Press release
Top Companies in Healthcare Regulatory Affairs Outsourcing Market - KLIFO , Asphalion S.L. etc., IQVIA Inc, Pharmalex Gmbh
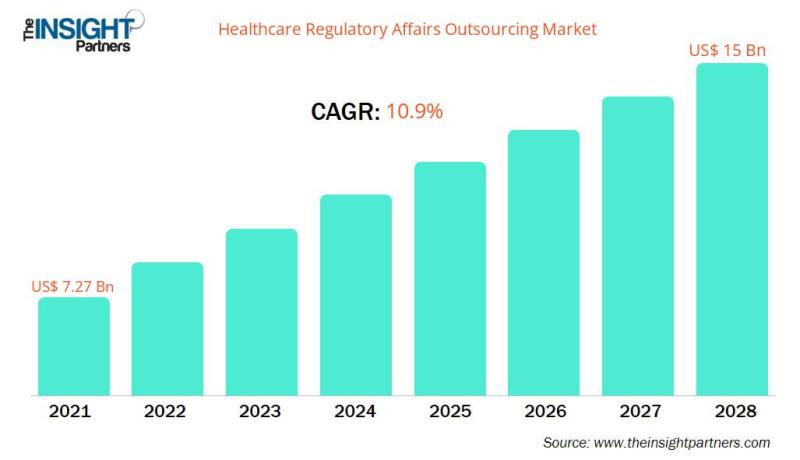
The healthcare regulatory affairs outsourcing market is projected to reach US$ 14,996.35 million by 2028 from US$ 7,274.73 million in 2021; it is expected to grow at a CAGR of 10.9 % from 2021 to 2028.The increasing regulatory pressure on healthcare companies and escalating demand for speedy approval of new products. However, dearth of skilled professionals is restraining the healthcare regulatory affairs outsourcing market growth.
Get a Sample PDF - https://www.theinsightpartners.com/sample/TIPRE00007611?utm_source=openpr&utm_medium=10324
Regulatory affairs outsourcing is the services offered to the pharmaceutical, biotech, and medical devices manufacturing industries. Regulatory affair outsourcing services help to achieve fast regulatory approvals. Regulatory affairs outsourcing industries are helping to get approval for new products, preparing protocols for conducting a clinical trial, publishing reports etc. An increase in demand for various services like regulatory consultation, medical writing and publishing of the regulatory documentation, clinical trial applications, and regulatory consulting and legal representations, patent application, product registration, and clinical trial applications has resulted in a surge in the adoption of healthcare regulatory affairs outsourcing business.
The Healthcare Regulatory Affairs Outsourcing Market continues to gain significant traction as pharmaceutical, biotechnology, and medical device companies adapt to an increasingly complex global regulatory environment. With evolving compliance standards, rising documentation requirements, and growing pressure to accelerate product approvals, organizations are increasingly relying on specialized outsourcing partners to manage regulatory workflows efficiently.
As innovation in healthcare advances at a rapid pace, the Healthcare Regulatory Affairs Outsourcing Market is becoming a strategic pillar for organizations aiming to bring new therapies and technologies to patients faster. Outsourcing is no longer limited to administrative support-it now plays a central role in regulatory intelligence, submission strategy, lifecycle management, and post-market compliance.
A Human-Centric Approach to Regulatory Transformation
Behind the growth of the Healthcare Regulatory Affairs Outsourcing Market is a simple goal helping patients access safe and effective healthcare solutions sooner. Regulatory teams worldwide face mounting workloads as product pipelines expand and compliance standards evolve. Outsourcing partners provide the flexibility and specialized expertise needed to manage these demands while allowing internal teams to focus on research, development, and innovation.
This shift toward collaboration reflects a broader transformation in the healthcare ecosystem, where speed, accuracy, and transparency are essential to delivering better patient outcomes.
Access Full Report - https://www.theinsightpartners.com/reports/healthcare-regulatory-affairs-outsourcing-market
Market Size, Share, Trends, Analysis, and Forecast by 2031
Expanding reliance on outsourcing for global regulatory submissions
Growing market share of providers offering digital and AI-driven regulatory solutions
Rising demand for lifecycle management and post-market surveillance services
Increasing partnerships between healthcare companies and outsourcing specialists
Strong long-term growth outlook supported by global healthcare innovation
Key Market Drivers
Rising Complexity of Global Regulations
Healthcare regulations vary significantly across regions, creating challenges for companies launching products internationally. The Healthcare Regulatory Affairs Outsourcing Market is expanding as organizations seek partners with expertise in regional compliance requirements.
