Press release
Regulatory Affairs & Compliance Market Set for Strong Growth to US$39.2 Billion by 2033, Led by North America's 38.5% Market Share
DataM Intelligence has released a new research report titled "Regulatory Affairs & Compliance Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms of both value and volume, along with emerging opportunities and the overall development outlook of the global Regulatory Affairs & Compliance Market.Ready to scale in the Regulatory Affairs & Compliance Market? Connect with the right partners and unlock new growth opportunities today:- https://www.datamintelligence.com/partner-identification-enquiry/regulatory-affairs-and-compliance-market?ram
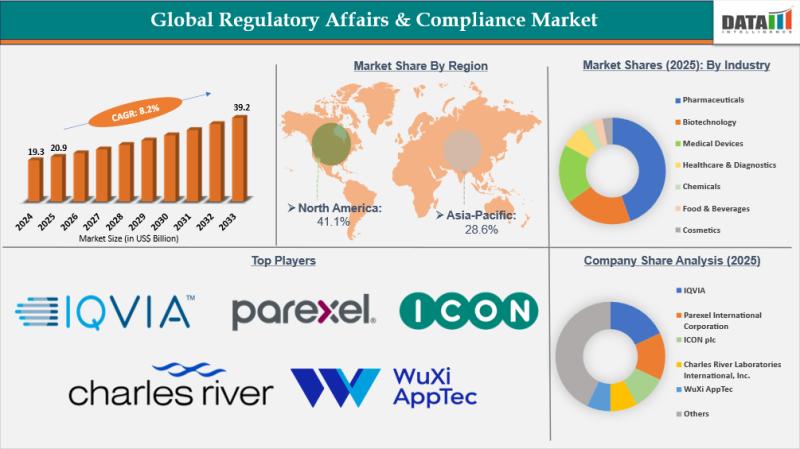
The Global Regulatory Affairs & Compliance market reached US$19.3 Billion in 2024, rising to US$20.9 Billion in 2025 and is expected to reach US$39.2 Billion by 2033, growing at a CAGR of 8.2% from 2026 to 2033.
The market is steadily expanding as pharmaceutical, biotech, and medical device firms navigate increasingly complex global regulations, driven by stringent FDA/EMA requirements, post-pandemic supply chain scrutiny, and the surge in digital health approvals. This growth underscores a shift toward proactive compliance strategies, leveraging AI-driven regulatory intelligence to accelerate drug approvals, ensure data privacy under GDPR, and mitigate risks in biologics and gene therapies with greater efficiency.
Get a Free Sample PDF Of This Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/download-sample/regulatory-affairs-and-compliance-market?ram
Key Industry Developments
United States:
✅ March 2026: Parexel International expanded its Regulatory Science Center of Excellence with AI-powered predictive modeling for FDA submissions, streamlining IND and NDA processes for over 50 biotech clients in oncology and rare diseases.
✅ February 2026: IQVIA launched a cloud-based compliance platform integrating real-time pharmacovigilance tracking, enhancing adverse event reporting compliance amid FDA's updated safety surveillance guidelines.
✅ December 2025: ICON plc acquired a regulatory tech startup to bolster its eCTD submission tools, reducing approval timelines by 30% for complex cell and gene therapies in Phase III trials.
Europe:
✅ January 2026: WuXi AppTec partnered with the EMA on a harmonized framework for biosimilar approvals, deploying automated compliance auditing software for faster market entry in immunology products.
✅ November 2025: Charles River Laboratories introduced a digital dossier management system compliant with EU MDR/IVDR, supporting post-market surveillance for medical devices across 20+ member states.
✅ October 2025: Labcorp enhanced its global regulatory consulting services with blockchain-based audit trails, addressing EMA's data integrity mandates for clinical trial submissions.
Key Players:
Parexel International (MA) Corporation | ICON plc | IQVIA | WuXi AppTec | Charles River Laboratories International, Inc. | Labcorp | Medpace
Strategic Leadership Analysis: Top 5 Key Players in Regulatory Affairs & Compliance Market 2026
-Parexel International (MA) Corporation: Advanced its RegAI suite with machine learning for automated gap analysis in global dossiers, accelerating FDA/EMA filings for biologics and supporting 40% faster market access.
-ICON plc: Rolled out ICONIK Regulatory platform upgrades featuring predictive risk modeling for compliance breaches, optimizing decentralized trial submissions under FDA's modernized guidelines.
-IQVIA: Introduced TotalView Regulatory Intelligence with real-time horizon scanning across 150+ agencies, enabling pharma clients to preempt changes in pharmacovigilance and labeling requirements.
