Press release
Key Players and Competitive Overview in the Clinical Trial Software Market
The clinical trial software market is undergoing significant transformation fueled by technological innovation and shifting industry demands. As pharmaceutical research continues to evolve, software solutions play an increasingly vital role in optimizing trial management, enhancing data accuracy, and accelerating drug development timelines. This overview explores the market's projected growth, the key players shaping its future, emerging trends, and important segmentation details.Clinical Trial Software Market Size and Growth Outlook
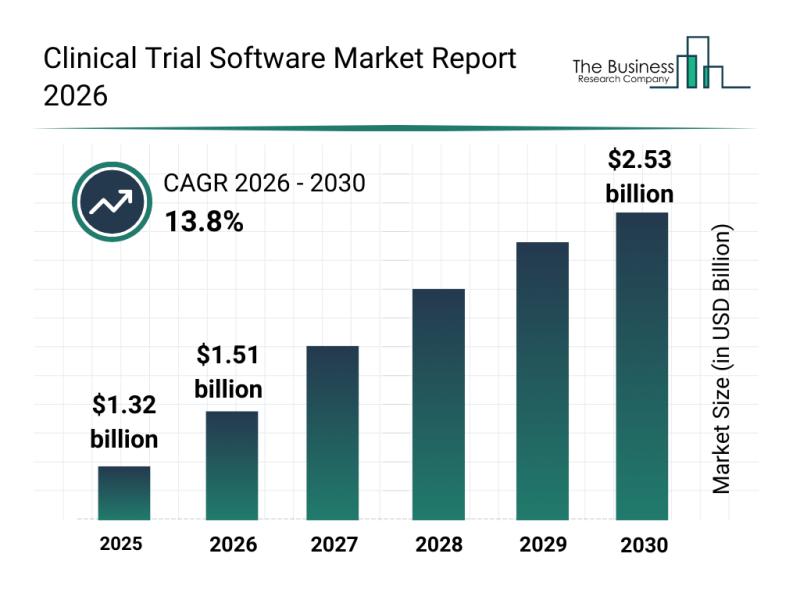
The clinical trial software industry is poised for rapid expansion, with its market size expected to reach $2.53 billion by 2030. This growth reflects a robust compound annual growth rate (CAGR) of 13.8%. Several factors are driving this upward trajectory, including the rising adoption of virtual and decentralized clinical trials, increased investments in SaaS-based trial platforms, broader integration of artificial intelligence into clinical workflows, the expansion of remote patient monitoring capabilities, and heightened regulatory emphasis on data integrity. Notable trends steering the market include a growing preference for cloud-based clinical trial software, the use of AI-powered data analytics tools, demand for decentralized trial platforms, the merging of electronic data capture (EDC) with clinical trial management systems (CTMS), and stronger attention to data security and compliance protocols.
Download a free sample of the clinical trial software market report:
https://www.thebusinessresearchcompany.com/report/clinical-trial-software-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Apr_PR
Top Companies Leading the Clinical Trial Software Industry
The clinical trial software landscape features several prominent companies that are driving innovation and market growth. These industry leaders include Oracle Corporation, Veeva Systems, Medidata Solutions, Signant Health Inc., Clario Inc., Greenphire Inc., Medrio Inc., Arisglobal LLC, Anju Software Inc., ClinCapture Inc., OpenClinica LLC, Castor, Viedoc, Clinion, OmniComm (TrialMaster), Curebase, Clinevo, RealTime-CTMS, REDCap Cloud, Zelta, Trialytix, Florence eBinders, Dot Compliance, ClinPlus CTMS, nQuery, and Jeeva Clinical Trials.
Strategic Acquisition to Enhance Clinical Trial Automation
In October 2023, Certara Inc., a US-based clinical software provider, acquired Formedix LLC, though the financial terms were not disclosed. This acquisition is aimed at boosting Certara's clinical trial automation capabilities by incorporating Formedix's expertise in data standardization and CDISC compliance. This integration is expected to streamline regulatory submissions, improve the efficiency of clinical trials, and accelerate drug development processes for pharmaceutical companies and regulatory authorities. Formedix LLC specializes in cloud-based software that simplifies clinical trial workflows.
The clinical trial software market is undergoing significant transformation fueled by technological innovation and shifting industry demands. As pharmaceutical research continues to evolve, software solutions play an increasingly vital role in optimizing trial management, enhancing data accuracy, and accelerating drug development timelines. This overview explores the market's projected growth, the key players shaping its future, emerging trends, and important segmentation details.
