Press release
Cost Analysis for Medical Supplies Manufacturing Plant 2026: Setup and Market Trends
Setting up a medical supplies manufacturing plant positions investors in one of the most resilient and essential segments of the global healthcare value chain, backed by sustained growth driven by rising healthcare expenditure, aging populations, expanding hospital infrastructure, increasing prevalence of chronic diseases, and the growing need for infection prevention and patient safety products. As healthcare systems worldwide scale up, governments prioritize domestic manufacturing of critical medical goods, and homecare adoption accelerates, the medical supplies industry continues to present compelling opportunities for manufacturers and entrepreneurs seeking long-term profitability in a mission-critical, demand-stable sector.Market Overview and Potential Growth:
The global medical supplies market demonstrates a steady growth trajectory, valued at USD 151.78 Billion in 2025. According to IMARC Group's comprehensive market analysis, the market is expected to reach USD 215.29 Billion by 2034, exhibiting a CAGR of 3.96% from 2026 to 2034. The market is primarily driven by rising global healthcare expenditure, a growing burden of chronic diseases, the expansion of hospital and outpatient care infrastructure, and increasing demand for single-use, sterile, and home-use medical consumables.
Request Sample: https://www.imarcgroup.com/medical-supplies-manufacturing-plant-project-report/requestsample
Medical supplies function as essential healthcare products used for the diagnosis, treatment, prevention, and monitoring of medical conditions. The production of these products requires a diverse set of raw materials, including non-woven fabric, gauze, medical-grade polymers (PVC, polypropylene, polyethylene), latex and nitrile, adhesives, elastic, sterilization chemicals, and pharmaceutical-grade packaging materials. Medical supplies serve essential functions in wound care, surgical procedures, infection control, fluid management, and patient monitoring. These products provide safety, sterility, and reliability for healthcare providers across applications ranging from routine primary care to complex surgical interventions, critical care, diagnostics, and homecare settings.
Modern medical supplies manufacturing encompasses disposable syringes, IV sets, catheters, surgical gloves, masks and PPE, bandages and wound dressings, sterile drapes, blood collection tubes, and a wide range of diagnostic consumables, each designed to meet strict regulatory and performance requirements. High-grade sterile products are manufactured under controlled clean-room environments conforming to ISO 13485, CE, USFDA, and WHO-GMP standards, ensuring consistent safety and quality. These supplies serve vital functions that help prevent hospital-acquired infections, enable accurate diagnosis, support surgical and emergency interventions, and ensure reliable patient care across the continuum of health services.
The market for medical supplies is experiencing stable growth due to the proliferation of hospitals, outpatient clinics, diagnostic labs, and homecare services worldwide.
The increasing prevalence of diabetes, cardiovascular disease, cancer, and respiratory conditions is expanding the need for long-term medical consumables, while aging populations in North America, Europe, and parts of Asia are driving demand for chronic-care and homecare products. For instance, the International Diabetes Federation projects that the global number of adults living with diabetes will continue to rise and reach nearly 853 million by 2050, underscoring a long-term demand base for insulin delivery devices, lancets, glucose strips, and related consumables. The industry outlook is further strengthened by government initiatives that support domestic healthcare manufacturing, reduce import dependence on critical medical products, and build resilient medical supply chains following lessons learned during recent global health disruptions.
Plant Capacity and Production Scale:
The proposed medical supplies manufacturing facility is designed with an annual production capacity ranging between 5 million - 20 million units, enabling economies of scale while maintaining operational flexibility. This capacity range allows producers to serve diverse end-user segments across hospitals, clinics, outpatient care centers, diagnostic labs, and homecare - ensuring steady demand and consistent revenue streams driven by rising healthcare expenditure, infection-prevention protocols, aging populations, expanding insurance coverage, and applications in wound care, surgical procedures, infusion therapy, diagnostics, personal protection, and routine patient care.