-WuXi AppTec: Launched WuXi RegTrack for end-to-end lifecycle management, integrating AI-driven submission automation that cut CMC review cycles by 25% for Asia-Pacific expansions.
-Charles River Laboratories International, Inc.: Expanded CRADL regulatory consulting with GLP-compliant digital twins for preclinical data, facilitating seamless transitions to IND stages for novel modalities.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/regulatory-affairs-and-compliance-market?ram
Main Drivers and Trends Shaping the Future of the Regulatory Affairs & Compliance Market
-Stringent Global Regulations: Escalating FDA/EMA/PMDA standards for biologics, gene therapies, and digital health are fueling demand for specialized compliance consulting and submission services.
-AI and Automation Adoption: AI tools for regulatory intelligence, eCTD automation, and predictive analytics are transforming manual processes into efficient, error-free workflows.
-Digital Health and Real-World Evidence: Boom in software-as-medical-device (SaMD) approvals and RWE integration requires tailored compliance strategies amid evolving frameworks.
-Pharma Outsourcing Surge: Cost pressures and expertise gaps drive CRO partnerships for end-to-end regulatory support in accelerated drug development pipelines.
-Market Hurdles: Harmonizing divergent regional requirements, cybersecurity risks in digital submissions, talent shortages in regulatory expertise, and rising costs of post-approval changes remain key barriers.
Regional Insights:
-North America: 38.5% (Largest share, propelled by FDA's accelerated pathways and concentration of top CROs/biotechs).
-Asia Pacific: 28% (Fastest growing, with 9.5% CAGR driven by China's NMPA reforms, India's CDSCO expansions, and Japan's PMDA digitization).
-Europe: 22% (Bolstered by EMA centralization, Brexit adjustments, and MDR/IVDR enforcement).
Market Opportunities & Challenges: Regulatory Affairs & Compliance Market 2026
-Opportunities: FDA's Expanded Access and Breakthrough Therapy designations fast-track rare disease filings, while ICH harmonization opens multi-regional submissions. AI interoperability via HL7 standards enables "Regulatory-as-a-Service" models, and EMA's adaptive pathways boost gene therapy compliance investments.
-Challenges: Divergent post-Brexit UKMHRA rules complicate EU-UK strategies; algorithmic transparency in AI tools faces FDA scrutiny. High costs of real-time pharmacovigilance amid "2026 Data Breach Waves" and legacy system migrations pose ongoing risks.
Purchase Corporate License | Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=regulatory-affairs-and-compliance-market?ram
Market Segmentation Analysis:
-By Service Type: Consulting Services Command Premium Amid Complexity
Consulting services lead with expert guidance on global filings and strategy. Outsourcing follows for full-service submissions, training builds internal capabilities, and software/tools enable automation; others include auditing and pharmacovigilance.
-By Category: Regulatory Consulting Dominates Strategy Needs
Regulatory consulting tops for submission planning, followed by compliance management, reporting & submissions, and intelligence & analytics for proactive monitoring.
-By Industry: Pharmaceuticals Drive Core Demand
Pharmaceuticals spearhead due to drug/biologic filings. Medical devices leverage MDR/IVDR expertise, biotechnology focuses on novel therapies, with others spanning food & cosmetics.
-By Deployment Mode: Cloud-Based Solutions Gain Traction
Cloud-based platforms offer scalability for global teams. On-premises suits sensitive data needs, hybrid balances flexibility and security.
-By Regulatory Lifecycle Stage: Pre-Market Approval Leads Pipeline Push
Pre-market approval dominates IND/NDA stages. Clinical trials require GCP compliance, post-market surveillance ensures ongoing adherence, with others covering renewals.
