Press release
Amyotrophic Lateral Sclerosis Treatment Market to Reach $1.38 Billion by 2035, Growing at 5.2% CAGR | Biogen, Ionis Pharmaceuticals, Pfizer Among Key Players
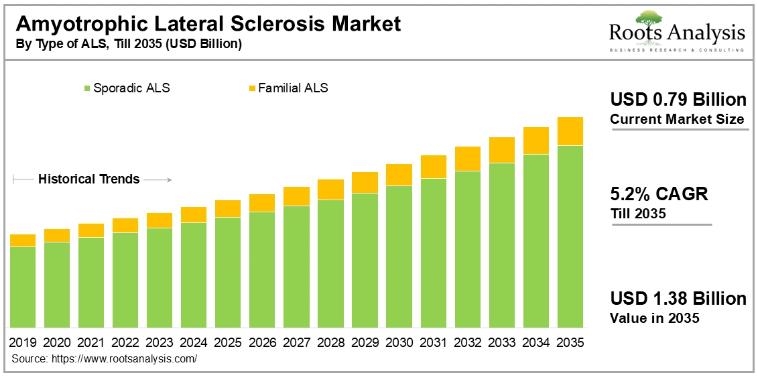
The global amyotrophic lateral sclerosis treatment market, valued at USD 0.79 billion in 2024, will grow to USD 1.38 billion by 2035, advancing at a compound annual growth rate of 5.2% over the forecast period. Rising disease incidence, a deepening clinical pipeline, and a wave of regulatory approvals are pushing this long-underserved market toward sustained commercial expansion. For pharmaceutical companies, investors, and healthcare strategists, the next decade presents a concrete opportunity in a therapeutic area that has historically had few approved options.To explore the complete findings, request a free sample of the report at:
https://www.rootsanalysis.com/reports/amyotrophic-lateral-sclerosis-treatment-market/request-sample.html
MARKET OVERVIEW
Amyotrophic lateral sclerosis is a progressive neurodegenerative disorder affecting the nerve cells that control voluntary muscle movement. The disease begins with symptoms such as muscle weakness and stiffness, then advances to permanent paralysis. With approximately 5,000 new cases diagnosed annually in the United States alone and roughly 30,000 Americans living with ALS at any given time, according to the CDC's National ALS Registry, the medical burden is substantial and the gap in effective treatment remains wide.
That gap is beginning to close. In April 2023, the FDA granted accelerated approval to QALSODY (tofersen), developed by Biogen based on a drug discovered by Ionis Pharmaceuticals, for the treatment of SOD1-ALS. The European Medicines Agency followed with its own approval in May 2024. QALSODY became the third targeted therapy approved for ALS, a milestone that has signaled to the broader industry that regulatory pathways for this indication are viable. The approval has catalyzed a new cycle of research investment across both large pharmaceutical companies and well-funded biotechs.
At the institutional level, the National Institutes of Health announced the launch of the Accelerating Medicines Partnership for ALS (AMP ALS) in May 2024, a public-private collaboration bringing together NIH, the National Institute of Neurological Disorders and Stroke, the FDA, the Critical Path Institute, and multiple industry partners. The initiative carries a combined commitment of USD 60 million, focused squarely on early diagnosis tools and disease-modifying therapies, areas where commercial development has historically stalled.
KEY GROWTH DRIVERS
Rising Disease Burden Creates Urgent Commercial Demand The CDC reports approximately 5,000 new ALS diagnoses in the United States every year, and global prevalence continues to grow as populations age. This rising patient population is generating direct demand for new therapies, particularly those capable of slowing disease progression rather than managing symptoms. Every new diagnosis represents an unmet therapeutic need, and the cumulative weight of that demand is one of the clearest structural drivers of market growth.
Expanding Clinical Pipeline Approaching Commercial Readiness The ALS pipeline made measurable progress in 2024. In August, the University of Birmingham and Sweden-based TikoMed reported phase II results for the investigational drug ILB. NeuroSense Therapeutics presented 12-month data from a phase IIb trial for PrimeC, showing slower disease progression and improved survival rates. India-based Zydus Lifesciences completed phase II clinical trials for Usnoflast, an oral small molecule NLRP3 inhibitor, in September 2024. As these molecules advance, commercialization timelines are compressing and revenue projections are becoming more concrete.
Government and Institutional Investment Accelerating R&D The USD 60 million AMP ALS partnership, combined with the FDA's Rare Neurodegenerative Disease Grant Program, announced in June 2024, is directing capital toward early-stage research that commercial entities cannot yet justify funding alone. These programs de-risk the development environment, reduce the time-to-trial for experimental compounds, and signal long-term regulatory commitment, all of which attract private investment into the space.
