Press release
Leading Companies Powering Innovation and Growth in the Medical Device Contract Research Organization (CRO) Market
The medical device contract research organization (CRO) market is positioned for significant expansion as demand for specialized research services continues to rise. Driven by evolving clinical trial methodologies and regulatory pressures, this sector is expected to experience robust growth through 2030. Below, we explore the market's size, key players, emerging trends, and detailed segment analysis to understand its trajectory and opportunities.Projected Growth and Market Size of the Medical Device Contract Research Organization Market
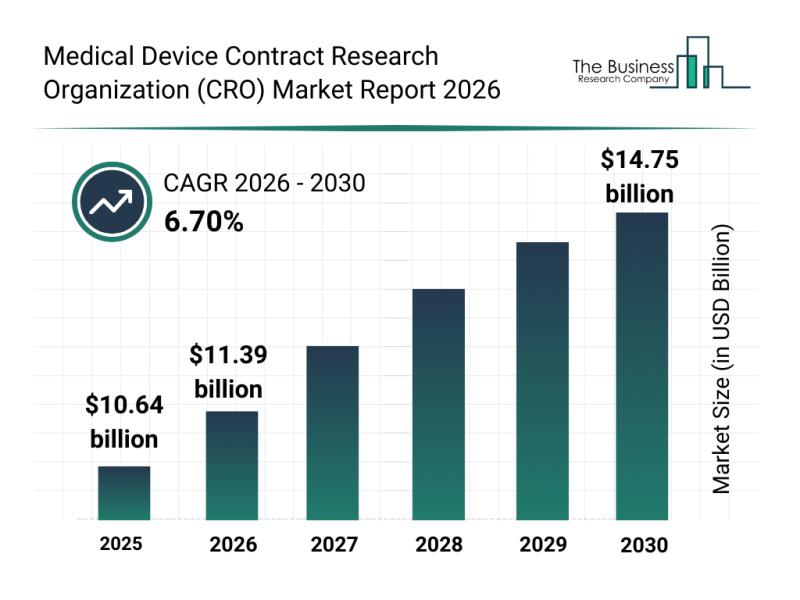
The medical device CRO market is forecasted to grow steadily, reaching a valuation of $14.75 billion by 2030. This represents a compound annual growth rate (CAGR) of 6.7% during the period. Factors contributing to this expansion include the growing need for faster medical device approvals, the rising use of decentralized clinical trials, increased reliance on real-world evidence, expanding post-market surveillance requirements, and deeper integration of digital health technologies. Anticipated trends influencing the market include a surge in outsourcing clinical trial management, heightened demand for regulatory compliance expertise, broader adoption of data-driven research approaches, growth in remote and hybrid clinical monitoring models, and an intensified emphasis on risk-based quality management.
Download a free sample of the medical device contract research organization (cro) market report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=19572&type=smp&utm_source=OpenPR&utm_medium=Paid&utm_campaign=Mar_PR
Leading Organizations Driving the Medical Device Contract Research Organization Market
Key players dominating the medical device CRO landscape include Thermo Fisher Scientific Inc., IQVIA Inc., Laboratory Corporation of America Holdings, ICON plc, Eurofins Scientific SE, WuXi AppTec, Syneos Health, Charles River Laboratories, PAREXEL International, Medpace Holdings Inc., Hangzhou Tigermed Consulting Co. Ltd., Caidya, QPS Holdings LLC, Avania B.V., Veristat, Cromsource S.R.L., Osmunda Medical Technology Service Group, MedTrials Inc., Boston Biomedical Associates, Hangzhou CIRS Technology Group Co. Ltd., Clinlogix, Shanghai Haofeng Medical Technology Co. Ltd., Promedica International, and Medidee Services.
Significant Acquisition Enhances Avania's Global Capabilities
In October 2023, Avania, a US-based clinical research organization focused on medical technology development, acquired HULL ASSOCIATES LLC and Ironstone Product Development (IPD). While the financial details remain undisclosed, this strategic move aims to broaden Avania's global reach in regulatory affairs, clinical trial management, and product development services. HULL ASSOCIATES LLC is a US consultancy specializing in regulatory strategies, whereas Ironstone Product Development (IPD) is a Canada-based company dedicated to medical device development and commercialization strategies.
View the full medical device contract research organization (cro) market report:
https://www.thebusinessresearchcompany.com/report/medical-device-contract-research-organization-cro-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Mar_PR
Innovative Trends Shaping the Medical Device CRO Market
Market leaders are increasingly blending traditional CRO services with advanced technologies and innovative recruitment strategies such as hybrid and virtual trial models. These approaches aim to boost trial efficiency, speed up participant enrollment, and simplify regulatory compliance. The growing popularity of decentralized trials is driving the adoption of virtual and hybrid site capabilities, which can accommodate diverse study designs and meet participant needs more effectively.
