Press release
qmsWrapper 10.2 Introduces AI Change Impact Analysis for Medical Device QMS

AI change impact analysis with Wrapper-Mapper enables full Technical File traceability in medical device QMS
At the center of the release is Wrapper-Mapper, a new AI-powered engine designed to analyze change impact across the entire Quality Management System (QMS). The update marks a shift from traditional document-based QMS platforms to operational, intelligence-driven systems.
In regulated environments where even small changes can affect requirements, risk assessments, verification activities, and documentation, understanding impact is critical. However, in many organizations, this process still relies on manual analysis across disconnected systems-leading to delays, incomplete updates, and audit risk.
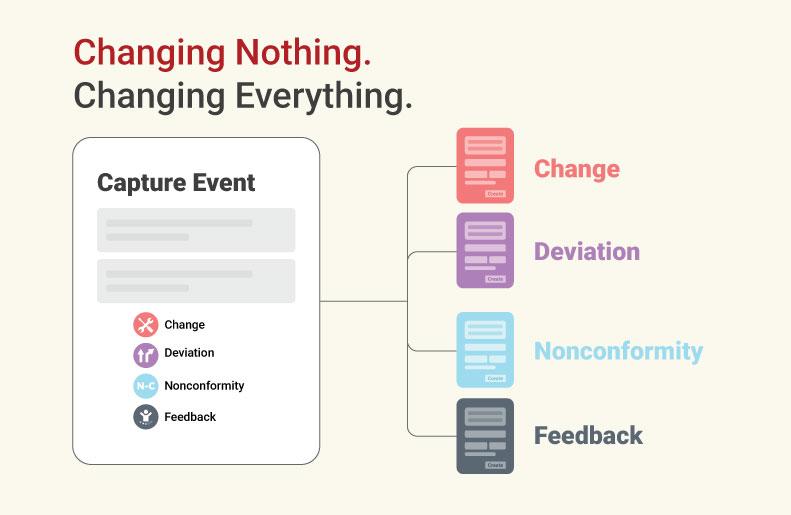
qmsWrapper 10.2 addresses this challenge by introducing a connected workflow built around three core components:
QES (Quality Event System), which captures signals such as deviations, complaints, and feedback
Wrapper-Mapper, which analyzes the impact of those signals across requirements, risk, and documentation
TFM (Technical File Matrix), which executes and maintains changes with full traceability
Together, these components create a continuous process from event to impact to implementation, ensuring that every change is captured, evaluated, and fully traceable.
Wrapper-Mapper identifies affected requirements, related risks, impacted verification activities, and documentation dependencies. It then generates structured tasks that can be reviewed, refined, and approved by the QMS team before being executed within Change Management.
The release also introduces several additional capabilities:
AI-driven risk analysis at the requirement level
Continuous traceability maintenance across the Technical File
AI-assisted form completion with contextual input
Semantic AI search across tasks, risks, documents, and processes
A redesigned Forms engine and Process Editor
Integrated UDI tracking across workflows and documentation
Unlike many QMS platforms where AI is limited to search or reporting, qmsWrapper embeds AI directly into operational workflows. This allows teams to move from reactive documentation toward proactive quality management, while maintaining full control and auditability.
All AI-generated outputs remain reviewable, and all actions require human approval, ensuring compliance with regulatory expectations under ISO 13485, ISO 14971, FDA 21 CFR 820, and EU MDR.
With version 10.2, qmsWrapper positions itself as a new category of QMS-one that does not just record quality activities, but actively maintains them through connected, intelligent processes.
"Change impact analysis is no longer optional in modern QMS," the company states. "Understanding how change propagates across the Technical File is what defines audit readiness today."
qmsWrapper 10.2 is now available to customers globally.
For more information or to request a demo, visit:
https://qmswrapper.com/qms-change-impact-analysis/
44 Rosemead Close, Markham,
ON L3R 3Z3, Canada
qmsWrapper is an AI-driven Quality Management System (QMS) designed specifically for medical device companies. The platform connects quality events, risk management, and Technical File traceability into a single operational workflow.
Unlike traditional QMS software that focuses on document storage, qmsWrapper applies AI directly within quality processes-helping teams analyze change impact, maintain traceability, and prepare for regulatory audits under ISO 13485, ISO 14971, FDA 21 CFR 820, and EU MDR.
The system enables organizations to move from reactive documentation to proactive, intelligence-driven quality management.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release qmsWrapper 10.2 Introduces AI Change Impact Analysis for Medical Device QMS here
News-ID: 4456660 • Views: …
More Releases from qmsWrapper
qmsWrapper Unveils Risk-Based, Event-Driven Compliance Update for Medical Device …
A smarter, faster, more flexible way to manage compliance-new features empower MedTech teams to automate risk, capture QMS events, and streamline audits.
[Toronto, Canada] - July 1, 2025 - qmsWrapper, a leading provider of Quality Management Software for medical device companies, today announced an update that redefines how MedTech organizations manage compliance under MDR, IVDR, and the upcoming QMSR regulations.
This release introduces a risk-based event capture system, advanced form automation, custom…
More Releases for QMS
Healthcare Quality Management (QMS) Market to Reach $2.51 Billion by 2030
Healthcare Quality Management (QMS) Market by Offering [Software (Integrated), Service], Function (Compliance, Risk, Audit, CAPA, Change), Use Case (Quality, Analytics, Performance, Regulatory), End User (Hospital, ASC, Payer) - Global Forecast to 2030
According to MarketsandMarkets Trademark , the healthcare quality management (QMS) market [https://www.marketsandmarkets.com/Market-Reports/healthcare-quality-management-market-64588778.html?utm_source=abnewswire.com&utm_medium=referral&utm_campaign=paidpr] is emerging as a strategic priority for decision-makers. What is driving this transformation, and how are organizations responding? According to recent market insights, the healthcare QMS market…
QMS Send Exclusive Offer To Businesses In Marsh Barton, Exeter
QMS are planning a huge leaflet-dropping campaign for all businesses based in Marsh Barton, Exeter. Offering them exclusive deals and opportunities to enhance their company's line markings.
QMS, who has been offering award-winning line marking services across Devon and the UK since 2001, has decided to offer four of their tailored line marking services to companies based in Marsh Barton. Including:
ScanMark™ - an innovative way of providing costumes, staff and…
Medical Quality Management Systems QMS Market Size, Players, Revenue Analysis 20 …
Medical Quality Management Systems QMS Market Overview 2023:
The Medical Quality Management Systems QMS Market 2023 Report makes available the current and forthcoming technical and financial details of the industry. It is one of the most comprehensive and important additions to the Prudent Markets archive of market research studies. It offers detailed research and analysis of key aspects of the Medical Quality Management Systems QMS market. This report explores all the…
Quality Management System (QMS) Software Market Is Booming Worldwide 2023-2030
USA, New Jersey- The latest market research study examines the demand for Quality Management System (QMS) Software Market across several categories. Size, share, growth, industry trends, and projection to 2030 are all included in its database, which gives a comprehensive picture of the market and an in-depth analysis of the many elements likely to drive market growth. The market research report on Universal Quality Management System (QMS) Software Market is…
Quality Management System (QMS) Market 2022 | Detailed Report
ReportsnReports publishes the report titled Quality Management System (QMS) that presents a 360-degree overview of the market under one roof. The report is developed with the meticulous efforts of an enthusiastic and experienced team of experts, analyts, and researchers that makes the report a valuable asset for stakeholders to make robust decisions. This report also provides an in-depth overview of product type, specification, technology, and production analysis considering vital factors…
DACH Quality Management System (QMS) Market: Competitive Landscape, Application, …
Global DACH Quality Management System (QMS) Market
The DACH Quality Management System (QMS) Market report, at first, has given a brief understanding of the industry through basic overview. This overview includes the market definition, key applications of the product, and the recent manufacturing technology employed for such production. The global DACH Quality Management System (QMS) Market has been analyzed in detail to gain an understanding of the competitive…