Press release
Parkinson's Disease Pipeline Accelerates with 200+ Therapies and 150+ Companies Driving Next-Gen Innovations | DelveInsight
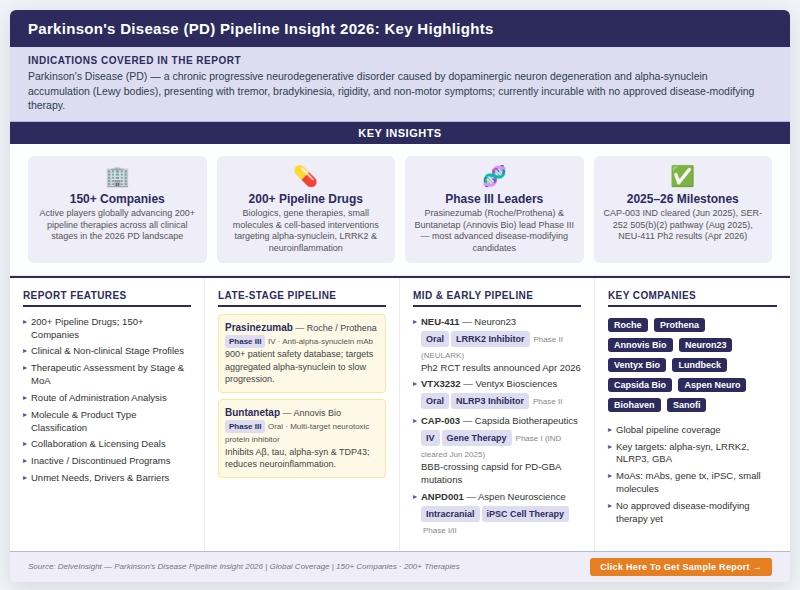
Key Parkinson's Disease Companies are Roche, Prothena Biosciences, Biohaven Pharmaceuticals, Neuraly Inc., Cerevance, IRLAB, Neuron23, Ventyx Biosciences, Lundbeck, Sanofi, Eli Lilly and Company, and othersThe Parkinson's disease therapeutic landscape is undergoing a transformative shift as over 150 leading biopharmaceutical companies actively advance more than 200 pipeline therapies targeting the underlying mechanisms of neurodegeneration. From disease-modifying biologics and gene therapies to innovative small molecules and cell-based interventions, companies such as Roche, Prothena Biosciences, Annovis Bio, Neuron23, Ventyx Biosciences, and Lundbeck are driving unprecedented innovation in Parkinson's disease (PD) research and development.
DelveInsight's latest report, "Parkinson's Disease Pipeline Insight, 2026, " delivers a comprehensive analysis of this rapidly evolving space, offering detailed insights into clinical and nonclinical drug candidates, emerging therapeutic strategies, and competitive intelligence across the global Parkinson's disease pipeline. The report further evaluates therapeutic segmentation by product type, development stage, route of administration, and molecule type, while also shedding light on inactive and discontinued programs.
Discover the complete Parkinson's Disease pipeline landscape and emerging therapeutic opportunities: Parkinson's Disease Pipeline Outlook [https://www.delveinsight.com/report-store/parkinsons-disease-pipeline-insights?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Understanding Parkinson's Disease: A Progressive Neurodegenerative Challenge
Parkinson's disease is a chronic and progressive neurological disorder that primarily impairs motor function due to the degeneration of dopamine-producing neurons in the brain. The condition is characterized by hallmark symptoms such as resting tremors, bradykinesia, muscular rigidity, and postural instability. As the disease progresses, patients also experience a wide range of non-motor symptoms, including cognitive decline, depression, anxiety, sleep disturbances, and autonomic dysfunction.
The underlying pathology of Parkinson's disease is multifactorial, involving both genetic predispositions and environmental triggers. Mutations in genes such as SNCA, LRRK2, PARK2, PINK1, and DJ-1 are associated with familial forms of the disease, while environmental exposures to toxins and pesticides further elevate risk. A key pathological hallmark is the accumulation of misfolded alpha-synuclein protein, leading to the formation of Lewy bodies that contribute to neuronal dysfunction and progressive neurodegeneration.