Speak to an Analyst: https://www.imarcgroup.com/request?type=report&id=22034&flag=C
Financial Viability and Profitability Analysis:
The medical supplies manufacturing business demonstrates healthy profitability potential under normal operating conditions. The financial projections reveal:
• Gross Profit Margins: 45-55%
• Net Profit Margins: 15-25%
These margins are supported by stable demand across hospitals, clinics, diagnostic centers, and homecare providers, value-added processing through clean-room molding, assembly, sterilization, and packaging lines that deliver consistent quality at scale, and the indispensable nature of medical supplies - where syringes, catheters, gloves, PPE, and wound care products are consumed routinely in every healthcare encounter and cannot be substituted. Because the majority of medical supplies are single-use consumables, demand is recurring and largely insulated from economic cycles, making the sector one of the more stable investment opportunities in healthcare manufacturing. The project demonstrates strong return on investment (ROI) potential with comprehensive financial analysis.
Cost of Setting Up a Medical Supplies Manufacturing Plant:
Operating Cost Structure:
Understanding the operating expenditure (OpEx) is crucial for effective financial planning. The cost structure includes:
• Raw Materials: 50-60% of total OpEx
• Utilities: 10-15% of OpEx
• Other Expenses: Labor, packaging, transportation, maintenance, depreciation, taxes
Raw materials at 50-60% of operating costs, with non-woven fabric and gauze as primary components, along with medical-grade polymers, latex/nitrile, adhesives, elastic, sterilization chemicals, and pharmaceutical-grade packaging materials. Utilities at 10-15%, largely driven by clean-room HVAC, sterilization (EtO, gamma, or steam), and compressed air requirements. By the fifth year, total operational cost is expected to increase substantially due to inflation, market fluctuations, and rising input material prices. Long-term contracts with reliable suppliers help stabilize pricing and ensure a steady supply of critical raw materials.
Capital Investment Requirements:
Setting up a medical supplies manufacturing plant requires substantial capital investment. The total investment depends on plant capacity, product mix, technology level, and location.
Land and Site Development: Location must offer easy access to key raw materials such as non-woven fabric, medical-grade polymers, gauze, adhesives, elastic, and packaging. Proximity to hospitals, distributors, and export gateways minimizes distribution costs. Robust infrastructure is essential, including reliable utilities, transportation access, and waste management systems. Compliance with local zoning laws, environmental regulations, and pharmaceutical manufacturing norms must also be ensured.
Machinery and Equipment: Machinery costs account for the largest portion of capital expenditure. Essential equipment includes:
• Injection molding machines
• Clean room facilities (ISO Class 7/8)
• Automated assembly lines
• Ethylene oxide (EtO) / gamma / steam sterilization chambers
• Testing and calibration instruments
• Automated blister and pouch packaging systems
Civil Works: Building construction with clean-room envelopes, controlled access, and optimized layouts. Separate zones must be designated for raw material storage, injection molding and component manufacturing, clean-room assembly, sterilization, quality control, and finished goods warehousing. Space for future expansion should also be factored in.
Buy Report Now: https://www.imarcgroup.com/checkout?id=22034&method=2175
Major Applications and Market Segments:
Medical supplies serve extensive applications across the healthcare ecosystem:
• Hospitals and Surgical Centers: Used in operating theaters, intensive care units, and wards in the form of sterile drapes, surgical gloves, sutures, syringes, IV sets, and wound care products
• Outpatient and Primary Care Clinics: Routine consumables such as examination gloves, bandages, dressings, injection devices, and diagnostic supplies
• Diagnostic Laboratories: Blood collection tubes, swabs, specimen containers, and precision disposables used across pathology, microbiology, and molecular diagnostics
• Homecare Settings: Insulin syringes, glucose test strips, incontinence products, wound dressings, respiratory masks, and mobility aids enabling care outside hospital environments
Process flow: Raw material preparation, injection molding / component fabrication, assembly of components, clean-room packaging, sterilization (EtO / gamma / steam), quality inspection, labeling, and secondary/tertiary packaging.
Why Invest in Medical Supplies Manufacturing?
Several compelling factors support investment in this sector:
• Essential and Recurring Demand: Medical supplies are consumed in every healthcare encounter, from routine checkups to major surgeries, making demand recurring, largely non-cyclical, and resilient to economic downturns
• Alignment with Global Megatrends: Aging populations, rising chronic disease burden, expanding health insurance coverage, and growing homecare adoption create sustained long-term need for medical consumables and devices
• Supply Chain Localization Advantage: Governments and hospital buyers increasingly prefer regional suppliers to ensure product availability, reduce lead times, and safeguard against global supply shocks such as those experienced during recent pandemic events
• Supportive Government Policies: Production-linked incentive schemes, import substitution programs, and investments in public healthcare infrastructure across emerging economies create favorable conditions for medical supplies manufacturing
• Scalable and Automation-Ready Production: Injection molding, automated assembly, and sterilization lines enable high-volume production with consistent quality and competitive unit economics, allowing manufacturers to serve domestic and export markets profitably
Manufacturing Process Excellence:
The production of medical supplies is a multi-step, tightly controlled operation:
• Raw material preparation and inspection (polymers, fabrics, adhesives, packaging)
• Injection molding and component fabrication
• Sub-assembly of components (needles, barrels, connectors, tubing)
• Clean-room assembly and inspection
• Primary packaging in sterile pouches or blisters
• Sterilization using EtO, gamma radiation, or steam
• Quality inspection, biocompatibility, and sterility assurance testing
• Labeling, secondary packaging, and dispatch
Comprehensive quality control is maintained throughout production. Analytical and validation instruments monitor sterility assurance levels (SAL), biocompatibility, tensile and mechanical integrity, and long-term stability in line with ISO 13485, CE, USFDA, and WHO-GMP standards.
Industry Leadership:
Leading manufacturers in the global medical supplies industry include:
• Medtronic plc
• Baxter International Inc.
• 3M Health Care
• Johnson & Johnson
• Stryker Corporation
All serve end-use sectors such as hospitals, outpatient care centers, diagnostic laboratories, emergency services, and homecare providers.
Recent Industry Developments:
July 2025: AstraZeneca announced a USD 50 Billion investment in the United States by 2030, building on America's global leadership in medicines manufacturing and R&D. The investment is expected to create tens of thousands of highly skilled direct and indirect jobs and expand capacity for next-generation medicines and related medical supplies serving patients in the U.S. and worldwide.
February 2025: Eli Lilly and Company announced plans to bolster its domestic medicine production across therapeutic areas by building four new pharmaceutical manufacturing sites in the United States, bringing the company's total U.S. capital expansion commitments to more than USD 50 Billion since 2020. The expansion is expected to strengthen domestic manufacturing capacity for injectable therapies and their associated medical supplies ecosystem.
Browse Full Report: https://www.imarcgroup.com/medical-supplies-manufacturing-plant-project-report
About Us:
IMARC Group is a global management consulting firm that helps the world's most ambitious changemakers to create a lasting impact. The company excels in understanding its client's business priorities and delivering tailored solutions that drive meaningful outcomes. We provide a comprehensive suite of market entry and expansion services. Our offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape, and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: (+1-201-971-6302)
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Cost Analysis for Medical Supplies Manufacturing Plant 2026: Setup and Market Trends here
News-ID: 4483323 • Views: …
More Releases from IMARC Group
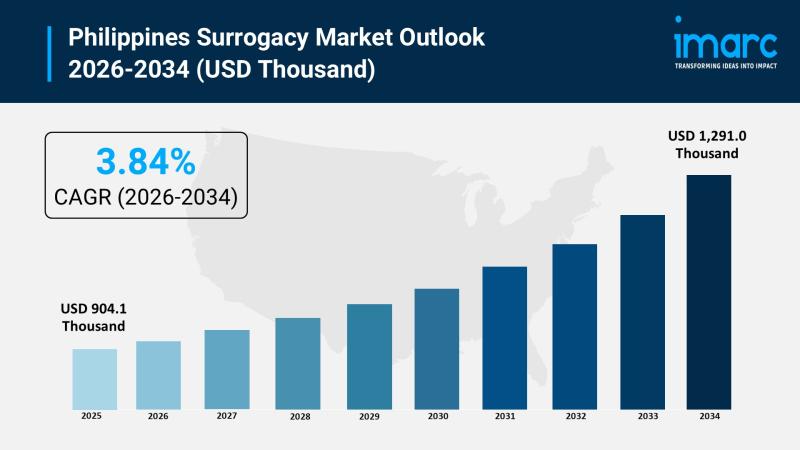
Philippines Surrogacy Market 2026 | Surge to Reach USD 1,291.0 Thousand by 2034
Market Overview
The Philippines surrogacy market size reached USD 904.1 Thousand in 2025 and is projected to reach USD 1,291.0 Thousand by 2034, exhibiting a growth rate (CAGR) of 3.84% during 2026-2034. The market encompasses diverse surrogacy arrangements, assisted reproductive technologies, and service provider types tailored to the needs of domestic and international intended parents, covering gestational and traditional surrogacy through IVF with ICSI, IVF without ICSI, and intrauterine insemination techniques…

Agarwood Oil Processing Plant DPR 2026: Setup, Investment Cost, Market Growth & …
