Press release
In Vitro Diagnostics (IVD) Market Outlook 2026-2033: Size, Share, and Strategic Developments
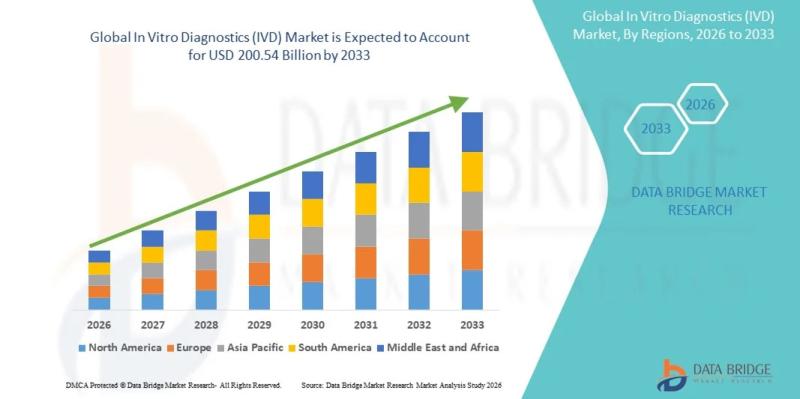
As per Data Bridge Market Research analysis, the In Vitro Diagnostics (IVD) Market was estimated at USD 135.73 billion in 2025. The market is expected to grow from USD 142.52 billion in 2026 to USD 200.54 billion in 2033, at a CAGR of 5.00% during the forecast period with driven by the rising demand for early disease detection, technological advancements in diagnostic platforms, increasing prevalence of chronic and infectious diseases, and expanding adoption of precision medicine and personalized healthcare solutions.The global In Vitro Diagnostics (IVD) market demonstrates steady expansion supported by the integration of automation, molecular diagnostics, and AI-enabled laboratory systems. Rising healthcare expenditure and increasing screening programs globally are further strengthening market penetration. Additionally, regulatory emphasis on early diagnosis and preventive healthcare is accelerating adoption across hospitals and diagnostic laboratories.
Get the full PDF sample copy of the report: Includes full table of contents, list of tables and figures, and graphs: https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-in-vitro-diagnostics-ivd-market
Market Size & Forecast
2025 Market Size: USD 135.73 Billion
2026 Projected Market Size: USD 142.52 Billion
2033 Projected Market Size: USD 200.54 Billion
CAGR (2026-2033): 5.00%
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Key Market Report Takeaways
Largest Region: North America holds the leading market share (~38-40%) due to advanced healthcare infrastructure, high testing volumes, and strong presence of key manufacturers
Fastest-Growing Region: Asia-Pacific, driven by expanding healthcare access, rising population base, and government-led diagnostic programs
Highest Market Share by Product Type: Reagents & consumables segment dominates due to recurring usage in diagnostic workflows
Dominant Application Segment: Infectious disease testing remains the leading application due to global disease burden and outbreak preparedness initiatives
Leading End-Use Segment: Hospitals and clinical laboratories account for the highest adoption of IVD solutions
Market Trends & Highlights
North America dominates the global IVD market due to strong reimbursement frameworks, advanced diagnostic infrastructure, and high adoption of automated laboratory systems.
Asia-Pacific is the fastest-growing region, driven by expanding healthcare investments, rising awareness of early disease detection, and increasing diagnostic laboratory networks.
Reagents and consumables segment remains dominant due to continuous demand across molecular, clinical chemistry, and immunodiagnostic workflows.
Rising burden of chronic diseases such as cancer, diabetes, and cardiovascular disorders is significantly increasing diagnostic testing volumes globally.
Adoption of molecular diagnostics, PCR-based testing, and next-generation sequencing is reshaping clinical diagnostics accuracy and speed.
Strategic collaborations and investments by global diagnostic leaders are enhancing innovation in point-of-care and decentralized testing solutions.
Details about the report and current availability can be viewed : https://www.databridgemarketresearch.com/reports/global-in-vitro-diagnostics-ivd-market
Market Dynamics
Market Drivers
1. Rising prevalence of chronic and infectious diseases
The increasing global incidence of diseases such as cancer, diabetes, and cardiovascular disorders is driving demand for early and accurate diagnostic solutions. Infectious disease outbreaks have further emphasized the importance of rapid testing infrastructure. This has led to higher testing volumes across hospitals and laboratories. The need for continuous monitoring and disease surveillance is strengthening IVD adoption globally.
2. Technological advancements in molecular diagnostics
Advancements in PCR, immunoassays, and next-generation sequencing have significantly improved diagnostic accuracy and turnaround time. Automation and AI integration in laboratory workflows are enhancing operational efficiency. These innovations are enabling high-throughput testing capabilities. As a result, laboratories are increasingly adopting advanced diagnostic platforms.
3. Expansion of personalized and precision medicine
The shift toward individualized treatment approaches is increasing reliance on diagnostic data. IVD tools are essential for identifying biomarkers and guiding targeted therapies. This trend is particularly strong in oncology and genetic testing applications. Pharmaceutical-diagnostic collaborations are further strengthening market growth.
4. Government initiatives for early disease detection
National screening programs and public health initiatives are promoting early diagnosis. Governments are investing in diagnostic infrastructure and laboratory networks. Subsidized testing programs are improving accessibility in developing economies. These initiatives are significantly expanding the diagnostic testing base.
5. Growing adoption of point-of-care testing (POCT)
Decentralized testing solutions are gaining traction due to demand for rapid results. POCT devices are reducing dependency on centralized laboratories. This is particularly important in emergency care and remote regions. The trend is enhancing diagnostic accessibility and efficiency.
Market Restraints
1. High cost of advanced diagnostic systems
Advanced molecular and automated diagnostic platforms require significant capital investment. Smaller laboratories and healthcare facilities face affordability challenges. Maintenance and operational costs further increase financial burden. This limits adoption in cost-sensitive markets.
2. Complex regulatory approval processes
IVD products are subject to stringent regulatory frameworks across regions. Approval timelines can delay product commercialization. Compliance requirements vary significantly across countries. This creates operational complexity for global manufacturers.
3. Reimbursement limitations in developing regions
Inadequate insurance coverage restricts patient access to diagnostic testing. Out-of-pocket expenses remain high in low-income economies. This reduces testing frequency and market penetration. Reimbursement gaps slow down overall adoption.
4. Skilled workforce shortages
Advanced diagnostic technologies require trained laboratory professionals. Many regions face shortages of skilled technicians and pathologists. This affects efficient utilization of diagnostic systems. Training and workforce development remain critical challenges.
Market Opportunities
1. Expansion in emerging economies
Asia-Pacific, Latin America, and the Middle East present significant growth potential. Rising healthcare investments are improving laboratory infrastructure. Increasing awareness of preventive healthcare is driving demand. These regions offer untapped diagnostic market opportunities.
2. Growth of decentralized diagnostics
Point-of-care and home-based testing solutions are expanding rapidly. These technologies reduce dependency on centralized labs. They improve accessibility in rural and remote areas. This shift is opening new revenue streams for manufacturers.
3. Integration of AI and digital pathology
Artificial intelligence is enhancing diagnostic accuracy and workflow automation. Digital pathology systems are enabling remote diagnostics and telemedicine integration. Predictive analytics is improving disease detection efficiency. This represents a major technological growth avenue.
4. Expansion of oncology and genetic testing
Rising cancer incidence is driving demand for biomarker-based diagnostics. Genetic screening is becoming integral to personalized treatment strategies. Pharmaceutical partnerships are accelerating innovation in this segment. This is creating high-value diagnostic opportunities.
Market Challenges
1. Intense market competition
The presence of established global players increases competitive pressure. Companies compete on innovation, pricing, and product differentiation. Continuous R&D investment is required to maintain market position. This creates barriers for new entrants.
2. Pricing pressure on diagnostic services
Healthcare providers are increasingly focusing on cost reduction. Diagnostic companies face pressure to reduce test pricing. This impacts profit margins across the value chain. Cost efficiency is becoming critical for sustainability.
3. Regulatory harmonization issues
Differences in global regulatory standards create market entry challenges. Manufacturers must comply with multiple regulatory frameworks. This increases time-to-market for new products. Regulatory uncertainty impacts strategic planning.
4. Supply chain and raw material volatility
Dependence on specialized reagents and components creates supply risks. Global disruptions can affect production and distribution. Price fluctuations in raw materials impact cost structures. This challenges operational stability.
Get Detailed Insights Before You Buy - Request Complete Market Intelligence Now. https://www.databridgemarketresearch.com/inquire-before-buying/?dbmr=global-in-vitro-diagnostics-ivd-market
Market Segment Insights
By Application
Infectious disease testing is the largest segment due to continuous global demand for outbreak detection and screening programs. Oncology testing is the fastest-growing segment driven by increasing cancer prevalence and biomarker-based diagnostics. Rising adoption of molecular testing technologies is further enhancing application diversity across chronic disease management.
By End Use
Hospitals dominate the market due to high patient inflow and integrated diagnostic facilities. Diagnostic laboratories are the fastest-growing segment owing to outsourcing trends and centralized testing efficiency. Increasing investments in laboratory automation are further strengthening this segment.
By Integration Type
Standalone diagnostic systems represent the largest share due to widespread installed base. Integrated systems are growing fastest as laboratories adopt automated and connected workflows to improve efficiency and reduce turnaround time.
By Deployment Model
On-premises systems dominate due to traditional laboratory infrastructure. Cloud-based diagnostic platforms are the fastest-growing segment, driven by data analytics integration and remote accessibility.
By Technology
Immunoassay technology holds the largest share due to extensive clinical usage. Molecular diagnostics is the fastest-growing segment supported by advancements in PCR, sequencing, and precision medicine applications.
Regional Insights
North America
North America leads the global IVD market due to advanced healthcare infrastructure, high diagnostic testing rates, and strong presence of leading manufacturers such as Roche Diagnostics, Abbott Laboratories, and Danaher Corporation. The region benefits from strong reimbursement systems and early adoption of innovative technologies.
Europe
Europe is an established market driven by strong regulatory frameworks and increasing adoption of molecular diagnostics. Countries such as Germany, France, and the UK are investing heavily in laboratory modernization. The presence of companies like bioMérieux strengthens regional innovation capacity.
Asia-Pacific
Asia-Pacific is the fastest-growing region due to rising healthcare expenditure, large population base, and expanding diagnostic infrastructure. China, India, and Japan are key contributors to growth. Government-led screening initiatives and increasing private laboratory networks are accelerating adoption.
Middle East & Africa
The region represents an emerging opportunity market supported by gradual healthcare infrastructure development. Rising investment in hospitals and diagnostic centers is improving access to IVD solutions. Demand for infectious disease testing is particularly strong due to regional health challenges.
Competitive Landscape (Advanced Level)
The global In Vitro Diagnostics (IVD) market is moderately consolidated, with the top 10 players accounting for approximately 60-65% of total market share. The competitive environment is characterized by continuous innovation, mergers and acquisitions, and expansion into emerging markets.
Key global leaders include F. Hoffmann-La Roche, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, and Danaher Corporation.
These companies focus on expanding molecular diagnostics portfolios, strengthening point-of-care testing capabilities, and integrating digital health technologies. Strategic collaborations with healthcare providers and research institutions are further enhancing market positioning and global reach.
Check out more related studies published by Data Bridge Market Research:
https://www.databridgemarketresearch.com/reports/global-poultry-intestinal-health-market
https://www.databridgemarketresearch.com/reports/global-cookie-mixes-market
https://www.databridgemarketresearch.com/reports/global-biostimulants-market
https://www.databridgemarketresearch.com/reports/global-floriculture-market
https://www.databridgemarketresearch.com/reports/global-non-alcoholic-wine-market
https://www.databridgemarketresearch.com/reports/point-care-testing-poct-market
https://www.databridgemarketresearch.com/reports/global-mayonnaise-market
https://www.databridgemarketresearch.com/reports/global-sauces-dressings-and-condiments-market
https://www.databridgemarketresearch.com/reports/global-luxury-jewellery-market
https://www.databridgemarketresearch.com/reports/global-molecular-biology-kits-and-reagent-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC: +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
Data Bridge Market Research:
Today's trends are a great way to predict future events!