-By Regulatory Authority: FDA Guidelines Set Global Pace
FDA leads North American dominance, EMA shapes Europe, PMDA/NMPA fuel Asia, with others like Health Canada and ANVISA.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription?ram
Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTW
Contact:
Fabian
DataM Intelligence 4market Research LLP
6th Floor, M2 Tech Hub, DataM Intelligence 4market Research LLP, Lalitha Nagar, Habsiguda, Secunderabad, Hyderabad, Telangana 500039
USA: +1 877-441-4866
UK: +44 161-870-5507
Email: fabian@datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Regulatory Affairs & Compliance Market Set for Strong Growth to US$39.2 Billion by 2033, Led by North America's 38.5% Market Share here
News-ID: 4477754 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Recycled Aluminum Market Set to Reach US$100.8 Billion by 2031 with 7.7% CAGR, F …
DataM Intelligence has released a new research report titled "Recycled Aluminum Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Global Battery Materials Recycling Market Set for Explosive Growth at 18% CAGR T …
DataM Intelligence has released a new research report titled "Battery Materials Recycling Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in…
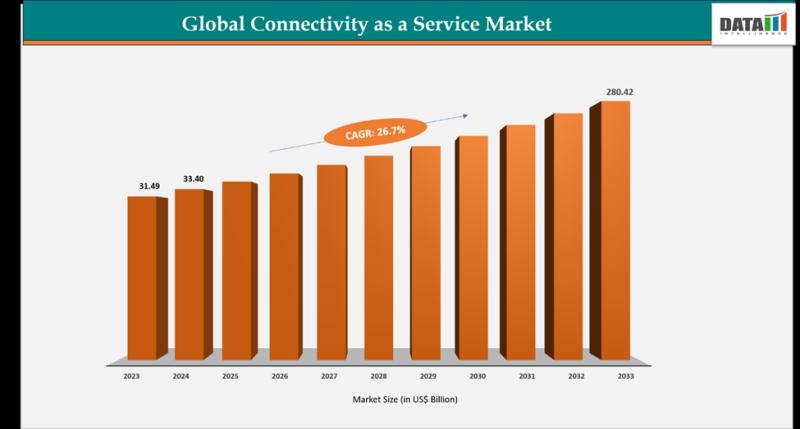
Connectivity as a Service Market Set for Explosive Growth to US$ 280.42 Billion …
DataM Intelligence has released a new research report titled "Connectivity as a Service Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size…
Bioactive Peptides Market Set for Steady Growth to US$ 4,114.21 Million by 2032, …
DataM Intelligence has released a new research report titled "Bioactive Peptides Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…
More Releases for Regulatory
Medical Device Regulatory Affairs Market Medical Device Regulatory Affairs Marke …
"Medical Device Regulatory Affairs Market" in terms of revenue was estimated to be worth $ 6.7 billion in 2024 and is poised to reach $ 18.3 billion by 2034, growing at a CAGR of 10.8% from 2025 to 2034 according to a new report by InsightAce Analytic.
Request For Free Sample Pages:
https://www.insightaceanalytic.com/request-sample/1913
Latest Drivers Restraint and Opportunities Market Snapshot:
Key factors influencing the global medical device regulatory…
Medical Device & IVD Regulatory Affairs Outsourcing Market: Navigating Regulator …
Global healthcare landscape, the Medical Device & IVD Regulatory Affairs Outsourcing Market has emerged as a critical component ensuring the safe and compliant introduction of medical devices and in-vitro diagnostic products to the market. As the industry witnesses significant shifts and challenges, here's an in-depth analysis of the current trends, dynamics, and future prospects within this market segment.
Download sample PDF copy of report: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=79264&utm_source=OpenPR_Ajay&utm_medium=OpenPR
Impact of COVID-19 on European Regulations
The outbreak of…
Regulatory Writing Market - Clear, Concise, Compliant: Redefining Regulatory Wri …
Newark, New Castle, USA - new report, titled Regulatory Writing Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Regulatory Writing market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Regulatory Writing market. The report offers an overview of the market, which…
Complex Regulatory Frameworks
It is challenging for new entrants to enter the FinTech industry because of its complex regulatory framework. All FinTech companies must comply with compliance requirements even before they begin operations, which increases their costs and creates a significant barrier for startups. While regulations are needed to protect consumers, a number of existing laws are slowing down the growth of many Indian FinTech companies, thereby extending their time to reach the…
South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Cr …
Presented report, South Africa Upstream Fiscal and Regulatory Report 2017 - Pending Legislation Creates Regulatory Uncertainty, presents the essential information relating to the terms which govern investment into South Africa’s upstream oil and gas sector. The report sets out in detail the contractual framework under which firms must operate in the industry, clearly defining factors affecting profitability and quantifying the state’s take from hydrocarbon production. Considering political, economic and industry…
Regulatory Affairs Outsourcing Market (Services - Regulatory Submissions, Clinic …
This research study analyzes the market for regulatory affairs outsourcing services in terms of revenue (US$ Mn). The stakeholders of this report comprises the clinical research organizations. The global regulatory affairs outsourcing market has been broadly segmented on the basis of services (Regulatory Submissions, Clinical Trial Applications and Product Registrations, Regulatory Writing and Publishing, Regulatory Consulting and Legal Representation and others regulatory affairs, and Geography (North America, Europe, Asia Pacific,…