Press release
Biosimilars Market Surge Driven by Patent Expiries, Cost-Effective Biologics, and AI-Enabled Therapy Optimization, Projected to Reach USD 125.9 Billion by 2035
Wilmington, DE, USA, April 28, 2026 - According to MarketGenics Global Research, the global biosimilars market is valued at USD 31.6 billion in 2025 and is projected to reach USD 125.9 billion by 2035, expanding at a CAGR of 13.4% during the forecast period.The market is undergoing a structural shift as healthcare systems transition toward cost-efficient biologic therapies, driven by patent expiries of blockbuster drugs and increasing demand for affordable treatment options across oncology, autoimmune diseases, and chronic conditions.
Biosimilars are increasingly integrated into clinical practice as therapeutically equivalent alternatives to high-cost biologics, enabling broader patient access while maintaining efficacy, safety, and regulatory compliance under frameworks such as FDA and EMA approvals.
Advancements in AI-enabled therapy monitoring, digital treatment optimization platforms, and real-world data integration are further strengthening biosimilar adoption by improving treatment personalization, adherence, and clinical outcomes.
Get Sample Copy of the Report: https://marketgenics.co/download-report-sample/biosimilars-market-04169
==============================
MARKET STRUCTURE & CONCENTRATION
==============================
The global biosimilars market is moderately consolidated, with the top five players accounting for over 45% of total market share in 2024, reflecting strong control over biologics manufacturing capabilities, regulatory approvals, and commercialization networks.
Leading companies including Amgen Inc., Novartis AG (Sandoz), Pfizer Inc., Samsung Bioepis, and Biocon Biologics Ltd. dominate through integrated biologics development ecosystems, combining clinical development, large-scale biomanufacturing, and global distribution capabilities.
Market leadership is defined by:
• Regulatory expertise - navigating complex biosimilar approval pathways and interchangeability requirements
• Manufacturing scale - high-cost biologics production infrastructure and process optimization
• Pipeline depth - strong portfolios of monoclonal antibodies and next-generation biologics
• Commercial reach - strategic partnerships with healthcare systems, payers, and providers
This structure creates high entry barriers due to capital-intensive development (USD 100-300 million per product), stringent clinical validation, and regulatory complexity, reinforcing dominance of established players.
==============================
SEGMENTAL DATA INSIGHTS
==============================
The monoclonal antibodies (mAbs) segment dominates the global biosimilars market, accounting for approximately 36% share, driven by extensive use in oncology, autoimmune diseases, and inflammatory conditions.
Biosimilar mAbs such as adalimumab, trastuzumab, and rituximab are expanding rapidly due to their role in treating high-burden diseases and their ability to significantly reduce treatment costs.
Recombinant proteins and hormones are gaining traction, while enzymes are expected to grow at a comparatively lower rate.
==============================
DEMAND TRENDS
==============================
Clinical demand is being driven by the growing burden of chronic diseases, including cancer, rheumatoid arthritis, and diabetes, where long-term biologic therapy is required.
Healthcare systems are increasingly adopting biosimilars to reduce treatment costs and expand access, particularly as biologics account for over 40% of pharmaceutical spending in developed markets.
Rising physician confidence, supported by regulatory approvals and real-world clinical evidence, is accelerating adoption across therapeutic areas, while favorable reimbursement policies and government initiatives are strengthening market penetration.
==============================
VALUE CHAIN & APPLICATION INTELLIGENCE
==============================
The biosimilars market operates through a biopharmaceutical development and commercialization pipeline, spanning from biologic reference product analysis to clinical validation and large-scale deployment:
• Upstream - Biologic Characterization & Cell Line Development
Reverse engineering of reference biologics, development of cell lines, and process optimization for biosimilar production.
• Midstream - Clinical Trials, Regulatory Approval & Manufacturing Scale-Up
Extensive analytical and clinical studies to demonstrate biosimilarity, followed by regulatory approval under FDA, EMA, and global frameworks.
• Downstream - Distribution, Reimbursement & Clinical Adoption
Integration into hospital formularies, payer systems, and treatment protocols across oncology, immunology, and chronic disease management.
• Clinical Application Layer - Long-Term Therapy & Disease Management
Biosimilars are widely used in chronic and life-threatening conditions, enabling sustained treatment at reduced cost and improved accessibility.
This value chain is defined by regulatory rigor, manufacturing complexity, and payer-driven adoption dynamics, shaping commercialization strategies globally.
==============================
TECHNOLOGY TRANSFORMATION
==============================
The market is evolving through integration of bioprocess engineering, digital health platforms, and AI-enabled treatment optimization.
Innovations such as AI-driven biosimilar selection tools, real-time patient monitoring systems, and cloud-based treatment management platforms are improving therapy outcomes, adherence, and healthcare efficiency.
Advancements in biologics formulation, including longer dosing intervals and improved stability, are enhancing patient convenience and treatment compliance.
==============================
PRICING & COST PRESSURE ANALYSIS
==============================
Despite lower pricing compared to originator biologics, biosimilars face high development and manufacturing costs, ranging between USD 100 million to USD 300 million per product.
Regulatory complexities, interchangeability challenges, and market access barriers impact adoption rates and commercialization timelines.
==============================
END-USE INTENSITY ANALYSIS
==============================
Demand intensity varies across healthcare settings:
• Hospitals - Primary centers for biologic therapy administration
• Specialty Clinics - High adoption in oncology and immunology treatments
• Biopharmaceutical Companies - Integration in drug development and lifecycle management
• Homecare Settings - Emerging adoption with self-administration formats
This multi-channel adoption supports scalable growth across healthcare systems.
==============================
REGIONAL ANALYSIS - DEMAND CENTERS
==============================
North America dominates the global biosimilars market, accounting for approximately 45-50% of total market share in 2024, driven by strong regulatory frameworks, high healthcare expenditure, and rapid adoption of biologic alternatives.
The United States leads global adoption, supported by the Biologics Price Competition and Innovation Act (BPCIA), increasing biosimilar approvals, and strong reimbursement systems.
Canada is expected to witness strong growth due to supportive pricing reforms and biosimilar switching policies.
==============================
INNOVATION & MATERIAL SHIFT
==============================
Manufacturers are focusing on advanced biologics development, formulation optimization, and personalized therapy approaches to enhance treatment outcomes and patient accessibility.
Increasing investment in biosimilar pipelines across oncology and immunology is expanding therapeutic coverage globally.
==============================
COMPETITIVE LANDSCAPE
==============================
• Amgen Inc.
• Novartis AG (Sandoz)
• Pfizer Inc.
• Samsung Bioepis
• Biocon Biologics Ltd.
• Celltrion Inc.
• Fresenius Kabi
• Merck KGaA
• Teva Pharmaceutical Industries Ltd.
• Sanofi S.A.
• Roche Holding AG
• Dr. Reddy's Laboratories
• STADA Arzneimittel AG
• Intas Pharmaceuticals Ltd.
• Coherus BioSciences
• Alvotech
• Apotex Inc.
• Reliance Life Sciences
• Shanghai Henlius Biotech, Inc.
• Other Key Players
==============================
STRATEGIC DEVELOPMENTS
==============================
In February 2025, Samsung Bioepis launched PYZCHIVA in the U.S. with an AI-enabled real-time monitoring platform, enabling personalized biosimilar therapy and improved treatment outcomes.
In April 2025, Pfizer introduced IXIFI in Canada alongside a cloud-based biosimilar management hub integrating patient data, biomarker insights, and treatment optimization tools.
==============================
FUTURE OUTLOOK & OPPORTUNITIES
==============================
The global biosimilars market is expected to create an incremental opportunity of approximately USD 94.4 billion by 2035.
North America remains the most attractive region, driven by regulatory support, increasing biologic drug costs, and expanding biosimilar adoption across therapeutic areas.
Biosimilars are positioned as a critical component of value-based healthcare systems, enabling cost optimization, expanded patient access, and long-term sustainability of biologic therapies.
==============================
THE GLOBAL BIOSIMILARS MARKET HAS BEEN SEGMENTED AS FOLLOWS:
==============================
By Product Type
• Monoclonal Antibodies (mAbs)
• Recombinant Hormones
• Recombinant Proteins
• Erythropoietin
• Granulocyte-Colony Stimulating Factor (G-CSF)
• Insulin
• Enzymes
• Fusion Proteins
• Others
By Application
• Oncology
• Autoimmune Diseases
• Diabetes
• Infectious Diseases
• Blood Disorders
• Hormonal Disorders
• Chronic Kidney Disease
• Ophthalmic Disorders
• Others
By Manufacturing Type
• In-house Manufacturing
• Contract Manufacturing
By Distribution Channel
• Hospital Pharmacies
• Retail Pharmacies
• Online Pharmacies
• Specialty Clinics
By Route of Administration
• Intravenous (IV)
• Subcutaneous (SC)
• Intramuscular (IM)
• Oral
• Others
By End-users
• Hospitals
• Specialty Clinics
• Research Institutes
• Ambulatory Surgical Centers
• Homecare Settings
• Diagnostic Laboratories
• Biopharmaceutical Companies
• Contract Research Organizations (CROs)
• Others
==============================
KEY MARKET TRENDS
==============================
Growing adoption of biosimilars as cost-effective alternatives to biologics is accelerating market expansion.
Integration of AI and digital health platforms is enhancing therapy optimization and patient monitoring.
Increasing regulatory support and reimbursement policies are driving market penetration.
Expansion of biosimilars across multiple therapeutic areas is strengthening long-term growth.
==============================
RESEARCH METHODOLOGY:
==============================
The study integrates product-level analysis, clinical adoption trends, and regulatory frameworks across global healthcare systems.
Analytical frameworks include cost structure analysis, competitive benchmarking, and innovation tracking.
The research further incorporates regional demand modeling and therapeutic application analysis to identify strategic growth opportunities.
Access the full report and strategic insights: https://marketgenics.co/reports/biosimilars-market-04169
==============================
RECOMMENDED REPORTS:
==============================
Healthcare Data Analytics Market: https://marketgenics.co/reports/healthcare-data-analytics-market-84339
Medical 3D Printing Market: https://marketgenics.co/reports/medical-3d-printing-market-00999
Contact:
Mr. Debashish Roy
MarketGenics Global Research
800 N King Street, Suite 304 #4208, Wilmington, DE 19801, United States
USA: +1 (302) 303-2617
Email: sales@marketgenics.co
Website: https://marketgenics.co
About MarketGenics
MarketGenics is a global market research and business advisory firm empowering decision-makers across startups, Fortune 500 companies, non-profit organizations, universities, and government institutions. The company delivers comprehensive market intelligence, industry analysis, and strategic insights across diverse sectors.
MarketGenics publishes detailed industry research reports combining granular quantitative analysis with expert insights on market trends, competitive landscapes, and emerging opportunities. These reports help organizations make informed strategic decisions, identify growth opportunities, and support sustainable business development.
In addition to research publications, MarketGenics supports organizations with strategic insights on product development, application modeling, market expansion strategies, and identifying niche growth opportunities.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Biosimilars Market Surge Driven by Patent Expiries, Cost-Effective Biologics, and AI-Enabled Therapy Optimization, Projected to Reach USD 125.9 Billion by 2035 here
News-ID: 4492143 • Views: …
More Releases from MarketGenics Global Research

Biodegradable Packaging Market to Reach USD 236.6 Billion by 2035 as Paper & Pap …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global biodegradable packaging market is valued at USD 114.8 billion in 2025 and is projected to reach USD 236.6 billion by 2035, expanding at a CAGR of 7.5% during the forecast period from 2026 to 2035.
The market is experiencing strong global momentum as governments, consumer brands, retailers, food service providers, and e-commerce companies accelerate the transition away…

Active Packaging Market to Reach USD 27.6 Billion by 2035 as Oxygen Scavenging S …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global active packaging market is valued at USD 16.3 billion in 2025 and is projected to reach USD 27.6 billion by 2035, expanding at a CAGR of 5.4% during the forecast period from 2026 to 2035.
The market is witnessing strong momentum due to increasing demand for shelf-life extension, food safety enhancement, spoilage reduction, pharmaceutical stability protection, and…
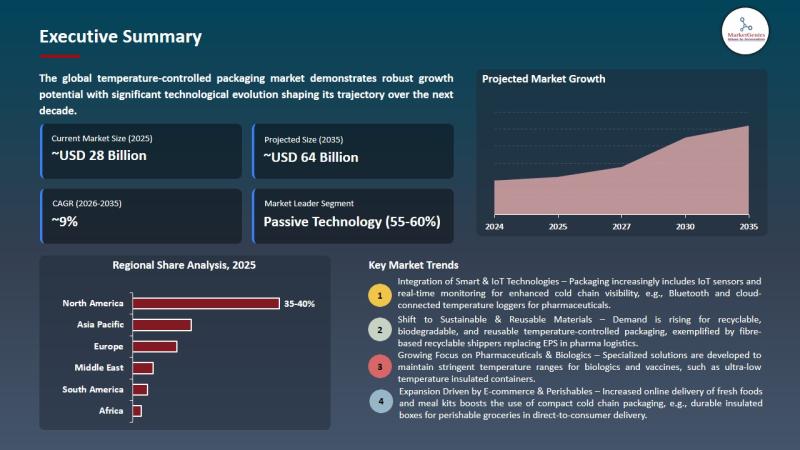
Temperature-Controlled Packaging Market to Reach USD 63.9 Billion by 2035 as Pas …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global temperature-controlled packaging market is valued at USD 28.2 billion in 2025 and is projected to reach USD 63.9 billion by 2035, expanding at a CAGR of 8.5% during the forecast period (2026-2035).
The market is witnessing substantial growth due to rising global demand for cold-chain logistics solutions across pharmaceuticals, biologics, vaccines, specialty drugs, perishable foods, and temperature-sensitive…

5G Chipset Market to Reach USD 160.3 Billion by 2035 as eMBB Applications Captur …
Wilmington, DE, USA, 8th May 2026 - According to MarketGenics Global Research, the global 5G chipset market is valued at USD 29.6 billion in 2025 and is projected to reach USD 160.3 billion by 2035, expanding at a CAGR of 18.4% during the forecast period (2026-2035).
The market is witnessing rapid expansion due to increasing global rollout of 5G infrastructure, rising smartphone penetration, accelerating industrial IoT adoption, deployment of AI-enabled connectivity…
More Releases for Biosimilar
Interchangeable Biosimilar Humira Market Share Driven by Biologic Therapy Adopti …
Interchangeable Biosimilar Humira Market
The global market for Interchangeable Biosimilar Humira was valued at US$ million in the year 2024 and is projected to reach a revised size of US$ million by 2031, growing at a CAGR of %during the forecast period
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-33I15005/Global_Interchangeable_Biosimilar_Humira_Market_Research_Report_2023
The Interchangeable Biosimilar Humira Market is experiencing significant market growth as healthcare providers and patients increasingly adopt biosimilar therapies for autoimmune and inflammatory conditions. Market trends indicate rising…
Key Trend Reshaping the Biosimilar Monoclonal Antibodies Market in 2025: Advance …
What Are the Projections for the Size and Growth Rate of the Biosimilar Monoclonal Antibodies Market?
In recent times, the biosimilar monoclonal antibodies sector has experienced a swift expansion. The market size, which stands at $8.04 billion in 2024, is projected to climb to $9.25 billion in 2025, marking a compound annual growth rate (CAGR) of 15.1%. Factors such as expired patents, an increased understanding of biosimilars, governmental strategies, heightened financial…
Key Trend Reshaping the Biosimilar Monoclonal Antibodies Market in 2025: Advance …
What Are the Projections for the Size and Growth Rate of the Biosimilar Monoclonal Antibodies Market?
In recent times, the biosimilar monoclonal antibodies sector has experienced a swift expansion. The market size, which stands at $8.04 billion in 2024, is projected to climb to $9.25 billion in 2025, marking a compound annual growth rate (CAGR) of 15.1%. Factors such as expired patents, an increased understanding of biosimilars, governmental strategies, heightened financial…
Biosimilar Market Treating More for Less: The Booming Infliximab Biosimilar Mark …
Infliximab Biosimilar Market worth $ XX Million by 2030 - Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Infliximab Biosimilar Market- by Application (Crohn's Disease, Psoriatic Arthritis, Rheumatoid Arthritis, Ulcerative Colitis, Ankylosing Spondylitis, Plaque Psoriasis and Others), End User (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy and Other Direct Distribution Channels), Trends, Industry Competition Analysis, Revenue and Forecast To 2030."
Get…
Biosimilar Monoclonal Antibodies Market
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the " "Global Biosimilar Monoclonal Antibodies Market by Product (infliximab, trastuzumab, rituximab, adalimumab, bevacizumab, cetuximab, ranibizumab, denosumab, eculizumab, and other pipeline products), Indication (oncology, inflammatory & autoimmune disorders, chronic diseases, blood disorders, and other indications), Clinical Trial/Pipeline Analysis, Future Trends, Industry Competition Analysis, Revenue and Forecast To 2031."
The Biosimilar Monoclonal Antibodies Market Size is valued at 5.02…
Infliximab Biosimilar Insight, 2022 | DelveInsight
DelveInsight's, "Infliximab Biosimilar Insight, 2022" report provides comprehensive insights about 35+ companies and 45+ marketed and pipeline drugs in Infliximab Biosimilars landscape. It covers the marketed and pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Interested to know more about the functioning of…