Setting up an agarwood oil processing plant positions investors in one of the most exclusive and high-value segments of the global luxury fragrance and natural essential oils value chain, backed by sustained global growth driven by rising demand for premium natural fragrances, increasing use in luxury perfumery, traditional medicines, aromatherapy, and religious practices, and growing consumer preference for rare and natural essential oils. As affluent consumers worldwide expand their spending…

Dry Coconut Leaves Based Biodegradable Straw Manufacturing Plant DPR & Unit Setu …
Setting up a dry coconut leaves based biodegradable straw manufacturing plant positions investors in one of the most innovative and rapidly expanding segments of the sustainable packaging and eco-friendly disposable products value chain, backed by sustained global growth driven by the worldwide movement against single-use plastic straws, rising consumer and regulatory demand for truly compostable alternatives, abundant availability of agricultural waste raw materials, and the growing adoption of natural plant-based…

Camelina Oil Processing Plant DPR & Unit Setup - 2026: Machinery Cost, CapEx/OpE …
Setting up a camelina oil processing plant positions investors in one of the most promising and rapidly expanding segments of the specialty oilseed and sustainable crop oil value chain, backed by sustained global growth driven by the rising demand for omega-3 rich plant-based oils, growing adoption of camelina oil in nutraceutical and functional food applications, expanding use as a low-carbon feedstock for sustainable aviation fuel (SAF) and biofuel production, and…
More Releases for Medical
ECG Analysis System Market Research Report 2022 - GE Medical, Medical Econet, Gr …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global ECG Analysis System. On the basis of historic growth analysis and current scenario of ECG Analysis System place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
ECG Analysis System Market Outlook: 2020 The Year On A Positive Note | GE Medica …
DataIntelo report titled Global ECG Analysis System Market provides detailed information and overview about the key influential factors required to make well informed business decision. This is a latest report, covering the current COVID-19 impact on the market. The pandemic of Coronavirus (COVID-19) has affected every aspect of life globally. This has brought along several changes in market conditions. The rapidly changing market scenario and initial and future assessment of…
Medical Tracheostomy Tube Market Overall Study Report 2020-2027 | Players Medtro …
The latest report added by Stratagem Market Insights gives deep insights into the drivers and restraints in the Worldwide Medical Tracheostomy Tube Market. The research report "Global Medical Tracheostomy Tube Market Size and Growth Forecast to 2027" provide a comprehensive take on the overall market. Analysts have carefully evaluated the milestones achieved by the global Medical Tracheostomy Tube market and the current trends that are likely to shape its future.…
Global Embolization Particle Market 2019 - Sirtex Medical, Merit Medical, Cook M …
The global "Embolization Particle Market" report delivers a comprehensive and systematic framework of the Embolization Particle market at a global level that includes all the key aspects related to it. The data is collected from different sources allied to the global Embolization Particle market and the research team meticulously analyze the gathered data with the help of various analytical tools and present their opinion based on analysis and calculations. The…
Medical Casting & Splinting Market 2019: Top Key players are 3M, DJO Global, BSN …
Medical Casting & Splinting Market 2019 Report analyses the industry status, size, share, trends, growth opportunity, competition landscape and forecast to 2025. This report also provides data on patterns, improvements, target business sectors, limits and advancements. Furthermore, this research report categorizes the market by companies, region, type and end-use industry.
Get Sample Copy of this Report@ https://www.researchreportsworld.com/enquiry/request-sample/13718924
Global Medical Casting & Splinting market 2019 research provides a basic overview of…
Global Oxygen Pressure Regulator Market 2017 : Precision Medical, Smiths Medical …
Oxygen Pressure Regulator
A market study based on the " Oxygen Pressure Regulator Market " across the globe, recently added to the repository of Market Research, is titled ‘Global Oxygen Pressure Regulator Market 2017’. The research report analyses the historical as well as present performance of the worldwide Oxygen Pressure Regulator industry, and makes predictions on the future status of Oxygen Pressure Regulator market on the basis of this analysis.
Get Free…
