Press release
Lecithin Manufacturing Plant DPR & Unit Setup Report 2026
Setting up a lecithin manufacturing plant positions investors within one of the steadily expanding and multifunctional segments of the global food ingredients and specialty chemicals industry, supported by increasing demand for natural emulsifiers, rising consumption of processed foods, and expanding applications across pharmaceuticals, cosmetics, and nutraceuticals worldwide. Lecithin is widely valued for its emulsifying, stabilizing, and dispersing properties, making it a key ingredient in chocolate, bakery products, dietary supplements, and personal care formulations. As consumer preference shifts toward clean-label and plant-based ingredients, demand for naturally derived additives continues to grow. With advancements in extraction technologies, expanding global trade, and the rapid growth of retail and e-commerce distribution channels, the lecithin sector offers compelling opportunities for manufacturers and entrepreneurs seeking scalable production and sustained profitability in a high-value ingredients market.Market Overview and Growth Potential
The global lecithin market demonstrates steady growth trajectory, valued at USD 1.00 Billion in 2025. According to IMARC Group's comprehensive market analysis, the market is projected to reach USD 1.69 Billion by 2034, exhibiting a CAGR of 6.0% from 2026-2034. This sustained expansion is driven by the growing demand for natural and clean-label emulsifiers in processed food and bakery applications replacing synthetic mono- and di-glycerides and other chemically derived emulsifiers, increasing consumer and formulator preference for non-GMO sunflower and rapeseed lecithin as an allergen-free alternative to soy lecithin, expanding nutraceutical and dietary supplement applications targeting cognitive health, liver function, and phosphatidylserine brain nutrition, growing pharmaceutical industry utilization of high-purity lecithin fractions in liposomal drug delivery and encapsulation systems, and rising animal feed application in aquaculture and livestock nutrition where lecithin enhances fat digestibility and growth performance.
Request for a Sample Report: https://www.imarcgroup.com/lecithin-manufacturing-plant-project-report/requestsample
Lecithin is a naturally occurring mixture of phospholipids-primarily phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylinositol (PI), and phosphatidic acid (PA)-along with neutral lipids and glycolipids, recovered as a valuable by-product from crude vegetable oil degumming during oilseed processing of soybeans, sunflowers, rapeseed, and other oil crops. Commercial lecithin is produced in several product grades and forms: standard fluid soybean lecithin as a brown viscous liquid at 95-99% phospholipid and neutral oil content; deoiled lecithin powder produced by solvent extraction removing neutral oil to yield a free-flowing dry powder with 95-98% phospholipid purity and enhanced dispersibility; modified lecithin including hydroxylated, acetylated, and enzymatically modified variants with improved emulsification performance in specific food applications; fractionated high-purity phosphatidylcholine concentrates for pharmaceutical and nutraceutical applications; and certified non-GMO sunflower lecithin produced from hexane-free cold-pressed sunflower oil processing meeting clean-label, organic, and allergen-free positioning requirements commanding significant pricing premiums above standard soy lecithin.
The lecithin market is witnessing robust demand due to the structural transition of global food manufacturing toward natural, minimally processed, and clean-label ingredient declarations, the explosive growth of the plant-based food and alternative protein sector requiring lecithin as a critical emulsifier and texture enhancer in plant-based meat, dairy alternative, and functional food formulations, and the expanding pharmaceutical liposomal delivery system pipeline driving high-purity PC fraction demand for drug encapsulation and bioavailability enhancement applications. According to the Economic Survey 2024-25, Government-led oilseed value chain development and food processing investment incentive programs further strengthen market prospects.
Plant Capacity and Production Scale
The proposed lecithin manufacturing facility is designed with an annual production capacity ranging between 5,000 - 15,000 tons, enabling economies of scale while maintaining operational flexibility. This capacity range allows manufacturers to cater to diverse market segments-from standard fluid soy lecithin for bakery, chocolate, and food ingredient distribution to deoiled lecithin powder for instant food, infant formula, and nutraceutical applications, non-GMO and organic-certified sunflower lecithin for clean-label food brands, phosphatidylcholine-enriched fractions for dietary supplement and cognitive health product manufacturers, pharmaceutical-grade high-purity PC and PE concentrates for liposomal drug delivery systems, and specialty modified lecithin for confectionery, margarine, and functional food formulation applications-ensuring steady demand and consistent revenue streams across food, nutraceutical, pharmaceutical, and animal nutrition industry verticals.
