Press release
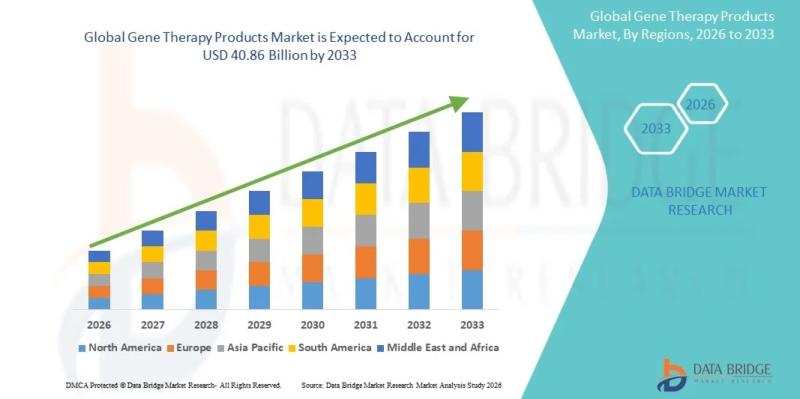
Global Gene Therapy Products Market to Reach USD 40.86 Billion by 2033, Growing at 22.15% CAGR from 2025
Market SummaryAs per Data Bridge Market Research analysis, the Gene Therapy Products Market was estimated at USD 8.24 billion in 2025. The market is expected to grow from USD 10.07 billion in 2026 to USD 40.86 billion in 2033, at a CAGR of 22.15% during the forecast period, driven by the rising demand for advanced genetic treatments, increasing prevalence of rare and chronic diseases, and continuous innovation in gene-editing technologies.
Get the full PDF sample copy of the report: https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-gene-therapy-products-market
Market Size & Forecast
2025 Market Size: USD 8.24 Billion
2026 Projected Market Size: USD 10.07 Billion
2033 Projected Market Size: USD 40.86 Billion
CAGR (2026-2033): 22.15%
Largest Region: North America
Fastest Growing Region: Asia Pacific
Key Market Report Takeaways
North America accounts for the largest market share (~45-48%), supported by strong R&D infrastructure and early adoption of advanced therapies
Asia Pacific is the fastest-growing region, driven by expanding healthcare investments and clinical trial activities
Viral vector-based gene therapy products hold the highest market share due to higher delivery efficiency
Oncology applications dominate the market, reflecting high demand for targeted cancer therapies
Hospitals and specialty clinics represent the leading end-user segment due to advanced treatment capabilities
Key Market Trends & Highlights
North America dominates the global market due to strong biotechnology ecosystems, regulatory approvals, and high healthcare spending
Asia Pacific is the fastest-growing region, driven by increasing clinical trials, government initiatives, and rising patient population
Oncology remains the dominant application segment, supported by growing cancer prevalence and advancements in targeted gene therapies
Market growth is fueled by rising demand for curative treatments, favorable regulatory pathways, and increasing investment in genetic research
Emerging technologies such as CRISPR gene editing, RNA-based therapies, and next-generation vectors are reshaping treatment paradigms
Strategic collaborations, licensing agreements, and funding initiatives are accelerating product development and commercialization
Market Dynamics
Market Drivers
Rising Prevalence of Genetic and Chronic Diseases
The increasing incidence of genetic disorders, cancer, and rare diseases is a primary driver of the gene therapy market. Conditions such as hemophilia, spinal muscular atrophy, and inherited retinal diseases require long-term treatment solutions, creating demand for curative therapies. North America and Europe report higher diagnosis rates due to advanced healthcare systems. Growing patient awareness and early diagnosis are further accelerating therapy adoption globally.
Technological Advancements in Gene Editing and Delivery Systems
Rapid progress in gene-editing technologies such as CRISPR-Cas9 and improvements in viral vector delivery systems have significantly enhanced therapeutic efficiency. These advancements reduce off-target effects and improve safety profiles. Biopharmaceutical companies are increasingly investing in next-generation platforms, particularly in the U.S. and Europe. This technological evolution is expanding the scope of treatable diseases.
Increasing Investments and Funding in Biotechnology
Substantial funding from venture capital firms, governments, and pharmaceutical companies is fueling market growth. Large-scale investments in clinical trials and manufacturing capabilities are accelerating product pipelines. Asia Pacific countries, including China and India, are witnessing rising investments in biotech infrastructure. These financial inflows are enabling faster commercialization and global market expansion.