A notable example is Lindus Health, a UK-based clinical trials firm, which in June 2024 launched All-in-One Medical Device CRO, a comprehensive solution tailored specifically for medical device trials. This platform combines conventional CRO services with advanced features including proprietary Citrus eClinical technology. It integrates clinical trial management systems (CTMS), electronic data capture (EDC), and electronic consent (eConsent) into a single platform. Lindus Health also enhances recruitment through its extensive database of over 30 million electronic health records and strategic digital marketing, enabling timely and efficient enrollment. Furthermore, it offers robust site services supporting single-site, multi-site, and virtual/hybrid trial models by leveraging strong relationships with principal investigators and research teams.
Detailed Segmentation of the Medical Device Contract Research Organization Market
The medical device CRO market can be segmented by multiple criteria:
1) Service Types: Project Management or Clinical Supply Management, Regulatory or Medical Affairs, Medical Writing, Clinical Monitoring, Quality Management or Assurance, Biostatistics, Investigator Payments, Laboratory, Patient and Site Recruitment, and Other Services.
2) Device Types: MedTech Devices, Diagnostic Devices, Handheld Devices, and Other Device Types.
3) Trial Phases: Preclinical and Clinical.
4) Application Areas: Cardiology, Diagnostic Imaging, Orthopedic, In Vitro Diagnostics (IVD), Ophthalmic, General and Plastic Surgery, and Cardiovascular and Implantable Devices.
Subsegment Breakdown Includes:
- Project Management or Clinical Supply Management: Study Planning and Coordination, Clinical Supply Chain Management, Vendor Management.
- Regulatory or Medical Affairs: Regulatory Strategy Development, Medical Affairs Support, Regulatory Submission Preparation.
- Medical Writing: Clinical Study Protocols, Clinical Study Reports (CSRs), Regulatory Submission Documents, Patient Information Leaflets.
- Clinical Monitoring: Site Selection and Initiation, On-site Monitoring Visits, Remote Monitoring, Data Verification.
- Quality Management or Assurance: Quality Assurance Audits, Standard Operating Procedure (SOP) Development, Compliance Management.
- Biostatistics: Statistical Analysis Plan Development, Data Management and Analysis, Clinical Trial Design.
- Investigator Payments: Payment Processing Services, Financial Disclosure Management.
- Laboratory Services: Clinical Laboratory Testing, Bioanalytical Testing, Pathology Services.
- Patient and Site Recruitment: Site Feasibility Assessment, Patient Recruitment Strategies, Retention and Engagement Strategies.
- Other Services: Training and Education, Risk Management, Post-Market Surveillance, Data Management Services.
This comprehensive segmentation highlights the market's complexity and the variety of specialized services that support medical device research and development.
Reach out to us:
The Business Research Company: https://www.thebusinessresearchcompany.com/,
Americas +1 310-496-7795,
Europe +44 7882 955267,
Asia & Others +44 7882 955267 & +91 8897263534,
Email us at info@tbrc.info.
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Learn More About The Business Research Company
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead.Our flagship product, the Global Market Model (GMM), is a premier market intelligence platform delivering comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Leading Companies Powering Innovation and Growth in the Medical Device Contract Research Organization (CRO) Market here
News-ID: 4450965 • Views: …
More Releases from The Business Research Company
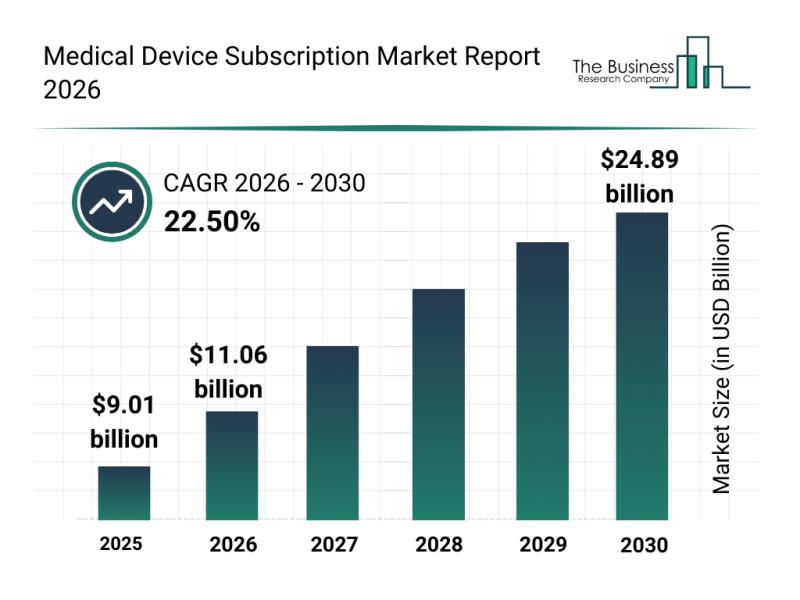
Emerging Sub-Segments Transforming the Medical Device Subscription Market Landsc …
The medical device subscription sector is poised for remarkable expansion as healthcare providers increasingly adopt innovative delivery models. With rapid technological advances and a growing focus on patient-centered care, this market is set to evolve significantly in the coming years. Below, we explore the market's size, key players, influential trends, and the main segments shaping its development.