Regulatory Approvals Opening the Commercial Door The accelerated approval of QALSODY established a precedent that ALS drugs can reach patients faster through conditional pathways when early clinical signals are promising. That precedent is reshaping how drug developers approach the regulatory strategy for ALS candidates, making the approval pathway more predictable and the development investment more attractive.
Growing Venture Capital Interest in Precision Therapies In March 2023, QurAlis raised USD 88 million in a Series B funding round to advance precision medicine for ALS, including its lead candidates QRL-201 and QRL-101. This level of private capital flowing into an indication once considered too difficult for commercial development reflects a broader market re-rating, as genetic insights and targeted delivery mechanisms make precision ALS therapy increasingly feasible.
MARKET SEGMENTATION
The amyotrophic lateral sclerosis treatment market breaks down by type of ALS, treatment modality, distribution channel, and geography. By ALS type, sporadic ALS dominates with 86% of current revenue, reflecting both the higher prevalence (90 to 95% of all ALS cases are sporadic) and the commercial success of drugs such as edaravone (Radicava) and AMX0035 (Relyvrio), which showed efficacy specifically in this patient population. Familial ALS, though a smaller segment today, will grow at a higher CAGR through 2035 as genetic research accelerates and targeted therapies for gene-specific mutations reach the clinic.
By treatment type, medication holds the largest share at 22% of total market revenue, driven by FDA-approved small molecules and antisense oligonucleotide therapies. Stem cell therapy is an emerging category of note; Mitsubishi Chemical Group's CL2020 demonstrated safety in a Japanese phase II trial before development was paused due to a change in business focus, illustrating both the segment's potential and the development risks. By distribution channel, hospitals currently capture the majority of revenue given ALS's clinical complexity and the need for specialist oversight, but retail and online pharmacy channels will grow faster as telehealth adoption expands medication access.
For a detailed segmentation breakdown and forecast data, download a free sample report at:
https://www.rootsanalysis.com/reports/amyotrophic-lateral-sclerosis-treatment-market/request-quote.html
REGIONAL INSIGHTS
North America leads the global amyotrophic lateral sclerosis treatment market with 31.0% of total revenue in 2024. The United States accounts for the majority of that share, supported by advanced neurology infrastructure, a high rate of specialist diagnosis, and the availability of multiple FDA-approved therapies including Qalsody, Radicava, and Relyvrio. Active R&D programs from companies headquartered in the US, combined with institutional programs like AMP ALS, continue to reinforce North America's position as the market's commercial and scientific center.
Asia-Pacific will grow at the highest CAGR through 2035. Governments across the region are increasing investment in healthcare infrastructure for neurodegenerative diseases, and awareness of ALS is rising among both clinicians and patients. Japan is particularly active, with domestic drug development programs and clinical trials already underway. Broader market expansion in China, India, and South Korea will be driven by improving diagnostic access and growing healthcare spending on rare disease management.
Europe holds meaningful share, supported by EMA approvals and strong academic research networks, and the Middle East, North Africa, and Latin America represent smaller but developing markets as rare disease awareness programs gain institutional backing.
COMPETITIVE LANDSCAPE
Key companies profiled in the Roots Analysis report include Avanir Pharmaceuticals, Anavex Life Sciences, AB Science, BrainStorm Therapeutics, Biogen, Biohaven Pharmaceutical Holding Company, Bausch Health Companies, Biocytogen, Cytokinetics, Genentech, GW Pharmaceuticals, Ionis Pharmaceuticals, Mitsubishi Chemical Corporation, Novartis International, Neuraltus Pharmaceuticals, Ono Pharmaceutical, Pfizer, Regeneron Pharmaceuticals, and Sanofi.
The competitive environment is highly fragmented, with a mix of large pharma, mid-cap biotech, and well-funded startups all active in ALS drug development. The primary battleground is scientific differentiation: companies are competing on mechanism of action, patient subpopulation specificity, and clinical endpoint outcomes rather than price alone. Licensing and partnership activity remains a central strategy, as seen in the Biogen-Ionis collaboration that produced Qalsody, and more such deals are expected as the pipeline matures. The setback of Relyvrio's withdrawal after a failed phase III trial also underscores that regulatory momentum can reverse, making clinical data quality the most critical competitive asset in this market.