Favorable Regulatory Environment and Accelerated Approvals
Regulatory bodies such as the FDA and EMA are providing expedited approval pathways for gene therapies targeting rare diseases. Programs like orphan drug designation and breakthrough therapy designation support faster market entry. This regulatory support reduces time-to-market and encourages innovation. As a result, more companies are entering the gene therapy space.
Growing Demand for Personalized Medicine
The shift toward personalized and precision medicine is driving the adoption of gene therapies. These treatments target the root cause of diseases at the genetic level, offering long-term or permanent solutions. Patients and healthcare providers are increasingly preferring tailored therapies over conventional treatments. This trend is particularly strong in developed healthcare markets.
https://www.databridgemarketresearch.com/reports/global-gene-therapy-products-market
Market Restraints
High Cost of Gene Therapy Treatments
Gene therapies are among the most expensive medical treatments, often costing hundreds of thousands to millions of dollars per patient. High production costs, complex manufacturing processes, and limited scalability contribute to pricing challenges. This restricts accessibility, especially in developing regions. Reimbursement limitations further hinder widespread adoption.
Complex Regulatory and Approval Processes
Despite supportive frameworks, gene therapies face stringent regulatory requirements due to safety concerns. Extensive clinical trials and long approval timelines increase development costs. Regulatory variations across regions complicate global commercialization. These factors delay product launches and limit market growth.
Limited Manufacturing Capacity and Scalability Issues
The production of gene therapy products requires specialized facilities and expertise. Scaling up manufacturing while maintaining quality and consistency remains challenging. Supply chain constraints, particularly for viral vectors, impact production timelines. This limitation is more pronounced in emerging markets with inadequate infrastructure.
Safety Concerns and Risk of Adverse Effects
Potential risks such as immune reactions, off-target gene editing, and long-term safety uncertainties remain key concerns. These risks necessitate rigorous clinical testing and post-market surveillance. Negative outcomes in clinical trials can impact investor confidence. Such safety issues slow down regulatory approvals and market acceptance.
Pricing Pressure and Reimbursement Challenges
Healthcare systems face difficulties in accommodating high-cost therapies within reimbursement frameworks. Insurers often impose strict coverage criteria, limiting patient access. Pricing negotiations between manufacturers and payers create uncertainties. This challenge is particularly significant in cost-sensitive regions.
Market Opportunities
Expansion into Emerging Markets
Developing regions such as Asia Pacific and Latin America present significant growth opportunities due to increasing healthcare investments and patient populations. Governments are improving healthcare infrastructure and supporting biotech innovation. Expanding clinical trials in these regions is enhancing accessibility. This creates new revenue streams for market players.
Advancements in Non-Viral Delivery Systems
The development of non-viral delivery methods, such as lipid nanoparticles and electroporation, offers safer and more cost-effective alternatives. These technologies reduce immune response risks and improve scalability. Companies investing in these innovations can gain a competitive advantage. This segment is expected to witness rapid growth in the coming years.
Strategic Collaborations and Partnerships
Collaborations between biotech firms, pharmaceutical companies, and research institutions are accelerating product development. Licensing agreements and joint ventures enable resource sharing and risk mitigation. These partnerships are particularly prominent in North America and Europe. They play a crucial role in expanding global market reach.
Expansion of Gene Therapy Applications
Gene therapy is expanding beyond rare diseases into areas such as oncology, cardiovascular diseases, and neurological disorders. This diversification broadens the addressable market. Increasing research in multi-gene and complex disorders is unlocking new therapeutic possibilities. This trend is expected to drive long-term market growth.
Government Support and Funding Initiatives
Governments worldwide are increasing funding for genomic research and advanced therapies. Incentives such as tax benefits, grants, and public-private partnerships support innovation. Countries in Asia Pacific are actively promoting biotech sectors. These initiatives are fostering a favorable environment for market expansion.
Market Challenges
Infrastructure and Skilled Workforce Limitations
Gene therapy development requires advanced infrastructure and highly skilled professionals. Many regions lack the necessary expertise and facilities for large-scale production and administration. This creates bottlenecks in supply and limits adoption. Developing countries are particularly affected by these constraints.