Clinical Trial Software Market Size and Growth Outlook
The clinical trial software industry is poised for rapid expansion, with its market size expected to reach $2.53 billion by 2030. This growth reflects a robust compound annual growth rate (CAGR) of 13.8%. Several factors are driving this upward trajectory, including the rising adoption of virtual and decentralized clinical trials, increased investments in SaaS-based trial platforms, broader integration of artificial intelligence into clinical workflows, the expansion of remote patient monitoring capabilities, and heightened regulatory emphasis on data integrity. Notable trends steering the market include a growing preference for cloud-based clinical trial software, the use of AI-powered data analytics tools, demand for decentralized trial platforms, the merging of electronic data capture (EDC) with clinical trial management systems (CTMS), and stronger attention to data security and compliance protocols.
Download a free sample of the clinical trial software market report:
https://www.thebusinessresearchcompany.com/report/clinical-trial-software-global-market-report?utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Apr_PR
Top Companies Leading the Clinical Trial Software Industry
The clinical trial software landscape features several prominent companies that are driving innovation and market growth. These industry leaders include Oracle Corporation, Veeva Systems, Medidata Solutions, Signant Health Inc., Clario Inc., Greenphire Inc., Medrio Inc., Arisglobal LLC, Anju Software Inc., ClinCapture Inc., OpenClinica LLC, Castor, Viedoc, Clinion, OmniComm (TrialMaster), Curebase, Clinevo, RealTime-CTMS, REDCap Cloud, Zelta, Trialytix, Florence eBinders, Dot Compliance, ClinPlus CTMS, nQuery, and Jeeva Clinical Trials.
Strategic Acquisition to Enhance Clinical Trial Automation
In October 2023, Certara Inc., a US-based clinical software provider, acquired Formedix LLC, though the financial terms were not disclosed. This acquisition is aimed at boosting Certara's clinical trial automation capabilities by incorporating Formedix's expertise in data standardization and CDISC compliance. This integration is expected to streamline regulatory submissions, improve the efficiency of clinical trials, and accelerate drug development processes for pharmaceutical companies and regulatory authorities. Formedix LLC specializes in cloud-based software that simplifies clinical trial workflows.
Current Trends Positively Influencing Clinical Trial Software Growth
The clinical trial software market is benefiting from innovations that enhance the efficiency and accuracy of trials. One notable development is the focus on advanced ECG monitoring equipment such as reusable ECG patches, which improve patient comfort, allow for longer wear times, and provide continuous cardiac data monitoring. These reusable devices can be cleaned and sterilized for multiple uses, supporting both intermittent and continuous heart activity monitoring.
For instance, in May 2024, Canadian wearable medical device manufacturer Vivalink launched an all-in-one ambulatory cardiac monitoring solution designed for remote patient monitoring in ambulatory care. The system features reusable ECG patches with long-lasting battery life and expanded data storage, mobile applications for real-time data transmission, and clinician portals that deliver detailed analyses and customizable reports. This turnkey solution aims to simplify cardiac monitoring deployment, boost diagnostic precision, improve patient outcomes, and reduce operational burdens and costs for healthcare providers.
Key Segments Defining the Clinical Trial Software Market
This market is segmented by deployment method, software type, and end-user categories to provide a comprehensive understanding of its structure. Deployment options include On-Premises, Web-Based Clinical Trial Software, Cloud-Based Clinical Trial Software, and other deployments. Software categories cover Electronic Data Capture (EDC), Electronic Clinical Outcome Assessment (eCOA) or Electronic Patient-Reported Outcome (ePRO), and Electronic Informed Consent. End-users span pharmaceutical and biotechnology firms, contract research organizations (CROs), medical device manufacturers, and other users.
Detailed Subsegments Within Deployment Categories
Within On-Premises deployment, subsegments include Enterprise Clinical Trial Management Systems and Site-Based Clinical Trial Solutions. The Web-Based deployment covers Electronic Data Capture (EDC) systems, Clinical Trial Management Systems (CTMS), and Randomization and Trial Supply Management (RTSM). Cloud-Based Clinical Trial Software encompasses Software-as-a-Service (SaaS) platforms, AI-powered clinical trial solutions, and remote monitoring and decentralized trial systems. Other deployments comprise Hybrid Clinical Trial Solutions and custom-built clinical trial software options.
Reach out to us:
The Business Research Company: https://www.thebusinessresearchcompany.com/,
Americas +1 310-496-7795,
Europe +44 7882 955267,
Asia & Others +44 7882 955267 & +91 8897263534,
Email us at info@tbrc.info.