Despite advances in understanding disease biology, Parkinson's disease remains incurable. Current treatment approaches are largely symptomatic, relying on dopaminergic therapies such as levodopa, dopamine agonists, MAO-B inhibitors, and COMT inhibitors. In advanced stages, surgical interventions like deep brain stimulation may be employed. However, the lack of disease-modifying therapies continues to represent a significant unmet need, driving robust pipeline innovation.
Evolving Parkinson's Disease Pipeline: Innovation Redefining Therapeutic Strategies
The Parkinson's disease pipeline is witnessing a paradigm shift as research increasingly focuses on disease-modifying approaches rather than symptomatic relief. Emerging therapies are targeting critical pathways such as alpha-synuclein aggregation, neuroinflammation, mitochondrial dysfunction, and genetic mutations associated with disease progression.
Request a sample report to explore breakthrough innovations in Parkinson's disease therapeutics: Parkinson's Disease Clinical Trial Analysis [https://www.delveinsight.com/sample-request/parkinsons-disease-pipeline-insights?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
According to DelveInsight, more than 150 companies are actively engaged in Parkinson's disease drug development, collectively advancing over 200 pipeline candidates across various stages of clinical development. These therapies span early discovery phases to late-stage clinical trials, highlighting a strong and diversified research ecosystem.
Key Takeaways from the Parkinson's Disease Pipeline Report
* The Parkinson's disease pipeline comprises 200+ therapeutic candidates across all stages of development.
* Over 150 companies are actively contributing to innovation in this space.
* Increasing focus on disease-modifying therapies targeting alpha-synuclein, neuroinflammation, and genetic pathways.
* Rising interest in precision medicine approaches for genetically defined patient populations.
* Significant growth in biologics, gene therapies, and cell-based therapies.
* Strategic collaborations, licensing deals, and mergers are accelerating drug development timelines.
* Expanding clinical trial activity across global regions, incorporating advanced endpoints and biomarkers.
* In April 2026, Biohaven Pharmaceuticals, Inc announced results of a Phase 2/3, Double-Blind, Placebo-Controlled Study of BHV-8000 in Participants With Early Parkinson's Disease
* In April 2026, H. Lundbeck A/S announced results of an Interventional, Open-label, Trial Investigating the Safety, Tolerability, and Pharmacokinetic Properties of Lu AF28996 in Chinese Men and Women With Parkinson's Disease
* In April 2026, Neuron23 Inc announced results of a Phase 2, Randomized, Double-Blind, Placebo-Controlled Study and Open-Label Extension to Evaluate the Safety and Efficacy of NEU-411 in Companion Diagnostic-Positive Participants With Early Parkinson's Disease (NEULARK)
* In August 2025, Serina Therapeutics announced FDA support to advance SER-252 (POZ-apomorphine) into a registrational study for advanced Parkinson's via the 505(b)(2) NDA pathway.
* In July 2025, NKGen Biotech received FDA authorization for an Expanded Access Program (EAP) to use its NK cell therapy, troculeucel, in multiple neurodegenerative diseases-including Alzheimer's, Parkinson's, ALS, MSA, PSP, FTD, MS, and Lewy Body Dementia-beyond its current Phase 2a trial in moderate-stage Alzheimer's disease.
* In July 2025, the FDA cleared the new Magstim Rapid TMS system for research, clinical use, and treatment of pain. This next-generation non-invasive neuromodulation device supports studies and therapy for conditions including depression, OCD, Alzheimer's, Parkinson's, autism, and stroke, featuring upgraded touchscreen, user-friendly software, and improved data management.
* In June 2025, Capsida Biotherapeutics announced FDA clearance of its IND application for CAP-003, an IV-administered gene therapy for Parkinson's disease with GBA mutations (PD-GBA). This marks Capsida's second wholly owned clinical program using its proprietary BBB-crossing capsid and detargeted gene cargo, manufactured in its GMP facility.
Parkinson's Disease Emerging Drugs Profile: Advancing Toward Disease Modification
The report provides detailed insights into key pipeline candidates that are shaping the future of Parkinson's disease treatment.
Prasinezumab: Roche/Prothena Biosciences
Prasinezumab is a monoclonal antibody designed to target aggregated alpha-synuclein, a key driver of Parkinson's disease pathology. By reducing protein aggregation and limiting its spread across neurons, the therapy aims to slow disease progression. With an extensive safety database involving over 900 patients, the drug is currently in Phase III clinical development, positioning it among the most advanced disease-modifying candidates.
Buntanetap: Annovis Bio
Buntanetap represents a multi-target approach to neurodegeneration, inhibiting the formation of several neurotoxic proteins including amyloid beta, tau, alpha-synuclein, and TDP43. By improving neuronal function and reducing inflammation, the drug aims to restore brain health. It is currently progressing through Phase III clinical trials.
NEU 411: Neuron23
NEU 411 is a brain-penetrant LRRK2 inhibitor targeting genetically defined Parkinson's disease populations. By addressing LRRK2 overactivity, the therapy aims to slow disease progression in patients with specific mutations. It is currently in Phase II clinical trials.
VTX3232: Ventyx Biosciences
VTX3232 targets the NLRP3 inflammasome, a key regulator of neuroinflammation. By reducing microglial activation and inflammatory signaling, the therapy aims to address both motor and non-motor symptoms while potentially modifying disease progression. It is currently in Phase II development.
ANPD001: Aspen Neuroscience
ANPD001 is an innovative autologous cell therapy derived from induced pluripotent stem cells. By generating dopaminergic neurons from a patient's own cells and transplanting them into the brain, this approach aims to restore lost neuronal function. The therapy is currently in Phase I/II clinical trials.
Lu AF28996: Lundbeck
Lu AF28996 is a dual dopamine receptor agonist targeting both D1 and D2 receptors to restore dopaminergic signaling. This mechanism aims to improve motor symptoms while potentially offering enhanced efficacy compared to traditional therapies. The drug is currently in early-stage clinical development.
Download free sample report to know in detail: Parkinson's Disease Drugs and Therapies [https://www.delveinsight.com/sample-request/parkinsons-disease-pipeline-insights?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Parkinson's Disease Pipeline Segmentation and Therapeutic Assessment
The report offers a comprehensive segmentation of pipeline therapies across multiple dimensions: Clinical Development Phases
* Late-stage (Phase III)
* Mid-stage (Phase II)
* Early-stage (Phase I)
* Preclinical and discovery-stage candidates
* Inactive and discontinued programs
Parkinson's Disease Route of Administration
* Oral
* Intravenous
* Subcutaneous
* Parenteral
* Topical
Parkinson's Disease Molecule Types
* Small molecules
* Monoclonal antibodies
* Gene therapies
* Peptides
* Polymer-based therapies
* Recombinant fusion proteins
Parkinson's Disease Product Types
- Monotherapy
- Combination therapy
- Mono/combination approaches
This diverse therapeutic landscape reflects the complexity of Parkinson's disease and the need for multifaceted treatment strategies.
Parkinson's Disease Clinical Trial and Development Activities
The Parkinson's disease pipeline is characterized by a surge in clinical trial activity and strategic collaborations. Companies are increasingly partnering with academic institutions and research organizations to accelerate innovation. Licensing agreements, mergers, and acquisitions are further strengthening R&D capabilities.
Key players such as Roche, Prothena Biosciences, Annovis Bio, Neuron23, and Ventyx Biosciences are leading the development of advanced therapies targeting distinct disease pathways. Meanwhile, emerging biotech firms are exploring novel approaches such as gene editing, stem cell therapy, and microbiome modulation.
Stay ahead with in-depth clinical trial insights and partnership analysis: Parkinson's Disease Companies and FDA approvals [https://www.delveinsight.com/sample-request/parkinsons-disease-pipeline-insights?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Parkinson's Disease Market Drivers, Challenges, and Future Outlook
The growth of the Parkinson's disease pipeline is driven by several key factors, including the rising prevalence of the disease, increasing aging population, and advancements in neuroscience research. The growing demand for disease-modifying therapies is further fueling investment and innovation.
However, challenges remain. The heterogeneity of Parkinson's disease, lack of reliable biomarkers, and complexity of clinical trial design continue to hinder progress. Additionally, high development costs and regulatory uncertainties pose significant barriers.