Data Bridge Market Research is a market research and consulting firm known for its innovative and unique approach, coupled with unparalleled resilience and integrated methods. We are committed to identifying prime market opportunities and delivering insightful information to help your business excel in the marketplace. Data Bridge provides customized solutions to complex business challenges, ensuring a seamless decision-making process. Founded in Pune in 2015, Data Bridge is the result of profound wisdom and extensive experience.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release In Vitro Diagnostics (IVD) Market Outlook 2026-2033: Size, Share, and Strategic Developments here
News-ID: 4464626 • Views: …
More Releases from Data Bridge Market Research
Cell-Based Immunotherapy Market Growth Report: Advancements in Personalized Canc …
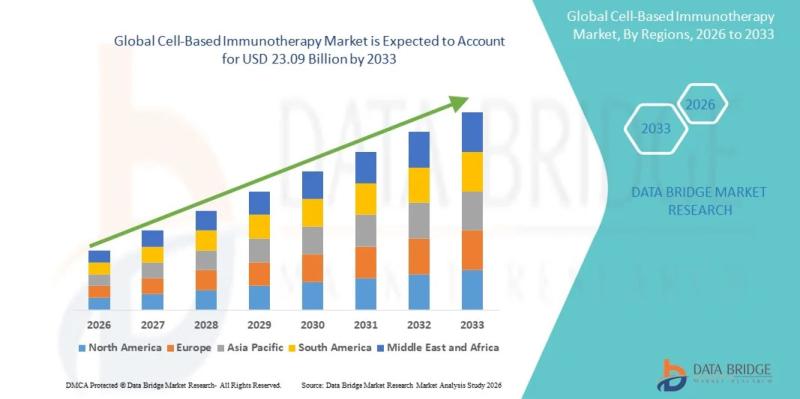
As per Data Bridge Market Research analysis, the cell-based immunotherapy market was estimated at USD 5.74 billion in 2025. The market is expected to grow from USD 6.83 billion in 2026 to USD 23.09 billion in 2033, at a CAGR of 19.01% during the forecast period with driven by the rising demand for advanced oncology treatments, increasing adoption of CAR-T and T-cell therapies, expanding clinical trial pipelines, and rising global…
Biostimulants Market Industry Overview, Regional Analysis & Competitive Landscap …
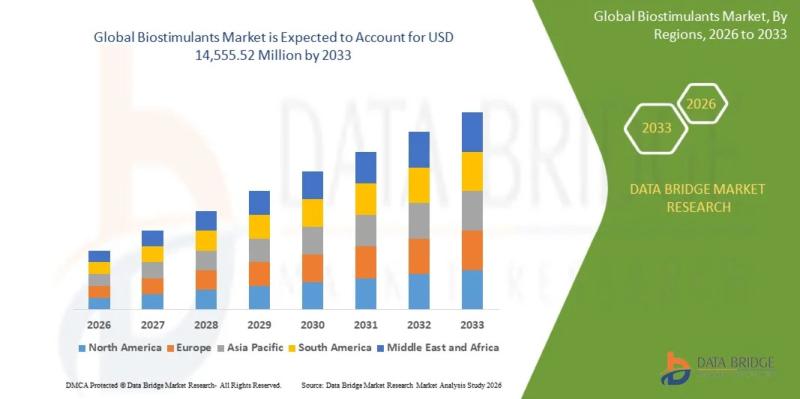
As per Data Bridge Market Research analysis, the Biostimulants Market was estimated at USD 5.78 billion in 2025. The market is expected to grow from USD 6.49 billion in 2026 to USD 14.56 billion in 2033, at a CAGR of 12.3% during the forecast period with driven by the rising demand for sustainable agricultural inputs, soil fertility enhancement solutions, increasing global food security pressures, and regulatory support for eco-friendly crop…
Point-Of-Care-Testing (POCT) Market Size, Share, Growth & Forecast (2026-2033): …
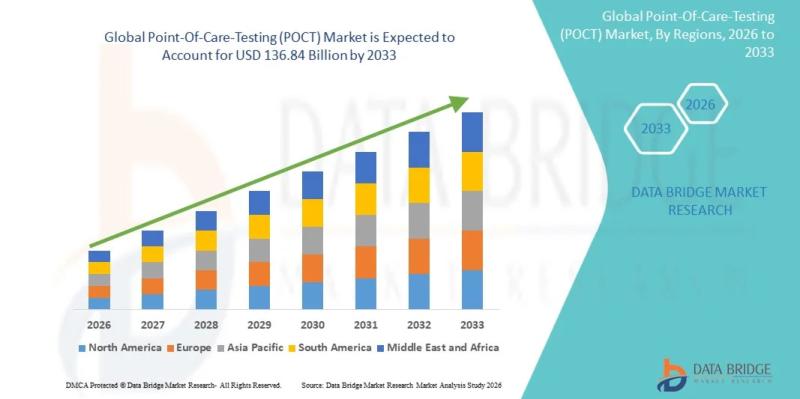
As per Data Bridge Market Research analysis, the Point-Of-Care-Testing (POCT) Market was estimated at USD 63.28 billion in 2025. The market is expected to grow from USD 69.74 billion in 2026 to USD 136.84 billion in 2033, at a CAGR of 10.2% during the forecast period.
The global Point-Of-Care-Testing (POCT) market is experiencing strong expansion, supported by rapid decentralization of diagnostic services, increasing prevalence of infectious and chronic diseases, and…
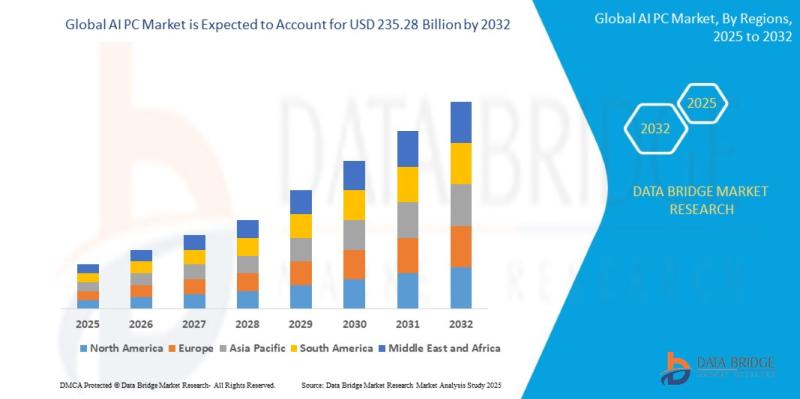
AI PC Market to Reach USD 235.28 Billion by 2032, Driven by Rapid Adoption of On …
As per Data Bridge Market Research analysis, the AI PC Market was estimated at USD 60.85 billion in 2025. The market is expected to grow from USD 73.70 billion in 2026 to USD 235.28 billion in 2032, at a CAGR of 21.30% during the forecast period of 2026-2032 with driven by the rising demand for on-device AI processing, increasing adoption of generative AI applications, expansion of AI-enabled chipsets, and rising…
More Releases for IVD
Transformative Trends Impacting the Cancer In Vitro Diagnostics (IVD) Market Lan …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Cancer In Vitro Diagnostics (IVD) Market Size By 2025?
The market size for cancer in vitro diagnostics (IVD) has seen significant growth in the past few years. The market value, which is expected to be $13.36 billion in 2024, is projected to increase to $14.32…
In Vitro Diagnostics (IVD) Market
With the watchful use of established and advanced tools such as SWOT analysis and Porter's Five Forces Analysis, this market report has been structured. While preparing this In Vitro Diagnostics (IVD) Market research report, few of the attributes that have been adopted include highest level of spirit, practical solutions, committed research and analysis, innovation, integrated approaches, and most up-to-date technology.
Every possible effort has been taken while researching and analysing…
Companion Animal IVD Market - Guiding the Path to Optimal Health: Empowering Vet …
Newark, New Castle, USA - new report, titled Companion Animal IVD Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Companion Animal IVD market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Companion Animal IVD market. The report offers an overview of…
IVD Market 2021 | Detailed Report
The IVD market research report delivers accurate data and innovative corporate analysis, helping organizations of all sizes make appropriate decisions. The IVD report also incorporates the current and future global market outlook in the emerging and developed markets. Moreover, the report also investigates regions/countries expected to witness the fastest growth rates during the forecast period.
The IVD research report also provides insights of different regions that are contributing market growth.…
Liquid Biopsy IVD Market 2021 | Detailed Report
According to Market Study Report, Liquid Biopsy IVD Market provides a comprehensive analysis of the Liquid Biopsy IVD Market segments, including their dynamics, size, growth, regulatory requirements, competitive landscape, and emerging opportunities of global industry. An exclusive data offered in this report is collected by research and industry experts team.
Get Free Sample PDF (including full TOC, Tables and Figures) of Liquid Biopsy IVD Market @ https://www.reportsnreports.com/contacts/requestsample.aspx?name=4623688
The report provides a…
Asia IVD Market
According to a new report published by Allied Market Research, the Asia Pacific In-vitro diagnostics market was valued at $12.9 billion in 2015, and is expected to reach $19.0 billion registering a CAGR of 5.6% during 2016 to 2022. The report offers a detailed analysis of the key segments, top investment pockets, changing dynamics, market size & estimations, and competitive scenario.
Download Free Sample Report @ https://www.alliedmarketresearch.com/request-sample/1256
The Asia-Pacific IVD market is…