Increasing Healthcare Innovation
The growth of biologics, personalized medicine, digital therapeutics, and AI-enabled healthcare solutions is increasing regulatory workload. Outsourcing provides access to specialized skills required to navigate emerging regulatory pathways.
Need for Faster Time-to-Market
In a competitive healthcare landscape, speed is critical. Outsourcing helps streamline regulatory processes, reduce delays, and accelerate product approvals.
Cost Efficiency and Resource Optimization
Building large in-house regulatory teams can be costly and resource-intensive. Outsourcing enables companies to scale operations efficiently while maintaining high compliance standards.
Global and Regional Analysis
North America
North America continues to lead the Healthcare Regulatory Affairs Outsourcing Market due to strong research activity, advanced healthcare infrastructure, and stringent regulatory frameworks. Companies are increasingly partnering with outsourcing providers to manage growing submission volumes and compliance requirements.
Europe
Europe is witnessing steady adoption of outsourcing services as regulatory frameworks evolve and digital health solutions gain momentum. Harmonization efforts and patient safety initiatives are driving demand for expert regulatory support.
Asia-Pacific
Asia-Pacific is emerging as a high-growth region in the Healthcare Regulatory Affairs Outsourcing Market. Expanding pharmaceutical manufacturing, increasing clinical trials, and rising healthcare investments are fueling demand for regulatory expertise.
Middle East, Africa, and Latin America
These regions are gradually strengthening regulatory frameworks, creating new opportunities for outsourcing providers to support market entry and compliance management.
Emerging Market Trends
The Healthcare Regulatory Affairs Outsourcing Market is being shaped by several transformative trends:
Integration of artificial intelligence and automation in regulatory workflows
Expansion of cloud-based regulatory information management systems
Increased focus on real-world evidence and post-market monitoring
Growing demand for global regulatory intelligence services
Rising emphasis on digital submission and documentation processes
Recent Market Developments
Healthcare organizations worldwide are investing in regulatory modernization and digital transformation initiatives. Companies are expanding outsourcing partnerships to strengthen compliance capabilities and improve efficiency. The growing adoption of advanced analytics and automation tools is reshaping regulatory processes and creating new opportunities for outsourcing providers.
These developments highlight the increasing importance of the Healthcare Regulatory Affairs Outsourcing Market in supporting innovation, compliance, and global healthcare accessibility.
Competitive Landscape and Future Outlook
The Healthcare Regulatory Affairs Outsourcing Market is becoming increasingly competitive as service providers expand capabilities and build global expertise. Strategic collaborations, service diversification, and technology integration are shaping the next phase of market evolution.
Looking ahead, the Healthcare Regulatory Affairs Outsourcing Market is expected to remain a key enabler of healthcare innovation. As regulatory requirements continue to evolve, outsourcing partnerships will play a critical role in helping organizations navigate complexity, accelerate approvals, and bring life-changing healthcare solutions to patients worldwide.
About the Industry
The Healthcare Regulatory Affairs Outsourcing Market empowers healthcare organizations with specialized expertise to manage regulatory compliance, streamline submissions, and support global product launches. As healthcare innovation continues to grow, outsourcing is becoming an essential strategy for success in the global healthcare landscape.
Get Premium Research Report Market Size and Growth Report at:
https://www.theinsightpartners.com/buy/TIPRE00007611?utm_source=openpr&utm_medium=10324
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
About Us:
The Insight Partners is a one stop industry research provider of actionable intelligence. We help our clients in getting solutions to their research requirements through our syndicated and consulting research services. We specialize in industries such as Semiconductor and Electronics, Aerospace and Defense, Automotive and Transportation, Biotechnology, Healthcare IT, Manufacturing and Construction, Medical Device, Technology, Media and Telecommunications, Chemicals and Materials.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Top Companies in Healthcare Regulatory Affairs Outsourcing Market - KLIFO , Asphalion S.L. etc., IQVIA Inc, Pharmalex Gmbh here
News-ID: 4481558 • Views: …
More Releases from The Insight Partners

Video Surveillance Storage Market Analysis 2025-2031: Growth, Trends, and Foreca …
The global video surveillance storage market is witnessing rapid expansion, driven by the exponential growth of video data and the increasing need for secure, scalable, and efficient storage systems. According to The Insight Partners, the market is projected to reach approximately US$ 20.21 billion by 2031, growing at a compound annual growth rate (CAGR) of around 15.0% during 2025-2031.