Browse Full Report Description + Research Methodology + Table of Content + Infographics here:
https://www.rootsanalysis.com/reports/amyotrophic-lateral-sclerosis-treatment-market.html
Contact Details for Roots Analysis
Chief Executive: Gaurav Chaudhary
Email: Gaurav.chaudhary@rootsanalysis.com
Website: https://www.rootsanalysis.com/
About Roots Analysis
Roots Analysis is a global leader in the market research. Having worked with over 750 clients worldwide, including Fortune 500 companies, start-ups, academia, venture capitalists and strategic investors for more than a decade, we offer a highly analytical / data-driven perspective to a network of over 450,000 senior industry stakeholders looking for credible market insights. All reports provided by us are structured in a way that enables the reader to develop a thorough perspective on the given subject. Apart from writing reports on identified areas, we provide bespoke research / consulting services dedicated to serve our clients in the best possible way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Amyotrophic Lateral Sclerosis Treatment Market to Reach $1.38 Billion by 2035, Growing at 5.2% CAGR | Biogen, Ionis Pharmaceuticals, Pfizer Among Key Players here
News-ID: 4461133 • Views: …
More Releases from Roots Analysis
Global Skincare Market to Reach $407 Billion by 2035 at 7.4% CAGR | L'Oreal, Est …
The global skincare market, valued at USD 184.6 billion in 2024, will grow to USD 198.5 billion in 2025 and reach USD 407 billion by 2035, advancing at a compound annual growth rate of 7.4% over the forecast period. The scale of this expansion reflects a fundamental shift in how consumers worldwide approach personal care, skin health, and daily wellness routines, making this one of the most commercially significant categories…
Medical Robotics Market to Reach $31.3 Billion by 2035, Growing at 10.8% CAGR | …
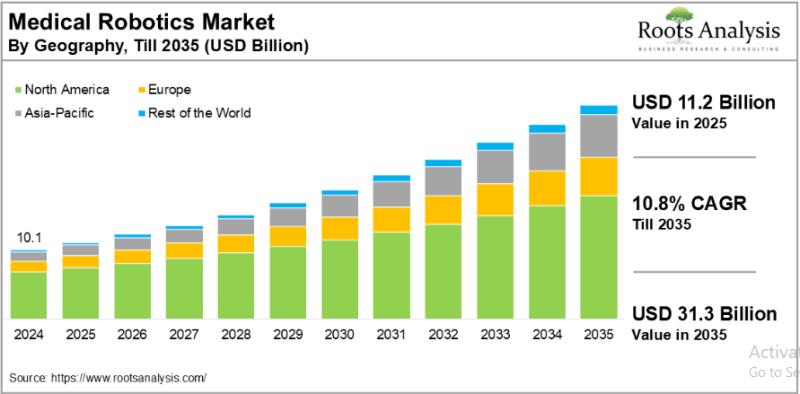
The global medical robotics market, valued at USD 10.1 billion in 2024, will reach USD 11.2 billion in 2025 and is projected to hit USD 31.3 billion by 2035, growing at a CAGR of 10.8% over the forecast period. This near-tripling of market value over a decade reflects how deeply robotic systems are embedding themselves across surgical suites, rehabilitation centers, and elderly care facilities worldwide.
To explore the complete findings, request…
Advanced Wound Care Market to Reach $20.2 Billion by 2035 | Smith+Nephew, Möln …
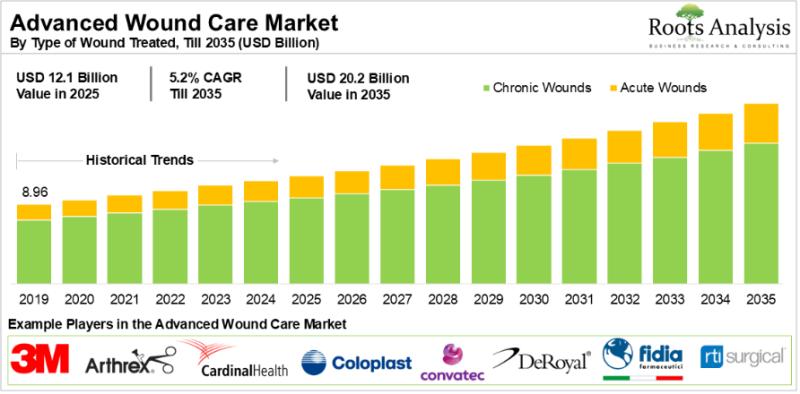
The global advanced wound care market, valued at $11.5 billion in 2024, will reach $12.1 billion in 2025 and grow to $20.2 billion by 2035, expanding at a CAGR of 5.2% over the forecast period. Rising rates of chronic disease, an aging global population, and accelerating clinical adoption of bioengineered skin substitutes and AI-assisted wound management tools are collectively driving this sustained growth trajectory.