Despite these challenges, the future outlook remains highly promising. Advances in artificial intelligence, biomarker discovery, and precision medicine are expected to revolutionize Parkinson's disease treatment. The integration of digital health technologies and real-world evidence is also enhancing clinical trial efficiency and patient outcomes.
Scope of the Parkinson's Disease Pipeline Report
* Coverage: Global
* Key Parkinson's Disease Companies: Roche, Prothena Biosciences, Biohaven Pharmaceuticals, Neuraly Inc., Cerevance, IRLAB, Neuron23, Ventyx Biosciences, Lundbeck, Sanofi, Eli Lilly and Company, and others
* Key Parkinson's Disease Therapies: Prasinezumab, Buntanetap, NEU 411, VTX3232, ANPD001, Lu AF28996, and more
* Therapeutic Assessment: By product type, clinical stage, route of administration, and molecule type
Request for free sample report: Parkinson's Disease Clinical Trial Studies and Approvals [https://www.delveinsight.com/sample-request/parkinsons-disease-pipeline-insights?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Table of Contents
1. Introduction
2. Executive Summary
3. Parkinson's Disease Overview
4. Pipeline Therapeutics
5. Therapeutic Assessment
6. Analytical Perspective
7. Late-Stage Products
8. Mid-Stage Products
9. Early-Stage Products
10. Inactive Products
11. Key Companies
12. Key Products
13. Unmet Needs
14. Market Drivers and Barriers
15. Future Perspectives
Transforming the Future of Parkinson's Disease Treatment
The Parkinson's disease pipeline is at a pivotal moment, marked by scientific breakthroughs and a strong commitment to addressing unmet clinical needs. With over 200 therapies under development and increasing focus on disease modification, the next decade is expected to bring transformative advancements in patient care.
Early diagnosis, timely intervention, and access to innovative therapies will be critical in improving patient outcomes and quality of life. As research continues to evolve, the integration of cutting-edge technologies and personalized medicine approaches will redefine the treatment paradigm for Parkinson's disease.
About DelveInsight
DelveInsight is a leading Life Science market research and business consulting company recognized for its off-the-shelf syndicated market research reports and customized solutions to firms in the healthcare sector.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Ankit Nigam
Email:Send Email [https://www.abnewswire.com/email_contact_us.php?pr=parkinsons-disease-pipeline-accelerates-with-200-therapies-and-150-companies-driving-nextgen-innovations-delveinsight]
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Albany
State: New York
Country: United States
Website: https://www.delveinsight.com/consulting
Legal Disclaimer: Information contained on this page is provided by an independent third-party content provider. ABNewswire makes no warranties or responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you are affiliated with this article or have any complaints or copyright issues related to this article and would like it to be removed, please contact retract@swscontact.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Parkinson's Disease Pipeline Accelerates with 200+ Therapies and 150+ Companies Driving Next-Gen Innovations | DelveInsight here
News-ID: 4484613 • Views: …
More Releases from ABNewswire
Focal Segmental Glomerulosclerosis Market to Surpass USD 837 Million by 2034, Gr …
Key companies actively involved in the Focal Segmental Glomerulosclerosis (FSGS) market include Travere Therapeutics, Dimerix, Goldfinch Bio, Vertex Pharmaceuticals, GlaxoSmithKline, Dimerix Bioscience Pty Ltd, ChemoCentryx, AstraZeneca, Genentech, Inc., Sanofi, Mallinckrodt ARD LLC, ACELYRIN Inc., Kyowa Kirin Co., Ltd., Bristol-Myers Squibb, among others.
The FSGS market across the seven major markets (7MM) was valued at nearly USD 734 million in 2022 and is anticipated to grow steadily throughout the forecast period.
DelveInsight's Focal…

EMPAVELI Sales Forecast to 2034 Signals Strong Growth Across PNH and Rare Kidney …
The global rare disease therapeutics landscape is undergoing a profound transformation, and at the center of this evolution is EMPAVELI (pegcetacoplan) - a first-in-class complement C3 inhibitor that is redefining treatment paradigms across hematology and nephrology.
According to DelveInsight's latest report, "EMPAVELI Sales Forecast, and Market Size Analysis - 2034," the therapy is poised for sustained commercial growth, driven by strong clinical differentiation, expanding indications, and increasing physician adoption.
As the healthcare…
Psoriatic Arthritis Market to Exceed USD 15 Billion by 2034 with 20+ Emerging Th …
Leading Psoriatic Arthritis companies working in the market are UCB Biopharma, Sun Pharmaceutical Industries Limited, BMS, Affibody AB, Janssen Biotech, Amgen, Eli Lilly and Company, Teva Pharma, Boehringer Ingelheim, AbbVie, Roche, Ampio Pharmaceuticals, Antares Pharma, Currax Pharmaceuticals, GlaxoSmithKline, Bayer, Sanofi, Astrazeneca, Johnson & Johnson Services, Pfizer and others.
The psoriatic arthritis (PsA) market is supported by well-established therapies such as HUMIRA, OTEZLA, COSENTYX, CIMZIA, and BIMZELX, which continue to dominate current…

Multiple Myeloma Treatment Pipeline Shows Strong Momentum as 75+ Pharma Companie …
DelveInsight's "Multiple Myeloma Pipeline Insights 2026" report provides comprehensive insights about 75+ Multiple Myeloma Companies and 80+ pipeline drugs in the Multiple Myeloma pipeline landscape. It covers the Multiple Myeloma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Multiple Myeloma therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Stay ahead in understanding…
More Releases for Parkinson
Key Trends Reshaping the Wolff Parkinson White Syndrome Market: Technological Ad …
Stay ahead with our updated market reports featuring the latest on tariffs, trade flows, and supply chain transformations.
Wolff Parkinson White Syndrome Market Size Growth Forecast: What to Expect by 2025?
The market for Wolff Parkinson White syndrome has registered consistent growth over the recent past. The expectation is that it will expand from $1.17 billion in 2024 to $1.22 billion in 2025, reflecting a compound annual growth rate (CAGR) of 4.5%.…
Evolving Market Trends In The Anti-Parkinson Drugs Industry: Innovative Advancem …
The Anti-Parkinson Drugs Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Expected Anti-Parkinson Drugs Market Size During the Forecast Period?
The anti-parkinson drugs market has seen significant growth in recent years, projected to increase from $10.37 billion in 2024 to $11.08 billion in…
What's Driving the Anti-Parkinson Drugs Market 2025-2034: Increasing Geriatric P …
What Are the Projections for the Size and Growth Rate of the Anti-Parkinson Drugs Market?
In recent times, the market size for anti-Parkinson drugs has experienced robust growth. The market, which is projected to rise from $10.37 billion in 2024 to $11.08 billion in 2025, boasts a compound annual growth rate (CAGR) of 6.9%. The past growth trend can be credited to an aging demographic, a surge in disease incidence, enhanced…
What's Driving the Anti-Parkinson Drugs Market 2025-2034: Increasing Geriatric P …
What Are the Projections for the Size and Growth Rate of the Anti-Parkinson Drugs Market?
In recent times, the market size for anti-Parkinson drugs has experienced robust growth. The market, which is projected to rise from $10.37 billion in 2024 to $11.08 billion in 2025, boasts a compound annual growth rate (CAGR) of 6.9%. The past growth trend can be credited to an aging demographic, a surge in disease incidence, enhanced…
Wolff Parkinson White Syndrome Treatment Market Overview 2024-2033
The Business Research Company has recently revised its global market reports, now incorporating the most current data for 2024 along with projections extending up to 2033.
Wolff Parkinson White Syndrome Global Market Report 2024 by The Business Research Company offers comprehensive market insights, empowering businesses with a competitive edge. It includes detailed estimates for numerous segments and sub-segments, providing valuable strategic guidance.
The Market Size Is Expected To Reach $1.43 billion…
Anti-Parkinson Drugs Market Overview
Exhibiting robust growth, the anti-parkinson drugs market is set to escalate from $9.73 billion in 2023 to $10.43 billion in 2024, with a notable Compound Annual Growth Rate (CAGR) of 7.2%. The trajectory continues on an upward trend, with an anticipated market size of $13.36 billion by 2028, sustaining a robust CAGR of 6.4%.
Increasing Geriatric Population Driving Demand:
The burgeoning geriatric population, coupled with a surge in Parkinson's disease cases,…