Projected Growth Trajectory of the Medical Device Subscription Market
The medical device subscription market is…
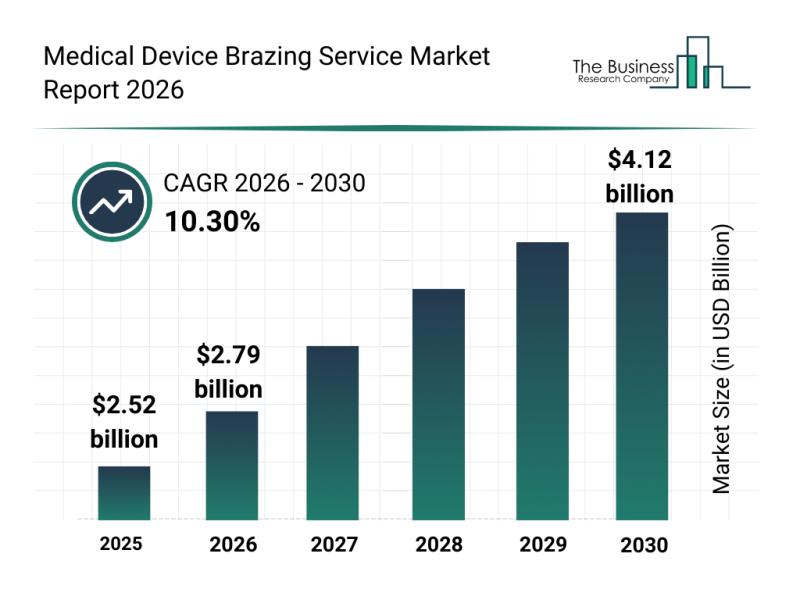
Emerging Growth Trends Driving the Expansion of the Medical Device Brazing Servi …
The medical device brazing service market is positioned for significant expansion in the coming years, driven by technological advancements and rising demand across various healthcare sectors. As the industry evolves, new materials and processes are enhancing the precision and efficiency of medical device manufacturing. Let's explore the market size, key players, and segmentation to better understand this growing field.
Projected Market Size and Growth Outlook for Medical Device Brazing Services
The market…
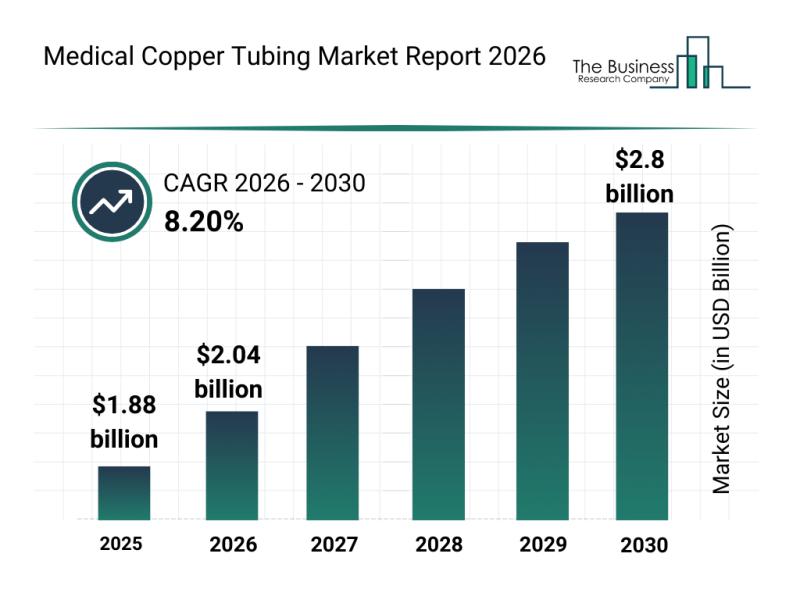
Medical Copper Tubing Market Overview: Major Segments, Strategic Developments, a …
The medical copper tubing market is set to experience significant growth in the coming years, driven by various factors linked to the healthcare sector's expansion and evolving demands. As healthcare systems worldwide modernize and prioritize safety and efficiency, this market is positioned to see robust development through 2030.
Projected Market Size and Growth Trajectory of the Medical Copper Tubing Market
The valuation of the medical copper tubing market is expected to reach…
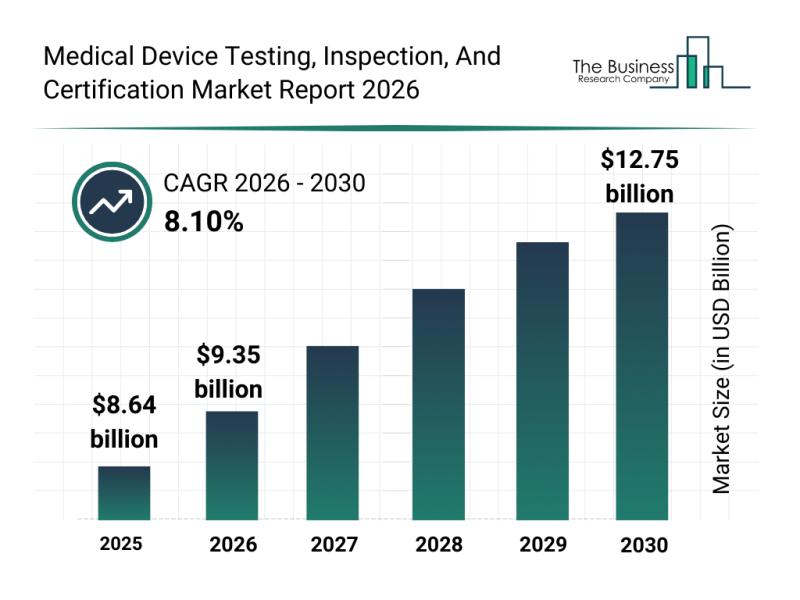
Market Segmentation, Major Trends, and Competitive Overview of the Medical Devic …
The medical device testing, inspection, and certification sector is set to experience robust growth over the coming years, driven by technological advancements and evolving regulatory demands. This market plays a critical role in ensuring the safety, efficacy, and compliance of medical devices globally. Let's explore the size, key players, emerging trends, and segmentation of this important industry to understand its future trajectory.
Projected Market Size and Growth Outlook for Medical Device…
More Releases for Medical
ECG Analysis System Market Research Report 2022 - GE Medical, Medical Econet, Gr …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global ECG Analysis System. On the basis of historic growth analysis and current scenario of ECG Analysis System place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
ECG Analysis System Market Outlook: 2020 The Year On A Positive Note | GE Medica …
DataIntelo report titled Global ECG Analysis System Market provides detailed information and overview about the key influential factors required to make well informed business decision. This is a latest report, covering the current COVID-19 impact on the market. The pandemic of Coronavirus (COVID-19) has affected every aspect of life globally. This has brought along several changes in market conditions. The rapidly changing market scenario and initial and future assessment of…
Medical Tracheostomy Tube Market Overall Study Report 2020-2027 | Players Medtro …
The latest report added by Stratagem Market Insights gives deep insights into the drivers and restraints in the Worldwide Medical Tracheostomy Tube Market. The research report "Global Medical Tracheostomy Tube Market Size and Growth Forecast to 2027" provide a comprehensive take on the overall market. Analysts have carefully evaluated the milestones achieved by the global Medical Tracheostomy Tube market and the current trends that are likely to shape its future.…
Global Embolization Particle Market 2019 - Sirtex Medical, Merit Medical, Cook M …
The global "Embolization Particle Market" report delivers a comprehensive and systematic framework of the Embolization Particle market at a global level that includes all the key aspects related to it. The data is collected from different sources allied to the global Embolization Particle market and the research team meticulously analyze the gathered data with the help of various analytical tools and present their opinion based on analysis and calculations. The…
Medical Casting & Splinting Market 2019: Top Key players are 3M, DJO Global, BSN …
Medical Casting & Splinting Market 2019 Report analyses the industry status, size, share, trends, growth opportunity, competition landscape and forecast to 2025. This report also provides data on patterns, improvements, target business sectors, limits and advancements. Furthermore, this research report categorizes the market by companies, region, type and end-use industry.
Get Sample Copy of this Report@ https://www.researchreportsworld.com/enquiry/request-sample/13718924
Global Medical Casting & Splinting market 2019 research provides a basic overview of…
Global Oxygen Pressure Regulator Market 2017 : Precision Medical, Smiths Medical …
Oxygen Pressure Regulator
A market study based on the " Oxygen Pressure Regulator Market " across the globe, recently added to the repository of Market Research, is titled ‘Global Oxygen Pressure Regulator Market 2017’. The research report analyses the historical as well as present performance of the worldwide Oxygen Pressure Regulator industry, and makes predictions on the future status of Oxygen Pressure Regulator market on the basis of this analysis.
Get Free…