatory requirements, improve batch consistency and accelerate clinical timelines. At the same time, reimbursement pressure across developed healthcare systems is pushing manufacturers to optimize production efficiency, reduce cost-per-dose and enhance scalability through automation and digital manufacturing systems.
Market Momentum Strengthens as Advanced Therapies Reshape Biomanufacturing Priorities
The Viral Vector and Plasmid DNA Manufacturing Market has become a critical enabler of next-generation healthcare innovation. Viral vectors are essential for gene delivery in gene therapy, while plasmid DNA plays a foundational role in vaccine production, cell therapy and gene editing applications.
As healthcare systems increasingly transition toward personalized and precision-based treatment models, the need for high-quality, scalable and compliant manufacturing solutions is intensifying. Businesses evaluating infrastructure modernization are prioritizing investments in biomanufacturing platforms that can support diverse vector types, flexible batch sizes and rapid clinical-to-commercial transitions.
Additionally, the rise of omics technologies-including genomics, transcriptomics and proteomics-is accelerating demand for customized DNA constructs and vector systems. This is creating new revenue opportunities for manufacturers capable of delivering high-purity, regulatory-compliant products at scale.
Operationally, the market is addressing key challenges such as production bottlenecks, process variability and regulatory complexity. Advanced manufacturing technologies, including single-use systems and automated bioreactors, are helping organizations improve yield, reduce contamination risks and enhance overall process efficiency.
Pipeline Strength, Trial Phases and Manufacturing Capacity Drive Strategic Investments
A defining feature of the Viral Vector and Plasmid DNA Manufacturing Market is the rapid expansion of the global therapeutic pipeline. Hundreds of gene therapy and cell therapy candidates are currently progressing through clinical development, with a growing number entering late-stage trials.
Pipeline Strength:
The increasing number of investigational therapies targeting rare diseases, oncology indications and genetic disorders is creating sustained demand for viral vectors such as lentivirus, adenovirus and adeno-associated virus (AAV), as well as plasmid DNA constructs. This pipeline expansion is directly translating into higher manufacturing requirements across preclinical and clinical stages.
Trial Phases:
As more candidates move from Phase I to Phase II and III trials, the scale and complexity of manufacturing requirements increase significantly. Late-stage trials require consistent, large-scale production with strict quality control, prompting companies to invest in capacity expansion and process optimization.
Manufacturing Capacity:
Capacity constraints remain a critical issue in the market. To address this, companies are expanding production facilities, investing in modular manufacturing units and forming strategic partnerships with contract development and manufacturing organizations (CDMOs). These initiatives are aimed at reducing lead times, ensuring supply continuity and supporting commercial-scale production.
Regulatory Pathway:
Regulatory frameworks in key markets such as the United States, Europe and Asia-Pacific are evolving to support advanced therapies while maintaining strict safety and efficacy standards. Compliance with Good Manufacturing Practices (GMP), validation protocols and documentation requirements is essential. Companies are increasingly aligning their manufacturing processes with regulatory expectations early in development to avoid delays during approval stages.
Get Customization in the report as per your requirements:
https://www.datamintelligence.com/customize/viral-vector-and-plasmid-dna-manufacturing-market?prasad
Market Segmentation Analysis
The Viral Vector and Plasmid DNA Manufacturing Market is segmented based on product type, application and end-user.
By Product Type:
The market includes viral vectors (such as AAV, lentivirus and adenovirus) and plasmid DNA. Viral vectors account for a significant share due to their widespread use in gene therapy and vaccine delivery, while plasmid DNA remains essential for upstream processes and as a critical raw material.
By Application:
Key applications include gene therapy, vaccinology, cell therapy and research applications. Gene therapy represents a major growth segment, driven by increasing approvals and clinical advancements. Vaccine development, particularly mRNA and DNA-based platforms, also contributes significantly to demand.