Get Sample Report@ https://www.theinsightpartners.com/sample/TIPRE00002821?utm_source=OpenPR&utm_medium=10852
For instant assistance or inquiries, please contact us at:…

User and Entity Behavior Analytics Market Growth, Trends & Forecast 2031
The User and Entity Behavior Analytics (UEBA) market has emerged as a critical segment within the broader cybersecurity and data analytics landscape. UEBA solutions focus on monitoring and analyzing user and entity activities across networks, devices, and applications to identify abnormal behavior and potential security threats. With the growing sophistication of cyberattacks and increasing digital transformation across industries, the demand for advanced behavioral analytics solutions has accelerated significantly.
According to The…

Driving Training Simulator Market Gains Momentum with Rising Focus on Road Safet …
According to a recent market study by The Insight Partners, the global driving training simulator market is experiencing steady growth, driven by increasing emphasis on road safety, rising demand for professional drivers, and rapid advancements in autonomous vehicle technologies. The market was valued at US$ 1.95 billion in 2021 and is projected to grow at a CAGR of 4.6% from 2022 to 2028.
Get Sample Report- https://www.theinsightpartners.com/sample/TIPRE00004985?utm_source=OpenPR&utm_medium=10927
For instant assistance or inquiries,…
Cryogenic Control Valve Market Poised for Steady Growth, Driven by LNG Expansion …
According to a latest market study by The Insight Partners, the global Cryogenic Control Valve Market is set for consistent growth over the next decade, supported by rising investments in liquefied natural gas (LNG) infrastructure, expansion of industrial gas applications, and increasing demand for high-precision flow control systems. As per the report, the Cryogenic Control Valve Market Size is expected to reach US$4.10 million by 2034, growing from US$2.72 million…
More Releases for Regulatory
Medical Device Regulatory Affairs Market Medical Device Regulatory Affairs Marke …
"Medical Device Regulatory Affairs Market" in terms of revenue was estimated to be worth $ 6.7 billion in 2024 and is poised to reach $ 18.3 billion by 2034, growing at a CAGR of 10.8% from 2025 to 2034 according to a new report by InsightAce Analytic.
Request For Free Sample Pages:
https://www.insightaceanalytic.com/request-sample/1913
Latest Drivers Restraint and Opportunities Market Snapshot:
Key factors influencing the global medical device regulatory…
Medical Device & IVD Regulatory Affairs Outsourcing Market: Navigating Regulator …
Global healthcare landscape, the Medical Device & IVD Regulatory Affairs Outsourcing Market has emerged as a critical component ensuring the safe and compliant introduction of medical devices and in-vitro diagnostic products to the market. As the industry witnesses significant shifts and challenges, here's an in-depth analysis of the current trends, dynamics, and future prospects within this market segment.
Download sample PDF copy of report: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=79264&utm_source=OpenPR_Ajay&utm_medium=OpenPR
Impact of COVID-19 on European Regulations
The outbreak of…
Regulatory Writing Market - Clear, Concise, Compliant: Redefining Regulatory Wri …
Newark, New Castle, USA - new report, titled Regulatory Writing Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Regulatory Writing market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Regulatory Writing market. The report offers an overview of the market, which…
Complex Regulatory Frameworks
It is challenging for new entrants to enter the FinTech industry because of its complex regulatory framework. All FinTech companies must comply with compliance requirements even before they begin operations, which increases their costs and creates a significant barrier for startups. While regulations are needed to protect consumers, a number of existing laws are slowing down the growth of many Indian FinTech companies, thereby extending their time to reach the…
South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Cr …
Presented report, South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Creates Regulatory Uncertainty, presents the essential information relating to the terms which govern investment into South Africa’s upstream oil and gas sector. The report sets out in detail the contractual framework under which firms must operate in the industry, clearly defining factors affecting profitability and quantifying the state’s take from hydrocarbon production. Considering political, economic and industry…
Regulatory Affairs Outsourcing Market (Services - Regulatory Submissions, Clinic …
This research study analyzes the market for regulatory affairs outsourcing services in terms of revenue (US$ Mn). The stakeholders of this report comprises the clinical research organizations. The global regulatory affairs outsourcing market has been broadly segmented on the basis of services (Regulatory Submissions, Clinical Trial Applications and Product Registrations, Regulatory Writing and Publishing, Regulatory Consulting and Legal Representation and others regulatory affairs, and Geography (North America, Europe, Asia Pacific,…