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Learn More About The Business Research Company
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead.Our flagship product, the Global Market Model (GMM), is a premier market intelligence platform delivering comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Key Players and Competitive Overview in the Clinical Trial Software Market here
News-ID: 4464049 • Views: …
More Releases from The Business Research Company
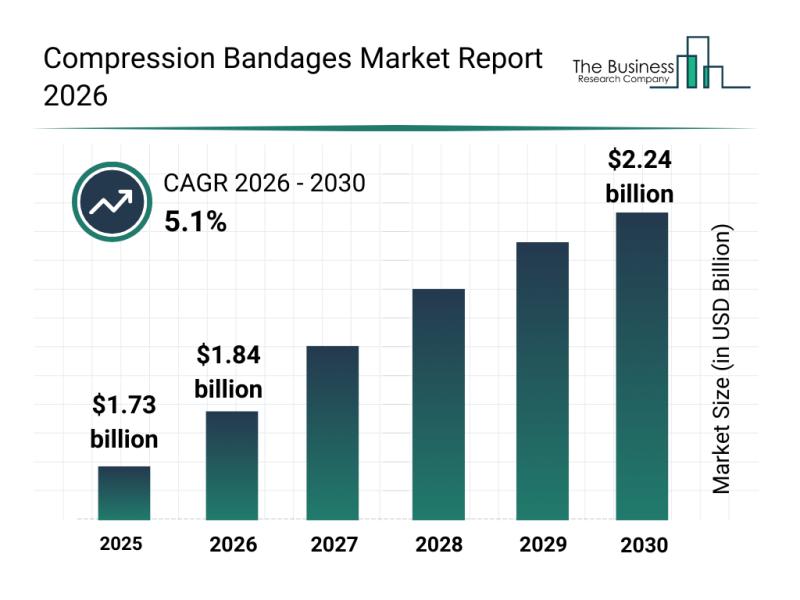
Future Perspective: Key Trends Shaping the Compression Bandages Market Until 203 …
The compression bandages sector is poised for considerable expansion as healthcare practices evolve and patient needs become more specialized. With rising awareness around vascular health and advancements in medical materials, this market is set to experience steady growth. Let's explore the current market size, leading companies, key trends, and segmentation that define this growing industry.
Projected Market Size for Compression Bandages by 2030
The compression bandages market is predicted to achieve…
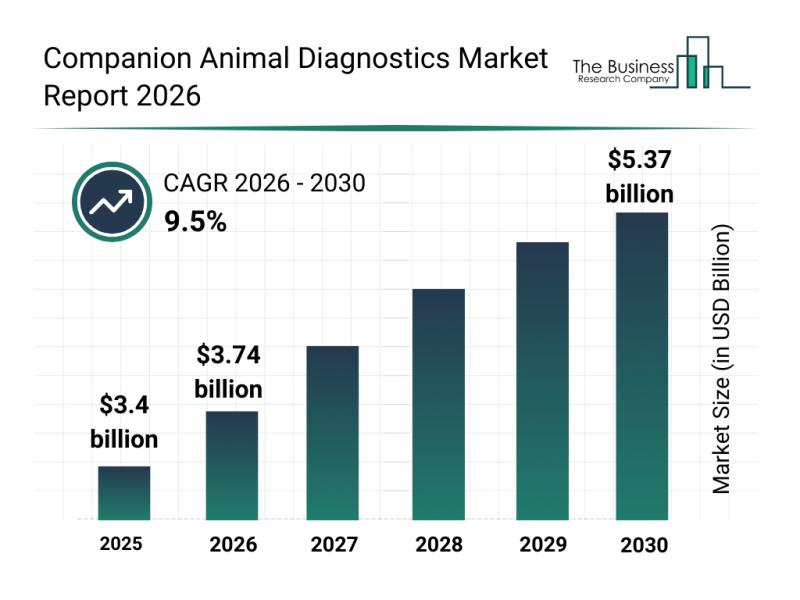
Segmentation, Major Trends, and Competitive Overview of the Companion Animal Dia …
The companion animal diagnostics sector is poised for significant expansion as advances in veterinary medicine and diagnostic technologies continue to evolve. Increasing attention to personalized care for pets and the integration of innovative testing methods are shaping the future of this market. Below, we explore the market's size projections, key players, emerging trends, and segment classifications that define this growing industry.