Ask Analyst for Customization:
https://www.imarcgroup.com/request?type=report&id=11038&flag=C
Financial Viability and Profitability Analysis
The lecithin manufacturing business demonstrates healthy profitability potential under normal operating conditions. The financial projections reveal:
• Gross Profit Margins: 30-40%
• Net Profit Margins: 15-22%
These margins are supported by stable and growing demand across food processing, nutraceutical, pharmaceutical, and animal nutrition sectors, value-added deoiled, non-GMO certified, fractionated, and pharmaceutical-grade lecithin product positioning enabling significant per-kilogram pricing premiums of 2-8 times standard fluid soy lecithin, and the integrated oilseed processing economics enabling gum recovery at very low incremental raw material cost compared to standalone lecithin production from purchased crude gum. The project demonstrates strong return on investment (ROI) potential, particularly for facilities integrated with existing soybean or sunflower oil processing operations where the crude gum by-product is currently underutilized or sold at commodity prices.
Cost of Setting Up a Lecithin Manufacturing Plant
Operating Cost Structure
Understanding the operating expenditure (OpEx) is crucial for effective financial planning and cost management. The cost structure for a lecithin manufacturing plant is primarily driven by:
• Raw Materials: 70-80% of total OpEx
• Utilities: 10-15% of OpEx
Raw materials constitute the dominant portion of operating costs, with crude soybean, sunflower, or rapeseed gum-the aqueous phospholipid fraction separated during vegetable oil degumming-being the primary feedstock input, the price of which directly tracks oilseed processing commodity economics and is influenced by seasonal soybean and sunflower crop yields in major producing countries. For standalone lecithin processors purchasing crude gum, the gum procurement price represents 55-65% of production cost; for integrated oilseed crushers, the gum is produced at processing cost, dramatically improving the lecithin recovery economics. Hexane or acetone solvent for deoiling operations, enzyme preparations for enzymatic modification, and primary packaging including food-grade drums and powder bags represent secondary material inputs. Establishing integrated crude gum supply relationships with oilseed crushing operations, and developing fractionation and purification capability for high-value PC and PE concentrates, are the most critical strategies for sustained lecithin production competitiveness and margin enhancement.
Capital Investment Requirements
Setting up a lecithin manufacturing plant requires substantial capital investment across several critical categories:
Land and Site Development: Selection of an optimal location with strategic proximity to major oilseed crushing operations-preferably integrated within or adjacent to an existing soybean, sunflower, or canola oil refinery to access crude gum by-product feedstock at competitive cost-and logistics connectivity to food ingredient distributors, nutraceutical manufacturers, pharmaceutical ingredient suppliers, and export terminals. The site must have reliable utilities including steam, cooling water, and electrical supply for evaporation and deoiling operations, solvent recovery and storage infrastructure for hexane-based deoiling lines, and food-grade manufacturing environment meeting HACCP and GMP standards.
Machinery and Equipment: The largest portion of capital expenditure (CapEx) covers specialized gum degumming, evaporation, deoiling, fractionation, and modification equipment essential for multi-grade lecithin production.
Key machinery includes:
• Crude gum receiving, storage, and conditioning tanks with heating systems to maintain flowability of viscous phospholipid slurry
• Multi-effect falling-film evaporators for concentrating crude hydrated gum by evaporating excess water to target moisture content
• Vacuum drum dryers or thin-film evaporators for final moisture reduction and producing fluid standard lecithin
• Hexane or acetone extraction and deoiling systems for producing deoiled lecithin powder from fluid lecithin
• Solvent recovery distillation columns for recovering and recycling hexane or acetone from deoiling operations
• Spray dryers and powder handling systems for producing free-flowing deoiled lecithin powder at target moisture and particle size
• Alcohol fractionation or chromatographic purification equipment for producing phosphatidylcholine-enriched lecithin fractions
• Quality control laboratory with phospholipid composition HPLC, acetone-insoluble content, moisture, and acid value testing capability
Civil Works: Food-grade manufacturing facility construction meeting HACCP and cGMP standards for food ingredient and nutraceutical production. The layout should incorporate crude gum receiving and heated storage, gum conditioning and water addition section, evaporation and concentration unit, fluid lecithin storage and drum filling area, deoiling and solvent extraction section with explosion-proof classification, solvent recovery plant, spray drying hall, powder processing and sifting area, fractionation section for high-purity PC production, quality control laboratory, cleanroom packaging area for pharmaceutical grades, cold storage for specialty grades, finished goods warehouse, utility block, and administrative offices.
Buy now: https://www.imarcgroup.com/checkout?id=11038&method=2175
Other Capital Costs: Pre-operative expenses, equipment installation and commissioning, FSSAI food ingredient manufacturing license, HACCP and ISO 22000 food safety management system certification, US FDA food ingredient notification for export market supply, non-GMO Project Verification for premium non-GMO soy lecithin, USDA Organic certification for organic sunflower lecithin, EU Novel Food or food additive notification for modified lecithin grades, solvent handling safety certification, initial crude gum procurement working capital, and contingency provisions for product quality qualification and customer approval trials during plant establishment.
Major Applications and Market Segments
Lecithin finds extensive applications across diverse food, nutraceutical, pharmaceutical, and industrial market segments, demonstrating its unique combination of functional, nutritional, and delivery technology properties:
Food Processing and Bakery: Primary and largest application segment where standard fluid and deoiled soy or sunflower lecithin serves as a natural emulsifier in chocolate and compound coatings improving flow properties and reducing cocoa butter content, bakery products improving dough machinability and extending shelf life, margarine and spreads for fat-water emulsion stability, instant food powders for wettability and dispersibility, pasta and noodle manufacturing for texture, and plant-based meat alternatives for fat emulsification and texture cohesion.
Infant Formula and Clinical Nutrition: Premium application where specifically selected and tested lecithin grades-predominantly non-GMO or organic soy or sunflower lecithin with stringent purity specifications-are incorporated in infant formula for fat emulsification and structural mimicry of human breast milk phospholipid composition, in clinical enteral nutrition products for fat digestion support, and in sports nutrition and meal replacement products for emulsification, mouthfeel, and phosphatidylcholine content contributing to cognitive function claims.
Nutraceuticals and Dietary Supplements: Rapidly growing premium segment where phosphatidylcholine-enriched lecithin concentrates (20-95% PC) are formulated into cognitive health supplements targeting brain function, memory, and focus; liver health supplements leveraging phosphatidylcholine's role in hepatic phospholipid membrane integrity and fat metabolism; sports performance supplements for choline precursor delivery; and cardiovascular health formulations, with the booming cognitive health and brain nutrition supplement category creating growing premium demand for high-purity PC fractions from sunflower and non-GMO soy sources.
Pharmaceutical and Drug Delivery: Specialty high-value application where pharmaceutical-grade lecithin fractions of defined phospholipid composition and microbiological purity are used as excipients in liposomal drug formulations encapsulating hydrophilic and lipophilic active pharmaceutical ingredients for enhanced bioavailability and targeted delivery, in topical emulsion cream and gel formulations as penetration enhancers, in parenteral lipid emulsions for intravenous nutrition, and as a processing aid in soft gelatin capsule formulations, with the expanding liposomal cancer therapy, vaccine adjuvant, and mRNA delivery system pipeline creating growing institutional demand for cGMP pharmaceutical-grade lecithin.
Animal Feed and Aquaculture Nutrition: High-volume application where soybean and rapeseed lecithin supplies the primary phospholipid fraction in aquaculture feed for shrimp, salmon, sea bass, and tilapia where phospholipids are essential during larval development stages and cannot be synthesized de novo, and in livestock and poultry feed formulations as a fat and energy source with additional phospholipid nutritional value, with the rapidly expanding global aquaculture industry creating growing institutional demand for consistent-quality marine and terrestrial animal nutrition-grade lecithin.
Why Invest in Lecithin Manufacturing?
Clean-Label Emulsifier Transition: The global food industry's structural transition from synthetic emulsifiers-sodium stearoyl lactylate, DATEM, monoglycerides, and polysorbates-toward naturally derived lecithin as a clean-label, consumer-friendly emulsifier is creating a systematic and regulatory-independent demand growth driver for lecithin that benefits producers regardless of overall food market growth rates, as reformulation programs by major food brands replacing synthetic E-number emulsifiers with lecithin create a growing and irreversible share shift within the emulsifier category.
Non-GMO Sunflower Lecithin Premium: The rapid growth of non-GMO and organic food markets in North America, Europe, and premium Asian urban consumer segments has created an extraordinary commercial opportunity for sunflower lecithin produced without solvent extraction and without GMO crop inputs, commanding retail ingredient pricing of 3-5 times standard commodity soy lecithin while serving the fastest-growing consumer segment-clean-label, allergen-free, and sustainability-committed food brands-with India's growing sunflower cultivation base in Karnataka, Andhra Pradesh, and Maharashtra providing a competitive domestic raw material advantage.
Cognitive Health Supplement Boom: The global explosion of consumer interest in brain health, cognitive performance, memory support, and nootropic supplements-driven by aging demographics, workplace performance optimization culture, and evidence-based preventive health investment-is creating structurally growing premium demand for phosphatidylcholine-enriched lecithin fractions as the primary active ingredient in cognitive health supplements, providing lecithin manufacturers with an extraordinarily high-margin product channel far above standard food emulsifier economics.
Pharmaceutical Liposomal Delivery Pipeline: The pharmaceutical industry's expanding adoption of liposomal delivery technology-validated by commercial successes including liposomal doxorubicin, liposomal amphotericin B, and mRNA lipid nanoparticle COVID-19 vaccines-is generating growing institutional demand for cGMP pharmaceutical-grade PC fractions from qualified suppliers, representing one of the highest-margin specialty lecithin application segments with demanding but clearly articulable quality specifications enabling premium pricing and contractual supply security.
Government Support: Government-led National Mission on Oilseeds supporting oilseed production and crushing capacity expansion, food processing PLI scheme incentives for value-added ingredient manufacturing, FSSAI modernization enabling premium ingredient grade marketing, Export Promotion Council support for specialty food ingredient exports, AYUSH nutraceutical manufacturing promotion, and oilseed processing cluster development programs further strengthen market prospects and directly support lecithin manufacturing investment.
Import Substitution Opportunities: India currently imports pharmaceutical-grade lecithin fractions, non-GMO certified lecithin, and high-purity PC concentrates predominantly from Germany (Lipoid GmbH), the United States (ADM), and the Netherlands, with growing domestic pharmaceutical formulation, nutraceutical brand, and clinical nutrition manufacturing creating significant import substitution opportunities for Indian lecithin processors establishing cGMP-certified fractionation and purification capability to supply domestic API and finished dose manufacturers at competitive pricing and shorter supply chain lead times.
Manufacturing Process Excellence
The lecithin manufacturing process involves several precision-controlled stages:
• Crude Gum Recovery: During oilseed crushing and crude oil degumming, water or acid is added to crude vegetable oil to hydrate and precipitate the phospholipid gum fraction, which is separated by centrifuge as crude hydrated gum containing 20-40% moisture, 30-50% phospholipids, and residual neutral oil and impurities
• Evaporation and Concentration: Crude hydrated gum is processed through multi-effect falling-film evaporators operating under gentle vacuum to progressively evaporate water and concentrate the phospholipid content, with temperature controlled carefully below phospholipid degradation thresholds to preserve functional quality and color
• Final Drying: Concentrated gum is further dried in vacuum drum dryers or thin-film evaporators to achieve the target moisture content and viscosity specification for fluid standard lecithin product, which is then cooled and stored in heated tanks to maintain pumpability
• Deoiling: Fluid lecithin is extracted with hexane or acetone in a counter-current extraction system to dissolve and remove the neutral oil fraction, leaving defatted phospholipid paste that is then dried to produce free-flowing deoiled lecithin powder with 95-98% phospholipid content
• Fractionation: For pharmaceutical and premium nutraceutical grades, deoiled lecithin is fractionated by alcohol solvent extraction or column chromatography to separate phosphatidylcholine-enriched fractions at 20-95% PC purity from PE and PI fractions, with purity confirmed by HPLC phospholipid class analysis
• Enzymatic Modification: Specialty modified lecithin grades are produced by incubation with phospholipase A2 or phospholipase D enzymes under controlled pH and temperature conditions generating lyso-phospholipids with enhanced emulsification performance for specific food emulsification applications
• Quality Testing and Packaging: Each production lot is tested for acetone-insoluble phospholipid content, moisture, acid value, peroxide value, phospholipid class composition by HPLC, heavy metals, pesticide residues, and microbiological compliance before grade certification and filling into food-grade steel drums, 25 kg kraft bags, or pharmaceutical-grade stainless steel containers with full certificate of analysis and application-specific compliance documentation
Industry Leadership
The global lecithin manufacturing industry is led by established oilseed processing companies and specialty phospholipid manufacturers with extensive extraction, fractionation, and purification capabilities and strong food ingredient, nutraceutical, and pharmaceutical customer relationships. Key industry players include:
• Cargill, Inc.
• ADM
• Lipoid GmbH
• Bunge Limited
• American Lecithin Company
• Global River Food Ingredients
These companies serve diverse end-use sectors including food processing and bakery, infant formula and clinical nutrition, nutraceuticals and dietary supplements, pharmaceutical and drug delivery, and animal feed and aquaculture nutrition, demonstrating the broad commercial applicability of lecithin across the full spectrum of food, health, and industrial applications globally.
Browse Full Report: https://www.imarcgroup.com/lecithin-manufacturing-plant-project-report
About Us:
IMARC Group is a global management consulting firm that helps the world's most ambitious changemakers to create a lasting impact. The company excel in understanding its client's business priorities and delivering tailored solutions that drive meaningful outcomes. We provide a comprehensive suite of market entry and expansion services. Our offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape, and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No:(D) +91 120 433 0800
United States: (+1-201971-6302)
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Lecithin Manufacturing Plant DPR & Unit Setup Report 2026 here
News-ID: 4481747 • Views: …
More Releases from IMARC Group

Brazil Cloud Security Market to Reach USD 2,003.29 Million by 2034, Growing at a …
Brazil Cloud Security Market Summary:
• The Brazil Cloud Security Market size reached USD 609.28 Million in 2025.
• The market is projected to reach USD 2,003.29 Million by 2034.
• The market is expected to grow at a CAGR of 12.64% during 2026-2034.
• Large enterprises dominate the market due to rising investments in cloud infrastructure and cybersecurity frameworks.
• Growth is driven by increasing cloud adoption, rising cyber threats, expansion of remote working…

Vietnam Coffee Market Growth, Demand and Opportunity Report 2026-2034
Vietnam Coffee Market Summary:
• The Vietnam Coffee Market size reached 73.44 Thousand Tons in 2025.
• Market is projected to reach 102.23 Thousand Tons by 2034.
• Southern Vietnam dominates with a significant market share.
• Growth driven by rising domestic coffee consumption, increasing café culture expansion, and growing premium coffee exports.
IMARC Group, a leading market research company, has released its latest report titled "Vietnam Coffee Market." The study provides a detailed analysis…
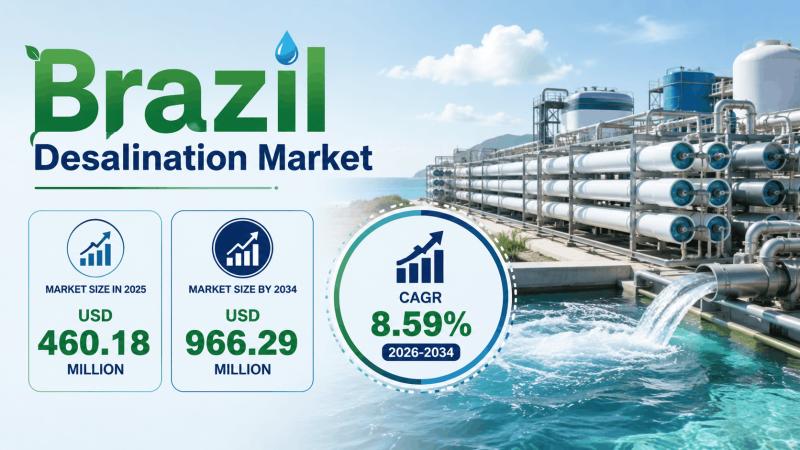
Brazil Desalination Market to Reach USD 966.29 Million by 2034, Growing at a CAG …
Brazil Desalination Market Summary:
• The Brazil Desalination Market size reached USD 460.18 Million in 2025.
• The market is projected to reach USD 966.29 Million by 2034.
• The market is expected to grow at a CAGR of 8.59% during 2026-2034.
• Rising water scarcity and increasing government investments in water infrastructure are driving market expansion.
• Growing adoption of renewable energy-powered desalination systems is supporting long-term industry growth.
Request Sample For PDF Report: https://www.imarcgroup.com/brazil-desalination-market/requestsample
The…

Hydrofluoric Acid Production Plant Setup DPR 2026: Machinery, Investment & Opera …
Setting up a hydrofluoric acid production plant positions investors in one of the most stable and essential segments of the specialty chemicals value chain, backed by sustained global growth driven by its extensive use in fluorochemicals production, petroleum refining, aluminum processing, and semiconductor manufacturing. Increasing demand for refrigerants, lithium-ion batteries, and specialty chemicals continues to support market growth. As electrification accelerates, semiconductor manufacturing intensifies, and fluorochemical applications diversify, the global…
More Releases for GMO
Transformative Trends Impacting the GMO Testing Market Landscape: Advancements I …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the GMO Testing Market Size By 2025?
The market size for gmo testing has seen significant expansion in the past few years. It's estimated to increase from $2.52 billion in 2024 to $2.75 billion in 2025, indicating a compound annual growth rate (CAGR) of 9.2%. This…
Non-GMO Flour Market Forecast Projects Steady Growth Amid Rising Awareness of GM …
InsightAce Analytic Pvt. Ltd. Announces the release of a market assessment report on the "Non-GMO Flour Market"-, By Product Type (Wheat Flour, Corn Flour, Rice Flour, Almond Flour), By Distribution Channel (Online Retail, Supermarkets, Health Food Stores, Wholesale), By End Use (Baking, Cooking, Brewing, Snacking), By Form (Whole Grain, Refined, Coarse), and Global Forecasts, 2024-2031 And Segment Revenue and Forecast To 2031."
The Non-GMO Flour Market is estimated to reach…
GMO Testing: Core Growth Enabler in the Rising Genetically Modified Crop Product …
Stay ahead with our updated market reports featuring the latest on tariffs, trade flows, and supply chain transformations.
What Will the GMO Testing Industry Market Size Be by 2025?
In recent times, the market size of GMO testing has witnessed a rapid expansion. It is set to soar from $2.52 billion in 2024 to a whopping $2.77 billion in 2025 at 10.0% compound annual growth rate (CAGR). This surging growth in the…
GMO Testing: Core Growth Enabler in the Rising Genetically Modified Crop Product …
Stay ahead with our updated market reports featuring the latest on tariffs, trade flows, and supply chain transformations.
What Will the GMO Testing Industry Market Size Be by 2025?
In recent times, the market size of GMO testing has witnessed a rapid expansion. It is set to soar from $2.52 billion in 2024 to a whopping $2.77 billion in 2025 at 10.0% compound annual growth rate (CAGR). This surging growth in the…
Rising Genetically Modified Crop Production Fuels Gmo Testing Market Growth: An …
The GMO Testing Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Projected Growth of the GMO Testing Market?
In the past few years, the size of the GMO testing market has expanded significantly. It is anticipated to rise from $2.52 billion in 2024…
Non-GMO Soybean Market Size and Forecast
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- The global Non-GMO Soybean Market is expected to record a CAGR of XX.X% from 2024 to 2031 In 2024, the market size is projected to reach a valuation of USD XX.X Billion. By 2031 the valuation is anticipated to reach USD XX.X Billion.
The impact of manufacturers on the market is significant across various industries, influencing supply chains, consumer choices, and economic growth. Manufacturers are key players in…