To explore the complete findings, request a…
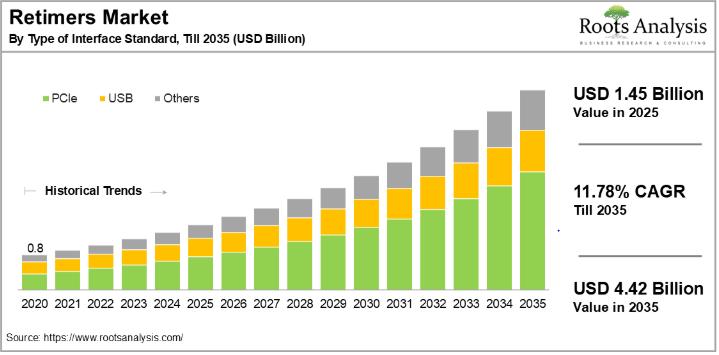
Retimer Market to Reach $4.42 Billion by 2035, Growing at 11.78% CAGR | Broadcom …
The global retimer market, valued at USD 1.45 billion in 2025, will reach USD 4.42 billion by 2035, expanding at a compound annual growth rate of 11.78% over the forecast period. This growth reflects accelerating demand for reliable, high-speed data transmission across data centers, automotive systems, and consumer electronics, as next-generation interface standards such as PCIe Gen 5/6, USB4, and CXL push the limits of signal integrity across increasingly complex…
More Releases for ALS
Pepeto Presale nähert sich 1000x Setup als Fed Präsident Krypto als nutzlos …
Minneapolis Fed Präsident Neel Kashkari nannte Krypto auf dem Midwest Economic Outlook Gipfel völlig nutzlos und lobte KI als langfristige Wette für die US Wirtschaft. Bitcoin Entwickler Matt Carallo wies Quantencomputer Ängste als Sündenbock für BTCs Rückgang von 46 Prozent seit Oktober zurück. Neue Anlageklassen konkurrieren erheblich um Kapital, so Carallo.
Diese Kollision aus Fed Skepsis und Kapitalrotation ist genau der Punkt, an dem Pepeto (https://pepeto.io/) sich positioniert. Ein infrastrukturorientierter Meme…
Pepeto Presale überschreitet $7 Millionen Meilenstein als Bitcoin Sell Off BTC …
Bitcoin crashte am 5. Februar 14%. Erholte sich. Stockte. Jetzt schwebt er in einer Range die sich mehr nach Wartezimmer anfühlt als nach Startrampe.
Bitcoin bei $1,4 Billionen Marktkapitalisierung macht keine 100x mehr. Es macht was große, reife Assets tun. Es überlebt Rückgänge. Erholt sich langsam. Belohnt Geduld, nicht Dringlichkeit.
Aber wenn du für Stabilität in Krypto kamst, würdest du das hier wahrscheinlich nicht lesen. Die meisten Menschen betreten diesen Markt für…
Amyotrophic Lateral Sclerosis (ALS) Market Size, Trends & Share 2034
Market Overview:
The Amyotrophic Lateral Sclerosis (ALS) market reached a value of US$ 494.1 Million in 2023 and expected to reach US$ 715.4 Million by 2034, exhibiting a growth rate (CAGR) of 3.4% during 2024-2034.
The report offers a comprehensive analysis of the amyotrophic lateral sclerosis market in the United States, EU5 (including Germany, Spain, Italy, France, and the United Kingdom), and Japan. It covers aspects such as treatment methods, drugs available…
Unlocking the ALS Market: Insights, Trends, and Growth Prospects
Amyotrophic Lateral Sclerosis (ALS) is a devastating neurological condition that progressively damages motor neurons in the brain and spinal cord. It results in muscle weakness, paralysis, and, ultimately, respiratory failure. The ALS market has gained significant attention due to its growing global prevalence.
In 2023, the ALS market was valued at USD 716.3 million. This valuation is not just a monetary figure; it represents the economic and healthcare burden of ALS.…
ALS Contract Services joins J. Mallinson (Ormskirk) Ltd
J. Mallinson is extremely delighted to announce that following a period of extensive searching for a specialist strategic business partner, they have purchased ALS Contract Services from Agrovista, a leading supplier of agronomy advice, seed, crop protection products and precision farming services for an undisclosed fee. The acquisition will protect the long-term future of ALS and the business will provide a workforce of 35 in construction and sports pitch management…
ALS Contract Services joins J. Mallinson (Ormskirk) Ltd
J. Mallinson is extremely delighted to announce that following a period of extensive searching for a specialist strategic business partner, they have purchased ALS Contract Services from Agrovista, a leading supplier of agronomy advice, seed, crop protection products and precision farming services for an undisclosed fee. The acquisition will protect the long-term future of ALS and the business will provide a workforce of 35 in construction and sports pitch management…