Regulatory Heterogeneity Across Regions
Differences in regulatory standards across countries complicate global product launches. Companies must navigate multiple approval processes, increasing time and cost. Lack of harmonization affects clinical trial design and data acceptance. This challenge slows international market expansion.
Supply Chain Constraints for Critical Components
The availability of key raw materials, such as viral vectors and plasmids, is limited. Supply chain disruptions can delay production and increase costs. Dependence on specialized suppliers adds risk to the manufacturing process. This challenge affects both established and emerging market players.
Market Fragmentation and Competitive Pressure
The presence of numerous biotech startups and established pharmaceutical companies intensifies competition. Continuous innovation is required to maintain market position. Smaller players face challenges in scaling operations and competing with global leaders. Pricing and differentiation pressures are increasing across the market.
Economic Uncertainty and Pricing Volatility
Fluctuations in global economic conditions impact healthcare spending and investment flows. High-cost therapies are particularly sensitive to budget constraints. Pricing volatility and reimbursement uncertainties create financial risks for manufacturers. This challenge affects long-term market sustainability.
Market Segmentation & Analysis
By Product Type
Gene therapy products are segmented into viral vector-based and non-viral vector-based therapies. Viral vectors dominate the market with over 65% share due to their high efficiency in gene delivery and proven clinical success. Non-viral vectors are the fastest-growing segment, driven by safety advantages and lower immunogenicity. Increasing research in alternative delivery methods is supporting segment growth. The viral vector segment is expected to maintain dominance due to established clinical pipelines.
By Application
The market is segmented into oncology, rare diseases, cardiovascular diseases, neurological disorders, and others. Oncology leads the segment with the highest market share (~40%), driven by the increasing prevalence of cancer and demand for targeted therapies. Rare diseases represent a significant segment due to orphan drug incentives. Neurological applications are the fastest-growing, supported by advancements in gene editing. Expanding therapeutic applications are broadening market scope.
By End-User
End-users include hospitals, specialty clinics, and research institutes. Hospitals dominate the segment due to advanced infrastructure and the ability to administer complex therapies. Specialty clinics are witnessing rapid growth due to increasing outpatient procedures. Research institutes play a crucial role in clinical trials and innovation. Growing collaboration between hospitals and research centers is enhancing treatment accessibility.
By Technology
The market is segmented into gene editing technologies, gene transfer technologies, and others. Gene editing technologies, particularly CRISPR-based systems, are the fastest-growing segment due to precision and efficiency. Gene transfer technologies currently dominate due to established usage. Continuous innovation is driving adoption across multiple therapeutic areas.
Analytical Insights
Largest Segment: Viral vector-based products due to high efficiency and clinical validation
Fastest Growing Segment: Non-viral delivery systems driven by safety and scalability advantages
Oncology dominates applications due to high disease burden and investment focus
Technological advancements are accelerating growth across all segments
Regional Analysis
By geography, the market is categorized into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America holds the largest market share, contributing approximately 45-48% of global revenue. The region benefits from advanced healthcare infrastructure, strong R&D investments, and early adoption of gene therapies. The United States leads due to the presence of major biotech companies and favorable regulatory frameworks. High healthcare expenditure and strong clinical trial activity further support market dominance.
Europe
Europe represents a mature market with steady growth driven by strong regulatory support and research initiatives. Countries such as Germany, the U.K., and France are key contributors due to robust healthcare systems. Increasing investments in genomic research and collaborations between academic institutions and industry players are driving growth. The region focuses on innovation and regulatory compliance.
Asia Pacific
Asia Pacific is the fastest-growing region, driven by rapid industrialization and expanding healthcare infrastructure. Countries like China, India, and Japan are investing heavily in biotechnology and clinical trials. Government initiatives and rising patient populations are accelerating demand. Increasing awareness and affordability improvements are supporting market expansion.
Latin America
Latin America is an emerging market with gradual growth supported by improving healthcare infrastructure. Brazil and Mexico are key contributors due to increasing adoption of advanced therapies. Economic limitations and regulatory challenges restrict rapid expansion. However, growing investments and partnerships are enhancing market potential.
Middle East & Africa
The Middle East & Africa region shows steady but slower growth due to infrastructure gaps and limited access to advanced therapies. Government initiatives and private sector investments are gradually improving market conditions. Increasing healthcare spending in select countries is supporting growth. Adoption remains limited compared to developed regions.
Key Insights:
Largest Region: North America
Fastest Growing Region: Asia Pacific
Competitive Landscape
Market Structure Overview
The gene therapy products market is moderately consolidated, with a mix of global pharmaceutical leaders, specialized biotechnology firms, and emerging startups. Competition is driven by technological innovation, clinical pipeline strength, and regulatory approvals. Companies are focusing on developing advanced gene-editing platforms and expanding therapeutic applications. Competitive landscape analysis highlights strategic positioning and innovation capabilities of key players.
Key Industry Players
Leading companies focus on strong R&D capabilities, diverse product pipelines, and global market presence. Market leaders emphasize innovation and regulatory approvals, while emerging players target niche applications. Companies are strengthening their positions through collaborations and technological advancements.
List of Key Industry Players:
Novartis AG
Gilead Sciences, Inc.
Spark Therapeutics
Bluebird Bio
BioNTech SE
Pfizer Inc.
Sarepta Therapeutics
CRISPR Therapeutics
Orchard Therapeutics
Competitive Strategies
Companies are actively adopting strategies such as product innovation, mergers and acquisitions, and strategic collaborations. Continuous investment in R&D is driving new product development. Partnerships between biotech firms and research institutions are accelerating innovation. Geographic expansion and distribution network strengthening are key focus areas. These strategies enhance competitive advantage and market positioning.
Emerging Players & Market Dynamics
Startups and niche innovators are playing a crucial role in market expansion by introducing cost-effective and specialized solutions. Increasing venture capital funding is supporting new entrants. Emerging players are focusing on novel technologies and underserved therapeutic areas. This dynamic environment is intensifying competition and driving innovation across the market.
Latest Developments
January 2025 - Novartis AG: Expanded its gene therapy manufacturing capacity in the U.S., enhancing production capabilities and supporting global demand growth.
October 2024 - Pfizer Inc.: Entered a strategic collaboration to develop next-generation gene therapies for rare diseases, strengthening its pipeline and innovation capabilities.
June 2024 - CRISPR Therapeutics: Received regulatory approval for a CRISPR-based therapy, marking a significant milestone in gene editing commercialization.
March 2024 - BioNTech SE: Invested in mRNA-based gene therapy platforms, expanding its technology portfolio and addressing broader therapeutic areas.
November 2023 - Gilead Sciences, Inc.: Acquired a biotech firm specializing in viral vector technology, enhancing its gene therapy capabilities.