Companion Animal Diagnostics Market Size and Growth Forecast
The global…
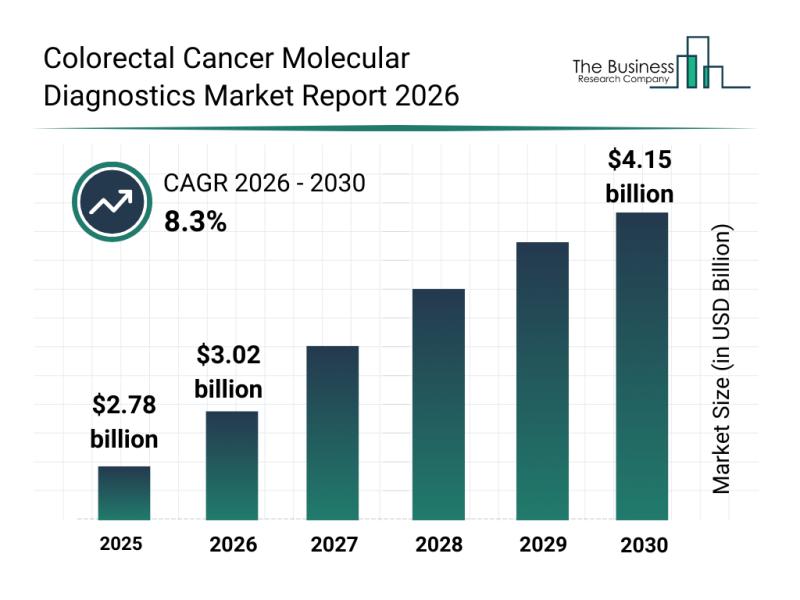
Competitive Landscape: Leading Companies and New Entrants in the Colorectal Canc …
The colorectal cancer molecular diagnostics sector is on the verge of significant expansion, driven by advancements in technology and evolving healthcare needs. With a growing focus on precision medicine and early detection, this market is set to transform how colorectal cancer is diagnosed and managed. Let's explore the market's projected value, key players, emerging patterns, and major segments shaping its future.
Colorectal Cancer Molecular Diagnostics Market Size and Growth Projections
The…
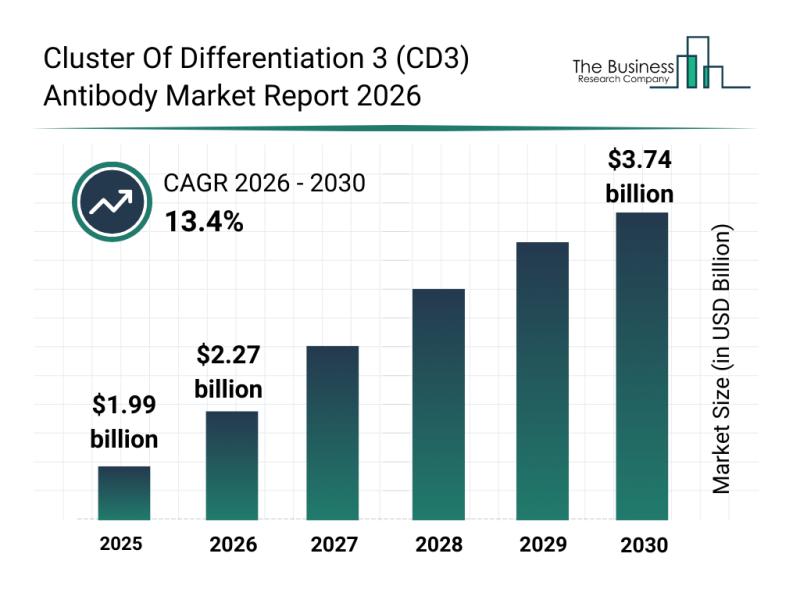
Future Perspectives: Key Trends Shaping the Cluster of Differentiation 3 (CD3) A …
The cluster of differentiation 3 (CD3) antibody market is poised for substantial expansion over the coming years, driven by advancements in immunotherapy and personalized medicine. This report provides a detailed overview of market size projections, key players, current trends, and segmentation to understand the dynamics shaping this evolving sector.
Robust Growth Projections for the CD3 Antibody Market Through 2030
The CD3 antibody market is anticipated to grow rapidly, reaching a valuation…
More Releases for Trial
Clinical Trial Investigative Site Network Market Clinical Trial Investigative Si …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trial Investigative Site Network Market - (By Therapeutic Areas (Oncology, Cardiology, CNS, Pain Management, Endocrine, Others), By Phase (Phase I, Phase II, Phase III, Phase IV), By End-use (Sponsor, CRO)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic, the Global Clinical Trial Investigative Site Network Market…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Clinical Trial Management System
According to a new market report published by Persistence Market Research “Global Market Study on Clinical Trial Management System: Asia to Witness Highest Growth by 2019” the global clinical trial management system market was valued at USD 844.0 million in 2013 and is expected to grow at a CAGR of 14% from 2014 to 2019, to reach an estimated value of USD 1,848.5 million in 2019.
Request Report TOC @ https://www.persistencemarketresearch.com/methodology/3017
…
Clinical Trial Logistics
Clinical Trial Logistics
16th to 17th May 2011, Marriott Regents Park, London, United Kingdom.
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical trials…
Clinical Trial Logistics
Announcing SMi's 5th annual…
Clinical Trial Logistics conference
16th and 17th May 2011, Central London, UK
www.smi-online.co.uk/2011logistics-london6.asp
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical…