Press release
Tetanus Clinical Trial Pipeline Accelerates as 20+ Pharma Companies Rigorously Develop Drugs for Market Entry | DelveInsight
DelveInsight's "Tetanus Pipeline Insight 2026" report provides comprehensive insights about 20+ companies and 20+ pipeline drugs in the Tetanus pipeline landscape. It covers the Tetanus Pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Tetanus Pipeline Therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.Curious about the latest updates in the Tetanus Pipeline? @ https://www.delveinsight.com/sample-request/tetanus-pipeline-insight [https://www.delveinsight.com/sample-request/tetanus-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Key Takeaways from the Tetanus Pipeline Report
* In January 2026- Tanabe Pharma Corporation initiated a phase 3 study is to evaluate immunogenicity of BK1310 for all antigens (anti-PRP, diphtheria toxin, pertussis, tetanus toxin, and polio virus), after 3 times of injection, when compared noninferiority with co-administration of ActHIB Registered and Tetrabik, as well as efficacy and safety, in healthy infants.
* DelveInsight's Tetanus Pipeline report depicts a robust space with 20+ active players working to develop 20+ pipeline therapies for Tetanus treatment.
* The leading Tetanus Companies such as Boryung Biopharma, Zydus Healthcare, G C Pharma, KM Biologics, Trinomab Biotech, Zydus Lifesciences, Changchun BCHT Biotechnology and others.
* Promising Tetanus Therapies such as TNM002, Human tetanus immunoglobulin (HTIG), GR2001, HTIG, BR-TD-1001, REVAXIS Registered , DT-Polio Registered and others.
Want to know which companies are leading innovation in Tetanus? @ Tetanus Clinical Trials Assessment [https://www.delveinsight.com/sample-request/tetanus-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
The Tetanus Pipeline Report provides disease overview, pipeline scenario and therapeutic assessment of the key pipeline therapies in this domain. The Tetanus Pipeline Report also highlights the unmet needs with respect to the Tetanus.
Tetanus Overview
Tetanus is different from other vaccine-preventable diseases because it does not spread from person to person. The bacteria are usually found in soil, dust, and manure and enter the body through breaks in the skin - usually cuts or puncture wounds caused by contaminated objects. Tetanus is uncommon in the United States, with an average of about 30 reported cases each year. Nearly all cases of tetanus are among people who did not get all the recommended tetanus vaccinations. This includes people who have never received a tetanus vaccine and adults who don't stay up to date on their 10-year booster shots. Tetanus is an infection caused by a bacterium called Clostridium tetani.
Tetanus Emerging Drugs Profile
* GC 3111A : GC Pharma
GC Pharma lead candidate GC 3111A is being developed for the treatment of Tetanus.GC3111A, belongs to the class of bacterial vaccines.It works as immunostimulants.GC 3111A is still in phase II trials for Diphtheria, Pertussis and Tetanus (Prevention) in South Africa.
* BR TD 1001 : Boryung Biopharma
BR TD 1001 is a lead candidate of Boryung Biopharma, which is being developed for the treatment of tetanus.BR TD 1001, was being investigated in Phase 3 study to evaluate immunogenicity by measuring the seroprotection rate against diphtheria and tetanus at 28 days after vaccination with BR-TD-1001 and Td-pur-inj.
* TNM 002 : Trinomab Biotech
TNM002 (human monoclonal antibody against tetanus toxin) is being developed for the treatment of Tetanus. TNM 002, works as Bacterial toxin inhibitors. It is Phase I stage of development for the treatment of Tetanus.
If you're tracking ongoing Tetanus Clinical trials @ Tetanus Treatment Drugs [https://www.delveinsight.com/sample-request/tetanus-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
The Tetanus Pipeline report provides insights into:-
* The report provides detailed insights about companies that are developing therapies for the treatment of Tetanus with aggregate therapies developed by each company for the same.
* It accesses the Different therapeutic candidates segmented into early-stage, mid-stage, and late-stage of development for Tetanus Treatment.
* Tetanus Companies are involved in targeted therapeutics development with respective active and inactive (dormant or discontinued) projects.
* Tetanus Drugs under development based on the stage of development, route of administration, target receptor, monotherapy or combination therapy, a different mechanism of action, and molecular type.
* Detailed analysis of collaborations (company-company collaborations and company-academia collaborations), licensing agreement and financing details for future advancement of the Tetanus market.
Tetanus Companies
Boryung Biopharma, Zydus Healthcare, G C Pharma, KM Biologics, Trinomab Biotech, Zydus Lifesciences, Changchun BCHT Biotechnology and others.
Tetanus Pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as,
* Intra-articular
* Intraocular
* Intrathecal
* Intravenous
* Ophthalmic
* Oral
* Parenteral
* Subcutaneous
* Topical
* Transdermal
Tetanus Products have been categorized under various Molecule types such as,
* Oligonucleotide
* Peptide
* Small molecule
From emerging drug candidates to competitive intelligence, the Tetanus Pipeline Report @ Tetanus Market Drivers and Barriers, and Future Perspectives [https://www.delveinsight.com/sample-request/tetanus-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Scope of the Tetanus Pipeline Report
* Coverage- Global
* Tetanus Companies- Boryung Biopharma, Zydus Healthcare, G C Pharma, KM Biologics, Trinomab Biotech, Zydus Lifesciences, Changchun BCHT Biotechnology and others.
* Tetanus Therapies- TNM002, Human tetanus immunoglobulin (HTIG), GR2001, HTIG, BR-TD-1001, REVAXIS Registered , DT-Polio Registered and others.
* Tetanus Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
* Tetanus Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
Discover what's next for the Tetanus Treatment landscape in this detailed analysis @ Tetanus Emerging Drugs and Major Players [https://www.delveinsight.com/sample-request/tetanus-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Table of Contents
* Introduction
* Executive Summary
* Tetanus: Overview
* Pipeline Therapeutics
* Therapeutic Assessment
* Tetanus- DelveInsight's Analytical Perspective
* Late Stage Products (Phase III)
* Mid Stage Products (Phase II)
* Early Stage Products (Phase I)
* BR TD 1001: Boryung Biopharma
* Preclinical and Discovery Stage Products
* GC 3111A: GC Pharma
* Inactive Products
* Tetanus Key Companies
* Tetanus Key Products
* Tetanus- Unmet Needs
* Tetanus- Market Drivers and Barriers
* Tetanus- Future Perspectives and Conclusion
* Tetanus Analyst Views
* Tetanus Key Companies
* Appendix
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email:Send Email [https://www.abnewswire.com/email_contact_us.php?pr=tetanus-clinical-trial-pipeline-accelerates-as-20-pharma-companies-rigorously-develop-drugs-for-market-entry-delveinsight]
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/report-store/tetanus-pipeline-insight
Legal Disclaimer: Information contained on this page is provided by an independent third-party content provider. ABNewswire makes no warranties or responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you are affiliated with this article or have any complaints or copyright issues related to this article and would like it to be removed, please contact retract@swscontact.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Tetanus Clinical Trial Pipeline Accelerates as 20+ Pharma Companies Rigorously Develop Drugs for Market Entry | DelveInsight here
News-ID: 4468853 • Views: …
More Releases from ABNewswire

Staphylococcal Infections Clinical Trial Pipeline Gains Momentum: 22+ Companies …
DelveInsight's "Staphylococcal Infections Pipeline Insight 2026" report provides comprehensive insights about 22+ companies and 23+ pipeline drugs in the Staphylococcal Infections pipeline landscape. It covers the Staphylococcal Infections Pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Staphylococcal Infections Pipeline Therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Curious about the latest updates…

Servo Manipulator Procurement Guide - Industry Trends and Best Practices
Servo Manipulator Procurement Guide: How to Choose the Right Solution for Your Automation Needs
In today's fast-evolving industrial landscape, automation is no longer optional-it is essential for manufacturers seeking efficiency, precision, and scalability. Among the most critical components in modern automated systems is the servo manipulator, a highly versatile robotic solution widely used in industries such as plastics, automotive, electronics, and packaging.
This Servo Manipulator Procurement Guide is designed to help procurement…

Global New Energy Enters a Transition to "Primary Energy", with China Leading th …
Driven by global energy security and the "dual carbon" goals, the new energy industry [https://www.steelcreate.com/news/global-new-energy-enters-a-transition-to-primary-energy-with-china-leading-the-coordinated-development-of-the-whole-industry-chain/webjs?curPage=product&source=2&source_id=105&is_list=0&is_custom=0]is accelerating its shift from an "alternative energy source" to a "primary energy source". Photovoltaics, wind power, energy storage and other sectors have achieved technological breakthroughs and scale expansion, with booming development in multiple regional markets. Relying on itswhole-industry-chain advantages and technological innovation capabilities, China is deeply involved in the coordinated development of global new energy,…
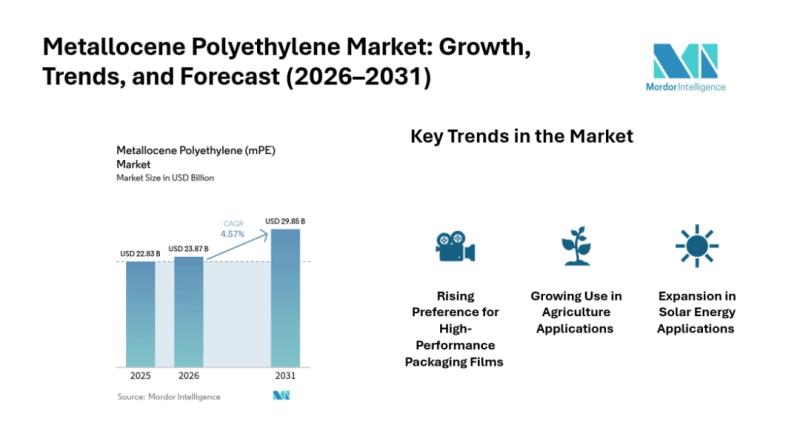
Global Metallocene Polyethylene Market Share Expanding at 4.57% CAGR, Reaching U …
Explore the Metallocene polyethylene market (2026-2031) with mordor intelligence. Get in-depth insights on market size, share, revenue, segments, key drivers, latest trends, and growth opportunities shaping the future of sustainable construction.
Metallocene Polyethylene Market Introduction
The Metallocene polyethylene market is witnessing steady expansion as industries increasingly prefer high-performance polymer materials for packaging, agriculture, and advanced applications. According to market analysis, the Metallocene polyethylene market size was valued at USD 22.83 billion in…
More Releases for Tetanus
Tetanus Market Value Projected to Expand by 2029
Tetanus Market size was valued at USD billion in 2023 and is expected to grow at a compound annual growth rate (CAGR) of 21.36% from 2024 to 2030. Welcome to our comprehensive exploration of the Tetanus market, where we uncover the trends, innovations, and dynamics shaping the landscape of tetanus prevention and treatment. Tetanus, a potentially life-threatening bacterial infection, continues to pose challenges globally, driving the demand for effective preventive…
Diphtheria, Pertussis, and Tetanus Vaccine Market
According to a new report published by Allied Market Research, titled, "Diphtheria, Pertussis, and Tetanus Vaccine Market by Product Type [DTaP (Daptacel, Infanrix, Kinrix, Pediarix, Pentacel, and Quaracel), Td (Decavac and Tenivac), and Tdap (Boostrix and Adacel)], Vaccine Type (Whole-Cell Vaccine and Acellular Vaccine), Age Group (Adult and Pediatric), and End User (Hospitals, Clinics, and Vaccination Centers) - Global Opportunity and Forecast, 2019-2026 ".
The Global market size of Diphtheria, Pertussis,…
Tetanus Therapeutics- Pipeline Analysis 2018 | Zydus Group
Tetanus, also known as lockjaw is a bacterial disease caused by Clostridium tetani. The bacteria are usually found in dust, soil, and manure and enter the body through breaks in the skin. The spores develop into bacteria when they enter the body.
Download the sample report @ https://www.pharmaproff.com/request-sample/1074
These spores can get into the body through broken skin, usually through injuries from contaminated objects. Certain breaks in the skin are more…
Tetanus-Pipeline Review H2 2018
Tetanus (Infectious Disease) pipeline guide helps in identifying and tracking emerging players in the market and their portfolios, enhances decision making capabilities and helps to create effective counter strategies to gain competitive advantage. The guide is built using data and information sourced from Publisher's proprietary databases, company/university websites, clinical trial registries, conferences, SEC filings, investor presentations and featured press releases from company/university sites and industry-specific third party sources. Additionally, various…
Tetanus-Pipeline Review H2 2018
Tetanus (Infectious Disease) pipeline guide helps in identifying and tracking emerging players in the market and their portfolios, enhances decision making capabilities and helps to create effective counter strategies to gain competitive advantage. The guide is built using data and information sourced from Publisher's proprietary databases, company/university websites, clinical trial registries, conferences, SEC filings, investor presentations and featured press releases from company/university sites and industry-specific third party sources. Additionally, various…
Tetanus - Pipeline Review, H1 2017
ReportsWorldwide has announced the addition of a new report title Tetanus - Pipeline Review, H1 2017 to its growing collection of premium market research reports.
Global Markets Direct's latest Pharmaceutical and Healthcare disease pipeline guide Tetanus - Pipeline Review, H1 2017, provides an overview of the Tetanus (Infectious Disease) pipeline landscape.
Tetanus is a serious illness caused by Clostridium bacteria. Signs and symptoms of tetanus include spasms and stiffness in jaw muscles,…