By End-User:
End-users include pharmaceutical and biotechnology companies, research institutes and contract manufacturing organizations. Pharmaceutical and biotech companies dominate the market due to their extensive R&D pipelines, while CDMOs are gaining traction as outsourcing becomes a strategic priority.
Each segment reflects a growing need for scalable, high-quality manufacturing solutions, with organizations aligning their procurement strategies to support long-term therapeutic development goals.
Regional Analysis
North America leads the Viral Vector and Plasmid DNA Manufacturing Market, supported by strong R&D infrastructure, favorable regulatory frameworks and significant investment in advanced therapies. The United States remains a key hub, driven by robust clinical trial activity and the presence of leading biopharmaceutical companies.
Europe follows closely, with countries such as the UK, Germany, France and Spain investing in biomanufacturing capabilities and supporting innovation through public and private funding initiatives. Regulatory alignment across the region is also facilitating market growth.
Asia-Pacific is emerging as a high-growth region, with China, Japan, South Korea and Taiwan expanding their biotechnology ecosystems. Increasing government support, rising clinical research activity and growing demand for advanced therapies are driving regional expansion. China, in particular, is investing heavily in manufacturing infrastructure, while Japan and South Korea are focusing on innovation and regulatory harmonization.
Other developed markets are also contributing to growth through strategic investments in healthcare infrastructure and advanced manufacturing technologies.
Purchase the Exclusive Report:
https://www.datamintelligence.com/buy-now-page?report=viral-vector-and-plasmid-dna-manufacturing-market?prasad
Recent Developments in the Global Viral Vector and Plasmid DNA Manufacturing Market
➡️March 2026 - Thermo Fisher Scientific: Announced expansion of its viral vector manufacturing facility in the U.S., increasing capacity to support late-stage clinical and commercial production. This move addresses growing demand for gene therapy manufacturing services.
➡️February 2026 - Lonza Group: Launched a new plasmid DNA manufacturing platform designed to improve scalability and reduce production timelines, enhancing its CDMO capabilities.
➡️January 2026 - Catalent, Inc.: Expanded its gene therapy manufacturing network with additional AAV production suites, strengthening its position in advanced therapy manufacturing.
➡️December 2025 - Samsung Biologics: Entered the viral vector manufacturing space with new facility investments, signaling increased competition and capacity in Asia-Pacific.
➡️November 2025 - FUJIFILM Diosynth Biotechnologies: Announced strategic investment in gene therapy manufacturing expansion in Europe, aimed at supporting growing clinical pipelines.
Competitive Landscape
The Viral Vector and Plasmid DNA Manufacturing Market is characterized by a mix of established biopharmaceutical companies and specialized CDMOs. Competition is driven by manufacturing capacity, technological capabilities and regulatory compliance expertise.
Key players are focusing on expanding global footprints, enhancing production scalability and integrating advanced technologies such as automation and AI-driven process optimization. Strategic partnerships and long-term supply agreements are also shaping the competitive landscape, enabling companies to secure stable revenue streams and strengthen market positioning.
Company Profiles
Thermo Fisher Scientific
Thermo Fisher Scientific plays a significant role in viral vector and plasmid DNA manufacturing through its extensive CDMO services. The company offers end-to-end solutions, from process development to commercial-scale production, leveraging advanced technologies and global infrastructure. Its strategic investments in capacity expansion position it as a key partner for organizations advancing gene and cell therapies.
Lonza Group
Lonza is a leading provider of biomanufacturing services, with strong capabilities in viral vector and plasmid DNA production. The company's focus on innovation, scalability and regulatory compliance makes it a preferred partner for advanced therapy developers. Its global network of facilities supports both clinical and commercial manufacturing needs.
Catalent, Inc.
Catalent specializes in advanced delivery technologies and manufacturing solutions for biologics and gene therapies. Its expertise in viral vector production, combined with continuous investment in facility expansion, enables it to meet growing demand across multiple therapeutic areas.
FUJIFILM Diosynth Biotechnologies
FUJIFILM Diosynth Biotechnologies is expanding its presence in gene therapy manufacturing through strategic investments in infrastructure and technology. The company's integrated approach to development and manufacturing supports efficient scale-up and regulatory compliance.
✅ Competitive Landscape
✅ Technology Roadmap Analysis
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Consumer Behavior & Demand Analysis
✅ Import-Export Data Monitoring
✅ Live Market & Pricing Trends
Contact Us-
Fabian
DataM Intelligence 4market Research LLP
6th Floor, M2 Tech Hub, DataM Intelligence 4market Research LLP, Lalitha Nagar, Habsiguda, Secunderabad, Hyderabad, Telangana 500039
USA: +1 877-441-4866
UK: +44 161-870-5507
Email: fabian@datamintelligence.com
About Us
DataM Intelligence is a market research and consulting firm that delivers comprehensive end-to-end business solutions, covering everything from in-depth research to strategic consulting. The company leverages key industry trends, insights, and developments to provide fast, reliable, and actionable solutions tailored to diverse client requirements.
It offers both syndicated and customized research reports supported by a strong and robust methodology. With an extensive database comprising 9000+ reports across 40+ industry domains, DataM Intelligence serves over 200 companies in more than 50 countries, helping organizations access critical business intelligence that drives informed decision-making and sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Viral Vector and Plasmid DNA Manufacturing Market to Expand Rapidly as Precision Medicine, Advanced Diagnostics and AI-Enabled Care Models Accelerate Global Demand here
News-ID: 4494394 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Air Cargo Security Equipment Market to Accelerate as AI-Driven Threats, Cybersec …
Austin, Texas, April 29, 2026: DataM Intelligence has released its latest analysis on the Air Cargo Security Equipment Market, highlighting how AI adoption, secure data infrastructure, compliance pressure and automation ROI are reshaping global security priorities. The market is projected to witness steady expansion over the forecast period, supported by increasing cargo volumes, heightened cybersecurity risks and regulatory enforcement across international air freight ecosystems.
Download PDF Sample Report:
https://www.datamintelligence.com/download-sample/air-cargo-security-equipment-market?prasad
The Air Cargo…
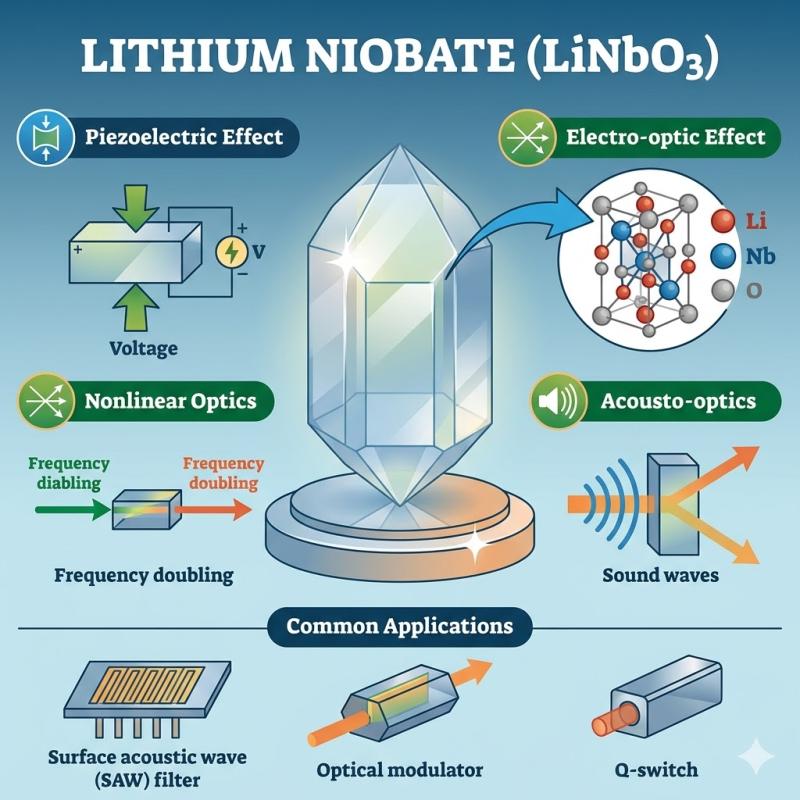
Lithium Niobate Market to Accelerate as Electrification, Charging Infrastructure …
Austin, Texas, April 29, 2026: DataM Intelligence has released its latest analysis on the Lithium Niobate Market, highlighting accelerating demand driven by electrification, grid modernization, energy security priorities and intensifying focus on critical-mineral supply risks across advanced economies.
