Press release
Virtual Clinical Trials Market Accelerates with Decentralized Research Trends and Forecast Outlook to 2033
Virtual Clinical Trials Market Gains Momentum as Decentralized Research Transforms Global HealthcareVirtual clinical trials are redefining the clinical research landscape by enabling decentralized, patient-centric study models that improve accessibility, efficiency, and data accuracy. The integration of digital technologies is significantly reducing traditional trial limitations, accelerating drug development, and expanding participation across diverse populations worldwide.
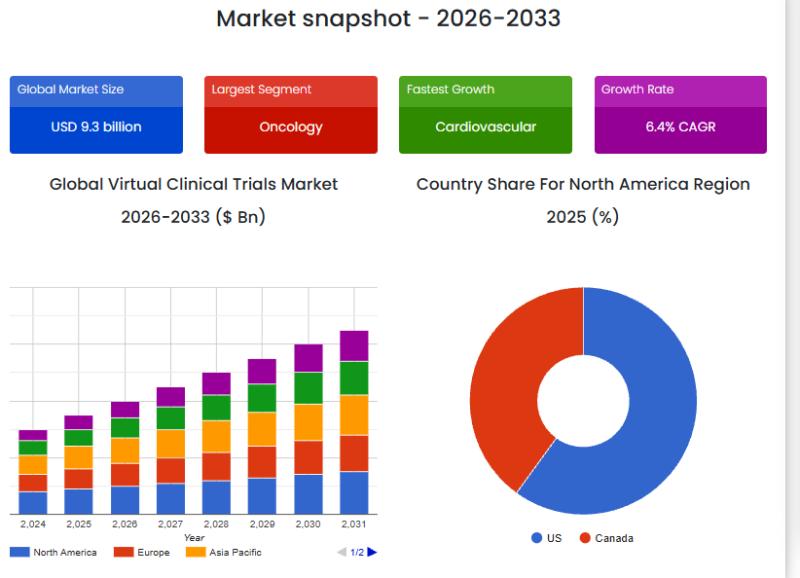
Global demand for virtual clinical trials is steadily increasing as healthcare systems embrace digital transformation. According to the latest market intelligence report by SkyQuest Technology, the Global Virtual Clinical Trials Market size was valued at USD 9.9 billion in 2024 and is poised to grow from USD 10.53 billion in 2025 to USD 17.3 billion by 2033, growing at a CAGR of 6.4% during the forecast period (2026-2033). This growth reflects the rising adoption of remote monitoring, telemedicine, and digital health tools to enhance clinical trial efficiency and patient engagement.
Get a Free Sample Report - https://www.skyquestt.com/sample-request/virtual-clinical-trials-market
What Are Virtual Clinical Trials?
Virtual clinical trials, also known as decentralized clinical trials (DCTs), utilize digital technologies such as telemedicine, wearable devices, mobile health applications, and remote monitoring tools to conduct clinical studies without requiring participants to visit centralized trial sites.
These trials allow patients to participate from their homes or local healthcare facilities, improving recruitment rates, retention, and overall patient experience. By leveraging real-time data collection and cloud-based platforms, virtual clinical trials enhance data accuracy, reduce operational costs, and accelerate study timelines compared to traditional clinical trial models.
Key Market Drivers:
Rising Adoption of Digital Health Technologies
The rapid integration of telehealth, wearable devices, and mobile applications is enabling real-time patient monitoring and seamless data collection. These technologies are making virtual clinical trials more efficient, scalable, and reliable.
Increasing Need for Patient-Centric Trial Models
Traditional clinical trials often face challenges related to patient recruitment and retention. Virtual trials eliminate geographical barriers, making it easier for diverse populations to participate, thereby improving study outcomes and inclusivity.
Cost Efficiency and Faster Trial Timelines
Virtual clinical trials significantly reduce costs associated with physical infrastructure, site management, and patient travel. This cost-effectiveness, combined with faster data collection, is encouraging pharmaceutical and biotechnology companies to adopt decentralized trial models.
Make an Inquiry to Address your Specific Business Needs - https://www.skyquestt.com/speak-with-analyst/virtual-clinical-trials-market
Market Segmentation:
By Study Design
• Interventional Trials - dominant segment driven by drug development activities
• Observational Trials - growing adoption for real-world evidence collection
• Expanded Access Trials
By Indication
• Oncology - leading segment due to high number of cancer trials
• Cardiovascular Diseases
• Neurological Disorders
• Infectious Diseases
• Others
By Technology
• Telemedicine Platforms
• Wearable Devices
• ePRO (Electronic Patient-Reported Outcomes)
• Mobile Health Applications
Regional Insights:
North America dominates the virtual clinical trials market, driven by advanced healthcare infrastructure, strong presence of pharmaceutical companies, and early adoption of digital health technologies. The United States plays a key role in market growth with significant investments in clinical research and innovation.
Europe is witnessing steady growth due to increasing regulatory support and rising adoption of decentralized trial approaches across countries such as Germany, the UK, and France.
Asia-Pacific is expected to be the fastest-growing region, fueled by expanding healthcare infrastructure, increasing clinical trial activities, and growing awareness of digital health solutions in countries like China and India.
Read Full Virtual Clinical Trials Market Report - https://www.skyquestt.com/report/virtual-clinical-trials-market
Competitive Landscape:
The global virtual clinical trials market is highly competitive, with key players focusing on technological innovation and strategic collaborations. Major companies include,
• IQVIA Holdings Inc.
• Parexel International Corporation
• Medpace Holdings Inc.
• ICON plc
• Oracle Corporation
• Medidata Solutions (Dassault Systèmes)
• Labcorp Drug Development
• PRA Health Sciences
• Signant Health
• Covance Inc.
Market Restraints:
Despite strong growth potential, the market faces several challenges. Data privacy and cybersecurity concerns remain critical, as virtual trials rely heavily on digital platforms and sensitive patient data.
Additionally, regulatory complexities and lack of standardized guidelines for decentralized trials across different regions can hinder adoption. Limited digital literacy among certain patient populations and technological barriers in developing regions may also restrict market growth.
About SkyQuest Technology Consulting:
SkyQuest Technology Consulting is a leading Strategy Consulting and Market Research firm, provides syndicated as well as customized research reports and growth consulting services, trusted by CXOs from Fortune 500 Companies, Start-ups, and MSMEs. The company comprises a team of expert research analysts and consultants, adding more than 1200 market research reports in our database each year. These reports offer in-depth analysis on 40+ industries & sub industries across 25 major countries worldwide, serving global clients across diverse industries. The company specializes in delivering customized intelligence, data-driven insights, and strategic advisory services that enable businesses to stay competitive and make informed decisions in rapidly evolving industries.
Contact Us:
SkyQuest Technology Consulting
1 Apache Way, Westford,
Massachusetts 01886
USA (+1) 351-333-4748
Email: sales@skyquestt.com
Visit Our Website: https://www.skyquestt.com/
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Virtual Clinical Trials Market Accelerates with Decentralized Research Trends and Forecast Outlook to 2033 here
News-ID: 4476356 • Views: …
More Releases from SkyQuest Technology
Artificial Intelligence in Healthcare Market Revolutionizing Diagnostics and Pat …
Artificial Intelligence (AI) in Healthcare Market Surges as Smart Technologies Transform Modern Healthcare Systems
Artificial intelligence (AI) is revolutionizing the healthcare industry by enabling faster diagnosis, personalized treatment, predictive analytics, and improved operational efficiency. Hospitals, pharmaceutical companies, and healthcare providers are increasingly integrating AI technologies to enhance patient care, streamline workflows, and reduce healthcare costs.
Global demand for AI-powered healthcare solutions is growing rapidly as digital transformation accelerates across the medical sector.…
Smart Energy Market Driven by AI-Powered Grid Modernization and Forecast Growth …
Smart Energy Market Expands as Digital Grid Technologies and Renewable Integration Accelerate Worldwide
Smart energy solutions are transforming the global energy sector by improving energy efficiency, enabling real-time monitoring, and supporting the integration of renewable power sources. Utilities, governments, and industries are increasingly adopting smart energy technologies to modernize power infrastructure and reduce carbon emissions.
Global demand for smart energy solutions is growing rapidly as countries invest in digital energy systems and…
Bioenergy Market Expands Rapidly with Renewable Fuel Innovations and Sustainabil …
Bioenergy Market Expands as Renewable Energy Demand and Sustainable Fuel Adoption Increase Globally
Bioenergy is emerging as a critical component of the global renewable energy transition, offering sustainable alternatives to fossil fuels through biomass, biofuels, and waste-to-energy solutions. Governments and industries worldwide are increasingly investing in bioenergy technologies to reduce carbon emissions and strengthen energy security.
Global demand for bioenergy solutions is growing steadily as countries focus on cleaner and more sustainable…
Additive Manufacturing Market Expands Rapidly with Industrial 3D Printing Innova …
Additive Manufacturing Market Accelerates as 3D Printing Revolutionizes Modern Manufacturing
Additive manufacturing, commonly known as 3D printing, is transforming industrial production by enabling faster prototyping, reduced material waste, and highly customized manufacturing processes. Industries such as aerospace, automotive, healthcare, and defense are increasingly adopting additive manufacturing technologies to improve efficiency and product innovation.
Global demand for additive manufacturing solutions is rising rapidly as organizations embrace digital manufacturing and Industry 4.0 technologies. According…
More Releases for Trial
Clinical Trial Investigative Site Network Market Clinical Trial Investigative Si …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trial Investigative Site Network Market - (By Therapeutic Areas (Oncology, Cardiology, CNS, Pain Management, Endocrine, Others), By Phase (Phase I, Phase II, Phase III, Phase IV), By End-use (Sponsor, CRO)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic, the Global Clinical Trial Investigative Site Network Market…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Clinical Trial Management System
According to a new market report published by Persistence Market Research “Global Market Study on Clinical Trial Management System: Asia to Witness Highest Growth by 2019” the global clinical trial management system market was valued at USD 844.0 million in 2013 and is expected to grow at a CAGR of 14% from 2014 to 2019, to reach an estimated value of USD 1,848.5 million in 2019.
Request Report TOC @ https://www.persistencemarketresearch.com/methodology/3017
…
Clinical Trial Logistics
Clinical Trial Logistics
16th to 17th May 2011, Marriott Regents Park, London, United Kingdom.
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical trials…
Clinical Trial Logistics
Announcing SMi's 5th annual…
Clinical Trial Logistics conference
16th and 17th May 2011, Central London, UK
www.smi-online.co.uk/2011logistics-london6.asp
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical…