Press release
Biologics Safety Testing Market Growth Accelerates with 9.7% CAGR Through 2033, Says Persistence Market Research
The biologics safety testing market plays a pivotal role in ensuring the quality, efficacy, and safety of biopharmaceutical products. As biologics-such as vaccines, monoclonal antibodies, and gene therapies-gain momentum, the need for rigorous safety testing becomes more critical. These complex therapeutics, often derived from recombinant DNA and advanced biotechnologies, require careful scrutiny to prevent contamination and ensure they meet regulatory standards.The biologics safety testing market is projected to reach USD 8.8 billion by 2033, growing at a compound annual growth rate (CAGR) of 9.7% from 2026 to 2033, up from USD 4.6 billion in 2026. This growth is driven by advancements in biopharmaceuticals, regulatory requirements, and the increasing adoption of high-throughput testing methods.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/18592
Market Overview and Key Trends
The biologics safety testing market has evolved in response to the growing use of biologics in treating a wide range of diseases, from cancer to autoimmune disorders and rare genetic conditions. Regulatory agencies such as the U.S. FDA, EMA, and other national authorities demand stringent safety protocols across all stages of development, manufacturing, and commercialization of biologics. These regulations include viral clearance, endotoxin, sterility, and residual DNA testing, which ensures product safety and efficacy.
The market's growth is largely attributed to the increasing approval and use of biologics, including monoclonal antibodies, gene therapies, and vaccines. As the biologics pipeline expands, so does the demand for high-quality testing services. Other key market drivers include the rise of biosimilars, the growth of vaccine production, and advances in cell and gene therapy research.
Regional Insights
North America remains the dominant region in the biologics safety testing market. The region's leadership is driven by its well-established biopharmaceutical infrastructure, advanced research and development (R&D) investments, and stringent regulatory frameworks. The U.S. is home to major pharmaceutical companies and testing organizations, while Canada also plays a significant role in biologics development. North America's adoption of advanced safety testing technologies, including automated testing systems and rapid microbiological methods, enhances the region's competitive edge in biologics safety testing.
Asia Pacific is the fastest-growing region in the biologics safety testing market. Countries such as China, India, and Japan have seen rapid advancements in biopharmaceutical manufacturing, driven by government initiatives and investments. These nations are increasingly adopting high-throughput automated testing systems to meet the rising demand for biologics and vaccines. Additionally, the growing presence of contract research organizations (CROs) in Asia Pacific has led to an expansion of outsourced testing services, further fueling market growth.
Market Dynamics
Drivers of Market Growth
Surge in Biologics Approvals: With the rapid expansion of biologics pipelines-particularly vaccines, monoclonal antibodies, and gene therapies-the need for comprehensive safety testing has skyrocketed. Regulatory agencies require extensive testing for viral clearance, sterility, endotoxins, and other contaminants before the approval of clinical trials or commercial release. As new therapies like gene and cell therapies are developed, testing methodologies must evolve to keep up with the increased complexity and production demands.
Stringent Regulatory Mandates: Global regulatory bodies impose strict requirements for identity, potency, and purity, with compliance necessary before products can be released for clinical use. Sterility and viral safety testing are crucial for ensuring patient safety, especially in injectable biologics. New testing approaches, such as recombinant Factor C endotoxin assays, are gaining traction for their ability to reduce reliance on animal-derived reagents, aligning with ethical and regulatory requirements.
Ethical and Sustainability Initiatives: Increasing focus on the ethical concerns surrounding animal-based assays has led to a shift toward animal-free testing solutions. For instance, the adoption of recombinant Factor C assays in endotoxin detection provides a more humane and sustainable alternative to traditional methods, further driving innovation in the biologics safety testing sector.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/18592
Restraints on Market Growth
Despite the overall growth, the biologics safety testing market faces certain challenges. One of the most significant obstacles is the high capital and operational costs associated with establishing and maintaining testing facilities. Biosafety testing laboratories require substantial investments in specialized infrastructure, such as BSL-2 or BSL-3 containment systems, and costly testing equipment. Additionally, the demand for skilled personnel to operate advanced testing platforms adds to operational expenses. Smaller biopharmaceutical companies may struggle to afford these costs, limiting their ability to perform in-house safety testing.
Opportunities in the Biologics Safety Testing Market
Expansion of Cell and Gene Therapy Pipelines: As cell and gene therapies gain traction, there is a growing need for specialized safety testing. Regulatory approvals for these therapies are driving demand for assays that target replication-competent viruses, residual DNA, and adventitious agents. The rapid development timelines associated with cell and gene therapies require fast, reliable testing solutions that ensure product safety while meeting stringent regulatory standards.
Adoption of Rapid Microbiological Methods: The shift from traditional culture-based techniques to rapid microbiological methods is revolutionizing biologics safety testing. Technologies such as PCR-based detection, automated imaging systems, and recombinant Factor C assays reduce testing times significantly, which is crucial for high-volume production. These methods enhance efficiency, ensuring faster batch releases and reducing supply chain delays.
Product Insights
In Vitro Testing Kits dominate the biologics safety testing market due to their cost-effectiveness, reproducibility, and growing preference over animal-based testing methods. These kits are integral to routine sterility, endotoxin, mycoplasma, and viral detection assays. The demand for in vitro testing kits is particularly high in vaccine production, monoclonal antibody manufacturing, and recombinant therapeutics. The increasing use of cell and gene therapies also boosts the need for these kits, as each production batch requires thorough safety verification before release.
End-User Analysis
Contract Research Organizations (CROs) and biopharmaceutical companies are the fastest-growing end-users in the biologics safety testing market. Outsourcing trends in the biopharmaceutical sector, coupled with the increasing volume of biologics in the pipeline, have made CROs essential partners for drug development and testing. CROs offer specialized testing services to pharmaceutical companies, enabling them to meet regulatory requirements efficiently and cost-effectively.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/18592
Market Segmentation
Product
Kits & Reagents
Instruments
Services
Test Type
Endotoxin
Sterility
Cell Line Authentication and Characterization
Adventitious Agent Detection
Bioburden
Other
Application
Vaccine and Therapeutics Development
Blood and Blood-related Products Testing
Cellular and Gene Therapy
Tissue-related Products Testing
Stem Cell Research
Regions
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Key Market Players
The biologics safety testing market is highly competitive, with major players offering a variety of testing solutions, reagents, and services. Companies such as Charles River Laboratories, Thermo Fisher Scientific, Lonza Group, SGS S.A., Eurofins Scientific, and Wuxi Apptec are among the leading players in the market. These companies are investing heavily in research and development to improve testing methods, create more efficient and automated systems, and develop animal-free assays.
For instance, Charles River Laboratories introduced the Endosafe Trillium rCR cartridge, an innovative, animal-free solution designed for precise bacterial endotoxin testing. Similarly, Merck KGaA established biosafety testing laboratories in Shanghai, China, to provide local access to testing services for clients in the Asia-Pacific region.
Conclusion
The biologics safety testing market is poised for robust growth, driven by the increasing use of biologics, stringent regulatory requirements, and advancements in testing technologies. As new biologic therapies-especially cell and gene therapies-emerge, the demand for specialized, high-throughput safety testing solutions will continue to rise. While challenges such as high capital costs remain, the opportunities for innovation in testing platforms, particularly in the realms of automation and animal-free methods, will help propel the market forward. With North America maintaining a strong market share and Asia Pacific emerging as the fastest-growing region, the biologics safety testing market is set to play a crucial role in shaping the future of biopharmaceutical development and manufacturing.
Contact Us:
Persistence Market Research
Second Floor, 150 Fleet Street, London, EC4A 2DQ, United Kingdom
USA Phone: +1 646-878-6329
UK Phone: +44 203-837-5656
Email: sales@persistencemarketresearch.com
Web: https://www.persistencemarketresearch.com
About Persistence Market Research:
At Persistence Market Research, we specialize in creating research studies that serve as strategic tools for driving business growth. Established as a proprietary firm in 2012, we have evolved into a registered company in England and Wales in 2023 under the name Persistence Research & Consultancy Services Ltd. With a solid foundation, we have completed over 3600 custom and syndicate market research projects, and delivered more than 2700 projects for other leading market research companies' clients.
Our approach combines traditional market research methods with modern tools to offer comprehensive research solutions. With a decade of experience, we pride ourselves on deriving actionable insights from data to help businesses stay ahead of the competition. Our client base spans multinational corporations, leading consulting firms, investment funds, and government departments. A significant portion of our sales comes from repeat clients, a testament to the value and trust we've built over the years.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Biologics Safety Testing Market Growth Accelerates with 9.7% CAGR Through 2033, Says Persistence Market Research here
News-ID: 4481067 • Views: …
More Releases from Persistence Market Research
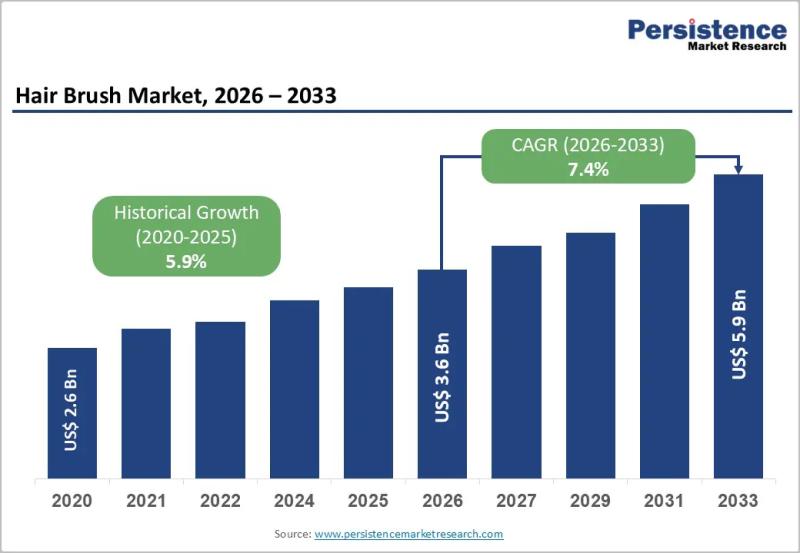
Hair Brush Market to Reach US$ 5.9 Billion by 2033 at 7.4% CAGR | Persistence Ma …
The global Hair Brush market size is likely to be valued at US$ 3.6 Billion in 2026 and is expected to reach US$ 5.9 Billion by 2033, growing at a CAGR of 7.4% during the forecast period from 2026 to 2033. The market is witnessing strong momentum owing to rising consumer awareness regarding personal grooming, hair health, and premium beauty accessories. Increasing disposable income, rapid urbanization, and the growing influence…

Mascarpone Cheese Market Growth Driven by Premium Dessert Demand Reaching US$5.8 …
The global mascarpone cheese market is anticipated to witness substantial growth over the forecast period, driven by evolving consumer preferences toward premium dairy products and the increasing popularity of international cuisines. The market is expected to be valued at US$ 3.6 billion in 2026 and is projected to reach US$ 5.8 billion by 2033, expanding at a robust CAGR of 7.1% between 2026 and 2033. Mascarpone cheese, known for its…

Soy Protein Market to Reach US$13.7 Bn by 2033 Driven by Vegan Food Demand
The global soy protein market is witnessing substantial growth momentum, driven by the increasing global demand for plant-based nutrition, sustainable food ingredients, and high-protein dietary alternatives. The market is expected to be valued at US$ 8.6 billion in 2026 and is projected to reach US$ 13.7 billion by 2033, expanding at a compound annual growth rate (CAGR) of 6.9% during the forecast period from 2026 to 2033. Soy protein has…
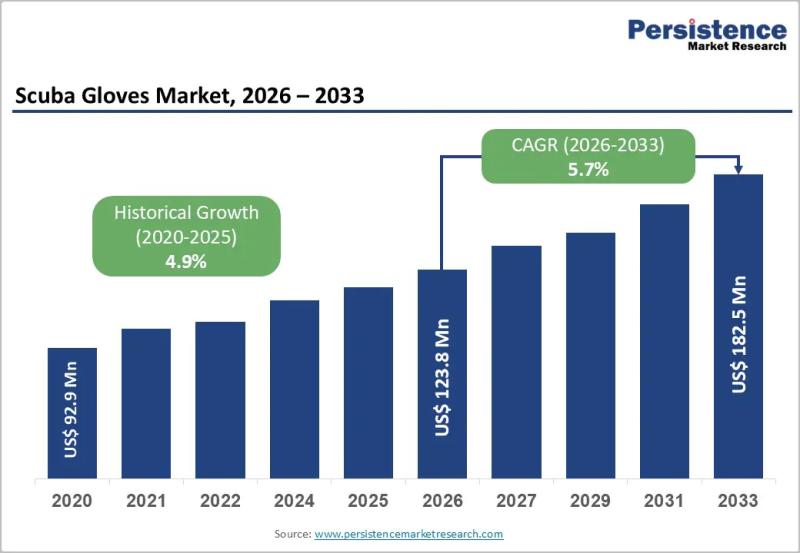
Scuba Gloves Market to Reach US$ 182.5 Million by 2033 at 5.7% CAGR | Persistenc …
The global scuba gloves market is witnessing notable expansion as underwater recreational activities, professional diving operations, and marine tourism continue to gain momentum worldwide. According to industry analysis, the global Scuba Gloves market size is likely to be valued at US$ 123.8 million in 2026 and is expected to reach US$ 182.5 million by 2033, growing at a CAGR of 5.7% during the forecast period from 2026 to 2033. Scuba…
More Releases for Test
Key Differences Between Megger Test, Tan Delta Test, and Hi-Pot Test for Electri …