Download PDF Sample Report:
https://www.datamintelligence.com/download-sample/lithium-niobate-market?prasad
The global Lithium Niobate Market is projected to witness steady growth over the forecast period, supported by rising deployment in optical communication systems, advanced electronics, energy-efficient components and emerging…
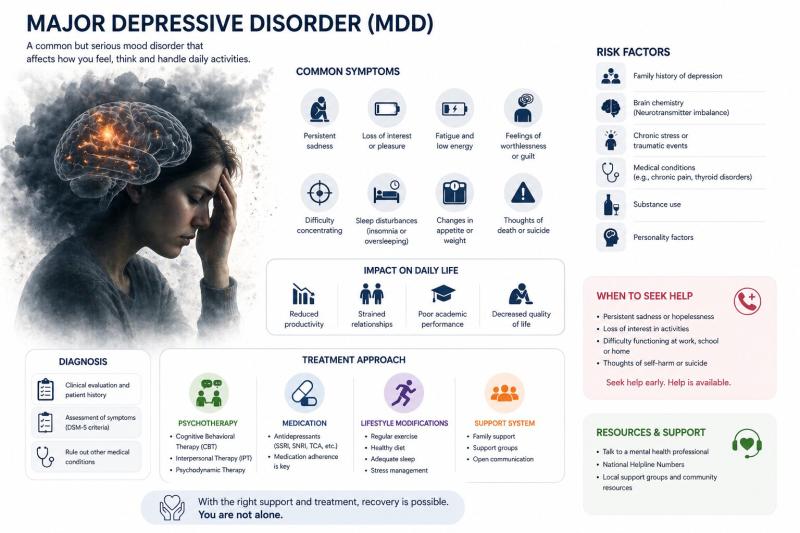
Major Depressive Disorder Market to Reach USD 14.57 Billion by 2033 at 8.8% CAGR …
The global major depressive disorder (MDD) market reached USD 7.44 billion in 2025 and is expected to reach USD 14.57 billion by 2033, growing at a CAGR of 8.8% during the forecast period from 2026 to 2033. The market is witnessing significant growth driven by the rising global prevalence of depression, increasing awareness around mental health, and expanding diagnosis rates across both developed and emerging regions. The growing societal and…
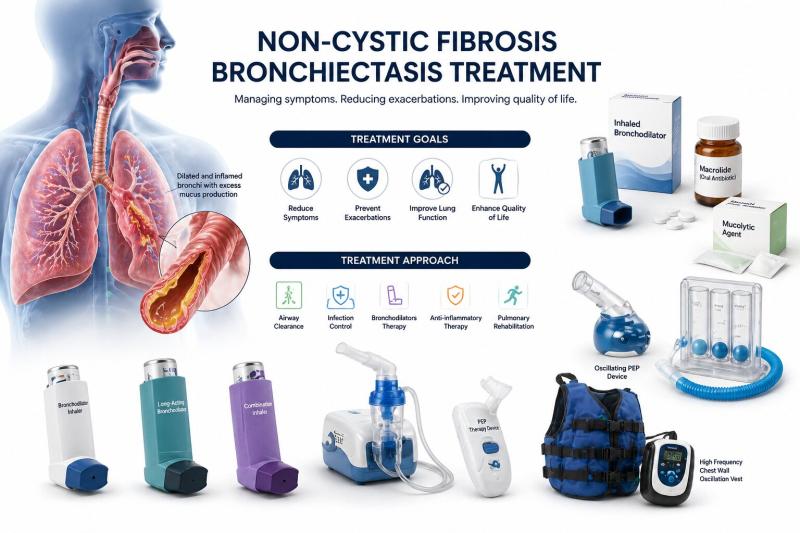
Non-Cystic Fibrosis Bronchiectasis Treatment Market to Reach USD 6.28 Billion by …
The global non-cystic fibrosis bronchiectasis (NCFB) treatment market reached USD 2.48 billion in 2025 and is expected to reach USD 6.28 billion by 2033, growing at a CAGR of 8.6% during the forecast period from 2026 to 2033. The market is witnessing steady growth driven by rising disease prevalence, increasing awareness, and improved diagnostic capabilities, particularly in aging populations and patients with chronic respiratory conditions. The growing burden of respiratory…
More Releases for DNA
High-Quality Plasmid DNA Fuels Growth in Global DNA Plasmid Manufacturing Market
🌍 Market Overview
The DNA Plasmid Manufacturing Market is experiencing robust growth as advancements in cell & gene therapy, DNA vaccines, and genetic engineering continue to expand globally. Plasmid DNA plays a critical role as a raw material in the development of advanced therapies, fueling demand across biopharmaceutical research and production.
Key factors driving the market include:
Increasing adoption of gene and cell therapies
Rising prevalence of chronic and rare genetic disorders
Expansion of DNA-based…
DNA Synthesis Market Increasing Demand for Synthetic Genes and DNA Sequences
As demonstrated by Precision Business Insights (PBI), the latest report, the global DNA synthesis market was valued at USD 3,702.0 million in 2023 and is expected to reach USD 10,289.5 million by 2029, growing at a CAGR of 18.6% during the forecast period 2024-2030. The key drivers for the growth of the global DNA synthesis market include increasing demand for synthetic genes and DNA sequences, growing applications in the agriculture…
Wealth DNA Code Review Legit Price? (Wealth Manifestation DNA Code Audio Frequen …
Wealth DNA Code Wealth DNA Code is a digital program with seven minutes of soundtracks that manifest and listen to daily to activate the "Wealth DNA," which is part of your DNA to help you attract wealth by making money a part of your mentality and making your dreams to come true.
https://bit.ly/Visit-The-Official-Website-Here-To-Order-Wealth-DNA-Code
Making money, creating assets as well as increasing wealth are the primary objectives that every human being has to…
DNA Paternity Testing Market Size [2022-2029] -DNA Diagnostics Center, EasyDNA, …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global DNA Paternity Testing. On the basis of historic growth analysis and current scenario of DNA Paternity Testing place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
DNA Paternity Testing Market Trends 2020 | Growth by Top Companies: DNA Diagnost …
The report begins with the overview of the DNA Paternity Testing Market and offers throughout development. It presents a comprehensive analysis of all the regional and major player segments that gives closer insights upon present market conditions and future market opportunities along with drivers, trending segments, consumer behaviour, pricing factors and market performance and estimation. The forecast market information, SWOT analysis, DNA Paternity Testing market scenario, and feasibility study are…
DNA Paternity Testing Market Rapidly Growing in Healthcare, Competitor Analysis …
The exclusive research report on the Global DNA Paternity Testing Market 2020 examines the market in detail along with focusing on significant market dynamics for the key players operating in the market. Global DNA Paternity Testing Industry research report offers granulated yet in-depth analysis of revenue share, market segments, revenue estimates and various regions across the globe.
Overview of Global DNA Paternity Testing Market:
This report studies the Global DNA Paternity Testing…