August 2023 - Sarepta Therapeutics: Launched a new gene therapy for muscular dystrophy, strengthening its position in the rare disease segment.
May 2023 - Bluebird Bio: Secured funding for expanding its gene therapy pipeline, supporting clinical trials and commercialization efforts.
https://www.databridgemarketresearch.com/reports/global-gene-therapy-market
https://www.databridgemarketresearch.com/reports/global-cell-line-development-market
https://www.databridgemarketresearch.com/reports/global-pharmaceutical-quality-control-market
https://www.databridgemarketresearch.com/reports/global-spatial-genomics-and-transcriptomics-market
https://www.databridgemarketresearch.com/reports/global-chemoinformatics-market
https://www.databridgemarketresearch.com/reports/global-blood-culture-test-market
https://www.databridgemarketresearch.com/reports/global-antispasmodics-drugs-market
https://www.databridgemarketresearch.com/reports/global-electronic-medical-records-emr-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Gene Therapy Products Market to Reach USD 40.86 Billion by 2033, Growing at 22.15% CAGR from 2025 here
News-ID: 4484945 • Views: …
More Releases from Data Bridge Market Research
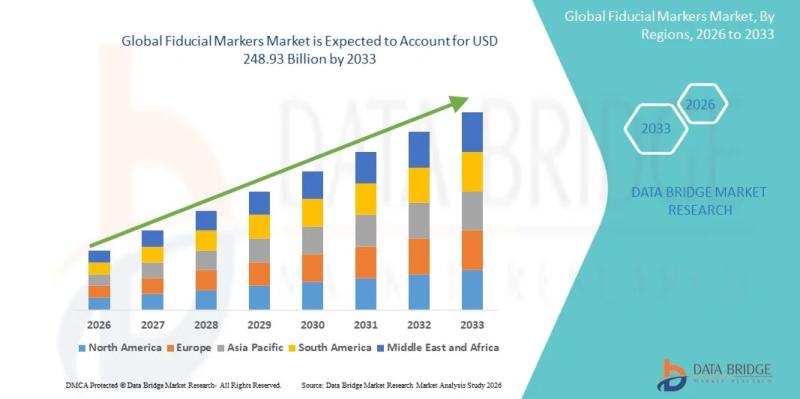
Fiducial Markers Market Analysis Report: Opportunities, Challenges, and Strategi …
As per Data Bridge Market Research analysis, the Fiducial Markers Market was estimated at USD 180.50 billion in 2025. The market is expected to grow from USD 187.90 billion in 2026 to USD 248.93 billion in 2033, at a CAGR of 4.10% during the forecast period with driven by the rising demand for advanced imaging technologies, increasing prevalence of cancer requiring precise radiotherapy, growing adoption of minimally invasive procedures, and…
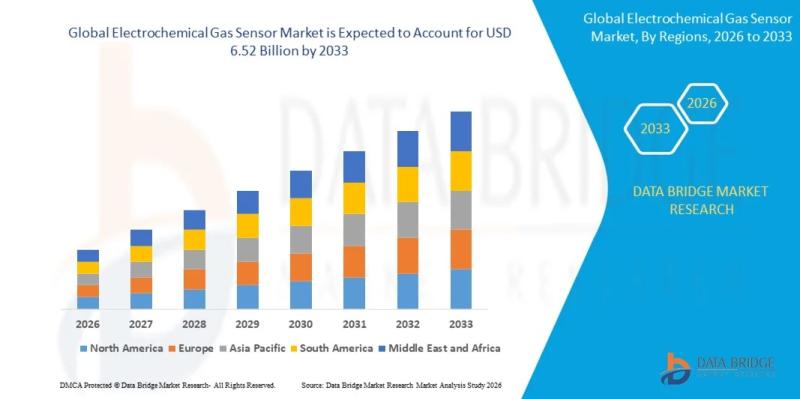
Electrochemical Gas Sensor Market Outlook: Demand, Innovation, and Opportunities
As per Data Bridge Market Research analysis, the Electrochemical Gas Sensor Market was estimated at USD 3.47 billion in 2025. The market is expected to grow from USD 3.75 billion in 2026 to USD 6.52 billion in 2033, at a CAGR of 8.20% during the forecast period with driven by the rising demand for industrial safety monitoring, environmental regulations, and increasing adoption of gas detection technologies across multiple end-use industries.
Growth…
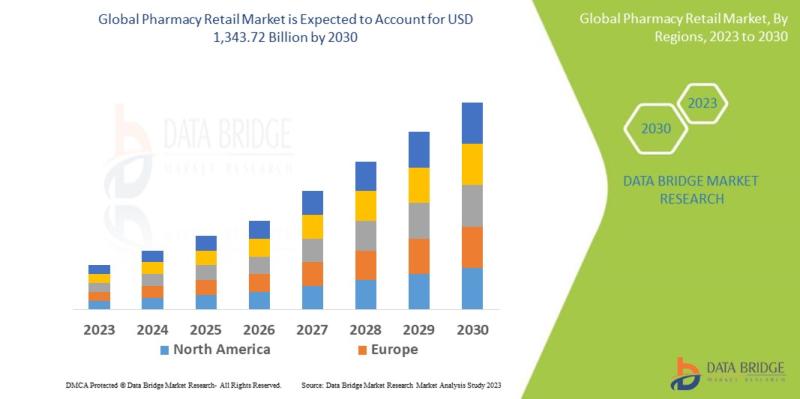
Global Pharmacy Retail Market Set to Grow at a CAGR of 10.10% from 2026 to 2030, …
Market Summary
As per Data Bridge Market Research analysis, the Pharmacy Retail Market was estimated at USD 830.80 billion in 2025. The market is expected to grow from USD 915.70 billion in 2026 to USD 1,343.72 billion in 2030, at a CAGR of 10.10% during the forecast period with driven by the rising demand for accessible healthcare services, expansion of e-pharmacy platforms, and increasing prevalence of chronic diseases.