Electrical insulation plays a critical role in ensuring the safety and efficiency of electrical systems. To assess the condition of insulation and identify potential issues, three common tests are used: the Megger test, Tan Delta test, and Hi-Pot test. Each test serves a unique purpose and provides valuable insights into the state of electrical insulation. Here's a closer look at the differences between these three essential tests.
Megger Test: Insulation Resistance…
Vitamin Test Market: Global Vitamin Test Analysis and Forecast (2023-2029)Vitami …
12.04.2024: Vitamin Test Market Overview
The development of companion diagnostic tools and advances in personalised treatment are driving considerable growth and revolution in the oncology Vitamin Test market. In the era of precision medicine, where healthcare is increasingly customised for individual individuals based on their own genetic and molecular profiles, this market segment is essential. Ongoing innovation and development define the oncology Vitamin Test market. To find particular biomarkers, genetic mutations,…
CAGR 8.1% Homecare Pregnancy Test Kits Market By Type of Test (Urine Test For H …
The Homecare Pregnancy Test Kits market report by Reports and Data provides an extensive overview of the vital elements of the Homecare Pregnancy Test Kits market and factors such as the drivers, restraints, latest trends, supervisory scenario, competitive landscape, technological advancements, and others. Further, it mentions the market shares associated with the market in terms of both value and volume along with the segmentation. Space-age industrial and digitalization tools are…
Home Safety Test Kits Market, Home Safety Test Kits Market Trends, Home Safety T …
“Home Safety Test Kits Market” 2020-2025 Research Report is a professional and in-depth study on the current state of the market. Global Home Safety Test Kits market containing a complete view of the market size, business share, profit estimates, SWOT analysis and the regional landscape of the Industry. The report explains key challenges and future development prospects of the market. The Global Home Safety Test Kits analysis is provided for…
Test Data Management (TDM) Market - test data profiling, test data planning, tes …
The report categorizes the global Test Data Management (TDM) market by top players/brands, region, type, end user, market status, competition landscape, market share, growth rate, future trends, market drivers, opportunities and challenges, sales channels and distributors.
This report studies the global market size of Test Data Management (TDM) in key regions like North America, Europe, Asia Pacific, Central & South America and Middle East & Africa, focuses on the consumption…
Hearing Screening and Diagnostic Devices Market Demands with Major Tests: pure T …
New Market Research Reports Title "Hearing Screening And Diagnostic Devices Market 2018" Has Been Added to Crystal Market Research Report database.
Hearing Screening and Diagnostic Devices - Competitive Insights:
The leading players in the market are Gn Otometrics A/S, Otodynamics, Nashua Hearing Group, Siemens Healthineers, Natus Medical Incorporated, Interacoustics A/S, Neurosoft S.A, Accent Hearing Pty Ltd, MAICO Diagnostics GmbH and IntriCon Corporation. The major players in the market are profiled in detail…
