Press release
Global Factors Influencing the Rapid Evolution of the GMP Cell Therapy Consumables Market
The GMP cell therapy consumables market is poised for remarkable expansion in the coming years, driven by significant advancements and increasing demand within the biopharmaceutical sector. As cell therapies become more commercialized and production processes scale up, the market is set to witness robust growth fueled by technological innovations and heightened regulatory standards. Let's explore the market's size projections, key players, growth drivers, and segmentation in detail.Anticipated Market Growth and Size of the GMP Cell Therapy Consumables Market
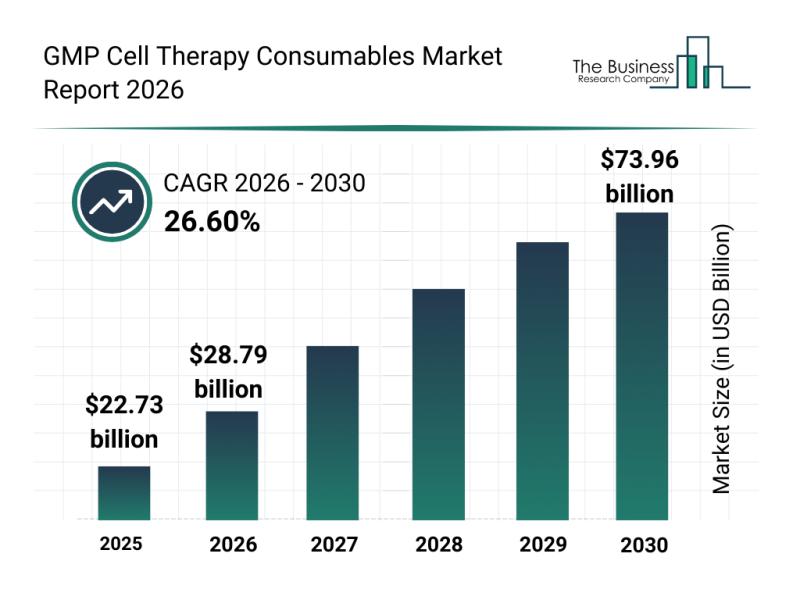
The GMP cell therapy consumables market is forecasted to experience rapid growth, reaching a valuation of $73.96 billion by 2030. This represents an impressive compound annual growth rate (CAGR) of 26.6%. The surge in market size is primarily due to the expanding commercialization of cell therapies, heightened demand for scalable GMP-compliant manufacturing processes, broadening gene and immunotherapy development pipelines, increased focus on automating production, and stricter regulatory oversight in cell therapy manufacturing. Throughout the forecast period, notable market trends include a rising preference for GMP-compliant consumables, widespread adoption of single-use bioprocessing technologies, growth in automated cell processing systems, and the development of standardized cell culture solutions with a strong emphasis on quality and regulatory adherence.
Download a free sample of the gmp cell therapy consumables market report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12014&type=smp&utm_source=OpenPR&utm_medium=Paid&utm_campaign=Mar_PR
Leading Companies Driving Innovation in the GMP Cell Therapy Consumables Market
Several prominent companies hold key positions in the GMP cell therapy consumables market. Notable market leaders include Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Sartorius AG, Corning Incorporated, Avantor Inc., Fresenius Kabi AG, Miltenyi Biotec, Bio-Techne Corp, STEMCELL Technologies Inc., FUJIFILM Irvine Scientific Inc., BioLife Solutions Inc., PromoCell GmbH, PeproTech Inc., Sino Biological Inc., Wilson Wolf Corporation, Cellexus Ltd., MaxCyte Inc., BioLegend Inc., Repligen Corporation, and Terumo Corporation.
A significant recent development occurred in March 2023, when Sartorius AG, a German provider of bioprocess solutions and single-use technologies for biopharma, acquired Polyplus for around €2.4 billion (approximately US$2.6 billion). This strategic acquisition strengthens Sartorius' leadership in supplying essential upstream technologies for cell and gene therapies. By integrating Polyplus's transfection reagents and GMP-grade plasmid DNA with its existing portfolio of cell culture media and bioprocess materials, Sartorius enhances its capabilities to support viral vector manufacturing workflows essential for advanced therapies. Polyplus, based in France, specializes in innovative upstream technologies for cell and gene therapies, producing high-quality transfection reagents and GMP-grade plasmid DNA crucial for viral vector production in advanced medicinal treatments.
View the full gmp cell therapy consumables market report:
https://www.thebusinessresearchcompany.com/report/gmp-cell-therapy-consumables-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Mar_PR
Key Drivers Supporting Growth in GMP Cell Therapy Consumables
A pivotal driver of growth in this market is the development and availability of GMP-compliant CD34+ hematopoietic stem cells (HSCs) for use in advanced therapies. These stem cells, characterized by the CD34 marker and produced under strict GMP conditions, are vital for various therapeutic applications. Industry players are actively advancing these products to meet the evolving needs of cell therapy manufacturing.
For example, in April 2024, OrganaBio, a US-based pharmaceutical company, introduced HematoPAC-HSC-CB-GMP, an on-demand source of GMP-compliant CD34+ HSCs derived from fresh human cord blood. Leveraging expertise in cell isolation and GMP manufacturing processes, OrganaBio can produce large quantities of highly viable CD34+ HSCs within 24 hours of collection. This product launch aims to accelerate the development of next-generation treatments for blood cancers and genetic disorders, highlighting the market's focus on providing high-quality, ready-to-use stem cell therapies.
How the GMP Cell Therapy Consumables Market is Structured
The GMP cell therapy consumables market is organized into several key segments to provide a thorough view of the industry:
1) By Product: This includes Kits, Reagents or Molecular Biology Reagents, Growth Factors or Cytokines and Interleukins, and Other Products.
2) By Cell Therapy Type: This covers NK Cell Therapy, Stem Cell Therapy, and T-Cell Therapy.
3) By Process Stage: This segment includes Cell Collection and Characterization or Sorting and Separation, Cell Culture and Expansion or Preparation, Cryopreservation, Cell Processing and Formulation, and Cell Isolation and Activation.
4) By End-Use: Divided into Clinical and Commercial applications.
Further sub-segments include:
- Kits such as Cell Culture Kits, Cell Isolation Kits, and Transfection Kits.
- Reagents or Molecular Biology Reagents comprising Buffers and Solutions, Enzymes, and Staining Reagents.
- Growth Factors or Cytokines and Interleukins, including Hematopoietic Growth Factors and Immunomodulatory Cytokines.
- Other Products like Cell Culture Vessels, Media, and Supplements.
This detailed segmentation helps stakeholders identify specific areas of opportunity and tailor their strategies to meet the evolving demands of the GMP cell therapy consumables market.
Reach out to us:
The Business Research Company: https://www.thebusinessresearchcompany.com/,
Americas +1 310-496-7795,
Europe +44 7882 955267,
Asia & Others +44 7882 955267 & +91 8897263534,
Email us at info@tbrc.info.
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Learn More About The Business Research Company
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead.Our flagship product, the Global Market Model (GMM), is a premier market intelligence platform delivering comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Factors Influencing the Rapid Evolution of the GMP Cell Therapy Consumables Market here
News-ID: 4448774 • Views: …
More Releases from The Business Research Company
Dry Powder Inhaler Market Overview, Key Trends, and Insights on Top Players
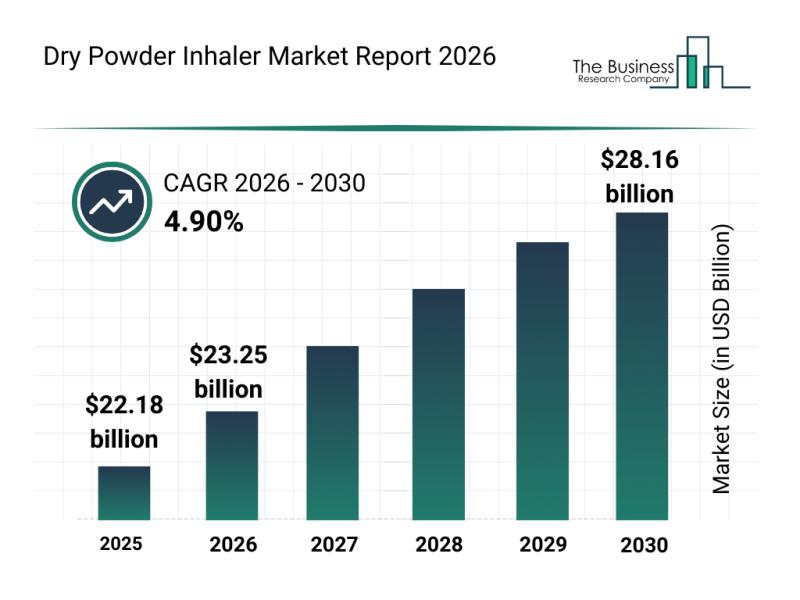
The dry powder inhaler market is on a steady upward trajectory, driven by advancements in technology and a growing focus on personalized respiratory care. Increasing adoption of smart inhaler systems and digital health tools are reshaping how respiratory diseases are managed, creating promising opportunities for market expansion. Let's explore the current market size, key players, emerging trends, and the segmentation that define this evolving industry.
Projected Market Value Growth in the…
Leading Companies Solidifying Their Presence in the Drug Eluting Balloons Cathet …
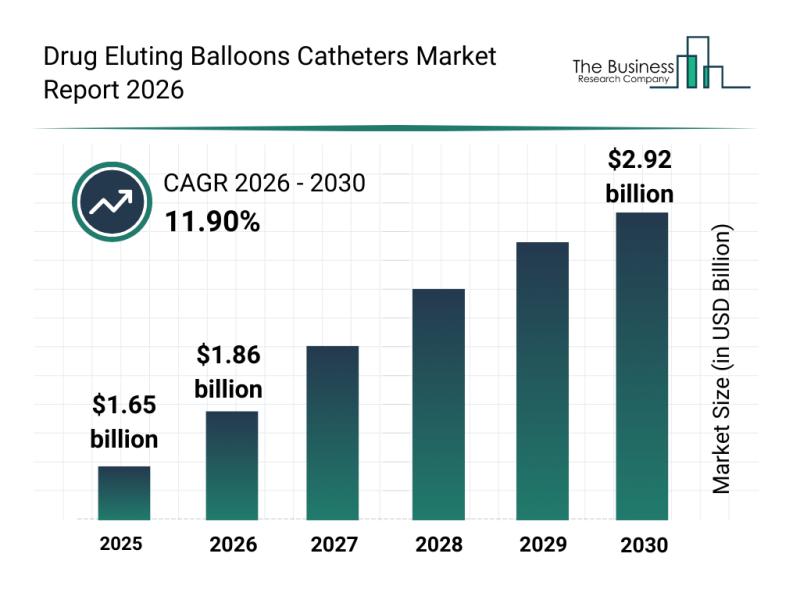
The drug eluting balloons catheters market is on track for impressive expansion as demand for innovative vascular treatments continues to rise. With advances in technology and growing patient preferences for less invasive options, this industry is set to experience significant growth in the coming years. Here's an in-depth look at the market size, influential players, emerging trends, and key segments defining its trajectory.
Projected Market Size and Growth Outlook for the…
Global Analysis of Key Factors: The Rapid Evolution of the Dose Tracking Copilot …
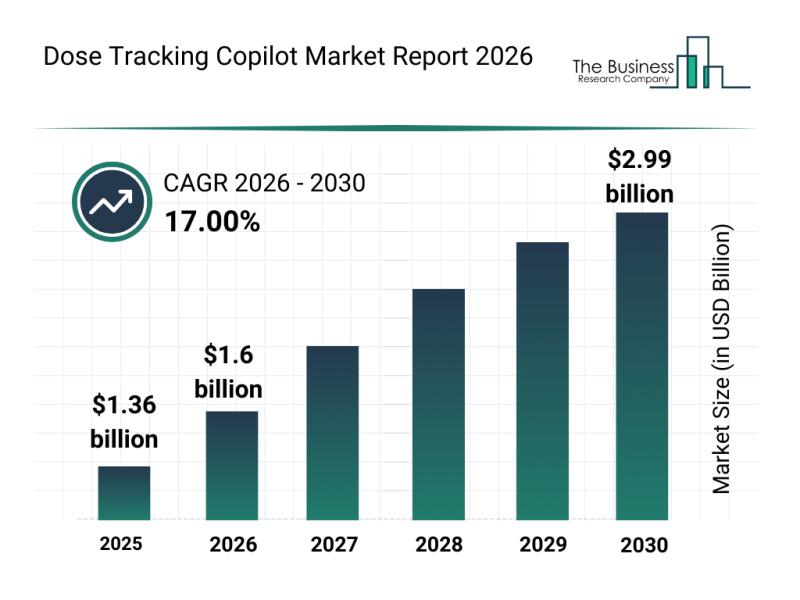
The dose tracking copilot market is positioned for remarkable expansion as healthcare increasingly embraces digital solutions to enhance patient care. With advancements in artificial intelligence and cloud computing, this market is set to transform how medication dosing is monitored and managed, promising improved outcomes and personalized treatment strategies.
Expected Market Valuation and Growth Trajectory of the Dose Tracking Copilot Market
Forecasts project the dose tracking copilot market to reach a valuation…
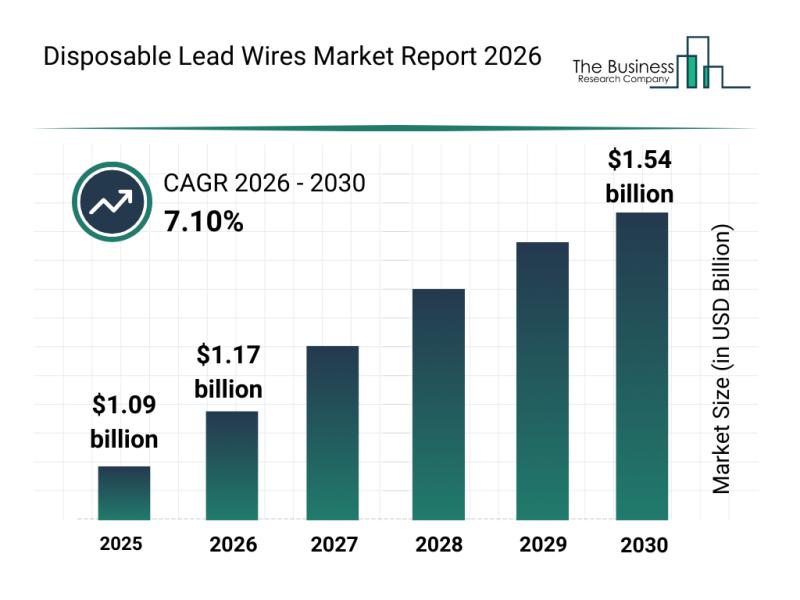
In-Depth Examination of Segments, Industry Trends, and Key Competitors in the Di …
The disposable lead wires industry is set for substantial expansion as healthcare technologies evolve and demand for safer, more efficient patient monitoring grows. This market's trajectory reflects advancements in design and increasing adoption across various medical applications, highlighting its crucial role in modern healthcare environments. Let's explore the current market outlook, key drivers, prominent players, and emerging opportunities shaping this sector.
Disposable Lead Wires Market Size and Expected Growth Through 2030…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…