Get the full PDF…
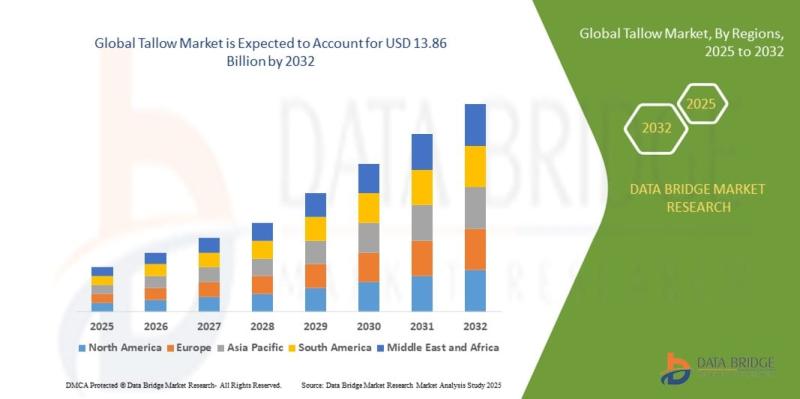
Strategic Insights into the Tallow Market: Trends, Forecast, and Competitive Lan …
As per Data Bridge Market Research analysis, the Tallow Market was estimated at USD 9.07 billion in 2025. The market is expected to grow from USD 9.63 billion in 2026 to USD 13.86 billion in 2032, at a CAGR of 6.2% during the forecast period with driven by the rising demand for bio-based products, expanding applications in biodiesel production, and increasing utilization in personal care and animal feed industries.
The market…
More Releases for Gene
DNA and Gene Cloning Services Market Expands with Growing Focus on Complex Gene …
InsightAce Analytic Pvt. Ltd. has announced the publication of a market research report titled "Global DNA and Gene Cloning Services Market by Type of Service Offered (Custom Cloning, Sub-cloning, Gene Synthesis, and Others), Type of Gene (Complex Gene, Standard Gene, and Others), End-User Industry (Pharmaceutical, Academic and R&D, and Biotechnology Companies, and Others)- Market Outlook and Industry Analysis 2034"
The DNA and Gene Cloning Services Market Size is valued…
Evolving Market Trends In The CRISPR Gene Editing Industry: Innovative Gene Ther …
The CRISPR Gene Editing Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Expected CRISPR Gene Editing Market Size During the Forecast Period?
The CRISPR gene editing market has grown exponentially in recent years. It will grow from $2.26 billion in 2024 to $2.88…
Cell and Gene Therapy Market Global Analysis By Type (Cell Therapy, Gene Therapy …
Gene and cell therapy uses genes and cells for the treatment of genetic diseases. Genetic diseases are caused by mutations, or errors in genes which can be passed down from one generation to another. Gene therapy aims to treat diseases by using genetic material, or DNA, to manipulate a patient's cells by replacing, changing or introducing genome into cells- either internally or externally. Cell therapy aims to treat diseases by…
Competitive and Opportunities Analysis of Gene Therapy Market of Gene Therapy Ma …
Global Gene Therapy Market accounted for US$ 2.05 billion in 2020 and is estimated to be US$ 12.29 billion by 2030 and is anticipated to register a CAGR of 19.8%. Gene therapy means fixing a working gene to an individual who features a damaged gene. The European Commission has approved this method for one particular treatment. The treatment by the merchandise Glybera uses an epidemic to infect muscle cells with…
Genetic Testing Market Size by Growth Opportunities, Top Key Players: GeneDx, In …
Genetic Testing Market Report provides an in-depth analysis of the overall market, The ripple effect of Coronavirus-Covid19 on the market needs to become part of strategy discussions to emerge strong. The report focuses on major key players, production details, their application, countries and also analyzes the global and key regions market potential and advantage, opportunity, and challenge, restraints, and risks.
The Top players Covered in report are GeneDx, Invitae, Pathway Genomics,…
Gene Synthesis Market by Top Manufacturers - Gene script, Gene Art (Thermofische …
The "Gene Synthesis Market" report Added by "Big Market Research", enumerates information about the industry in terms of market share, market size, revenue forecasts, and regional outlook. The report further illustrates competitive insights of key players in the business vertical followed by an overview of their diverse portfolios and growth strategies.
In the Gene Synthesis Market 2018 research report professionals describe the different facets of the industry with a specific goal…