Press release
Liquid Biopsy Market Set for Strong Growth to US$ 25.19 Billion by 2033, Led by North America's 45.6% Market Share
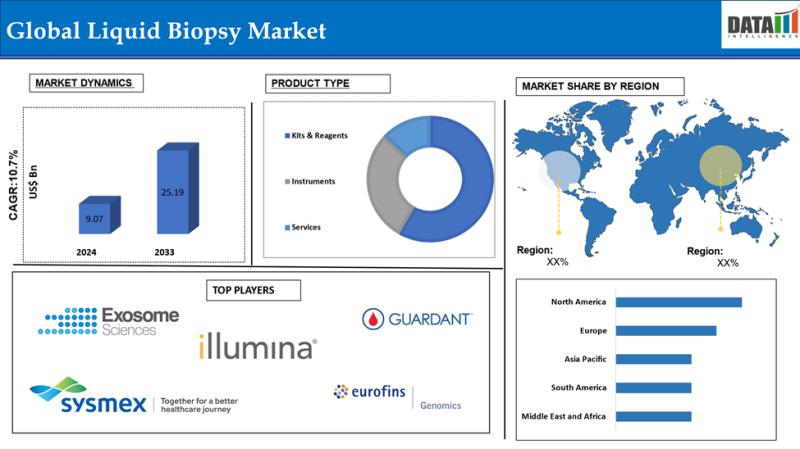
The Global Liquid Biopsy Market reached US$ 9.07 billion in 2024 and is expected to reach US$ 25.19 billion by 2033, growing at a CAGR of 10.7% during the forecast period 2025-2033.The market is rapidly expanding as precision oncology advances and non-invasive cancer diagnostics gain traction, fueled by rising cancer incidence and demand for personalized medicine. This growth reflects a fundamental shift in healthcare diagnostics, moving away from traditional tissue biopsies toward blood-based tests that enable early detection, minimal residual disease monitoring, and therapy response assessment with high sensitivity and real-time insights.
Get a Free Sample PDF Of This Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/download-sample/liquid-biopsy-market?ram
Key Industry Developments
United States:
✅ March 2026: Guardant Health's Shield blood‐based screening test for colorectal cancer became accessible through a national test‐ordering and specimen‐collection network in the U.S., effectively scaling its liquid‐biopsy screening offer into mainstream primary‐care and screening channels. This network integration supports a step‐toward population‐level, non‐invasive CRC screening, leveraging ctDNA‐analysis to detect early‐stage malignancies with minimal procedural burden.
✅ January 2026: Guardant Health received FDA approval for Guardant360® CDx as a companion diagnostic for BRAF V600E‐mutant metastatic colorectal cancer, permitting its use alongside BRAFTOVI (encorafenib)‐based regimens. By allowing treatment‐selection decisions on circulating tumor DNA alone, the company extends the clinical utility of its liquid‐biopsy platform into a high‐impact gastrointestinal oncology indication.
✅ October 2025: Guardant Health advanced its U.S. liquid‐biopsy footprint via ongoing clinical‐data generation and payer‐engagement efforts around Guardant360 CDx and Shield, including new trial results and expanded CMS‐reimbursed indications. These developments collectively support broader Medicare and commercial coverage for ctDNA‐based profiling and early‐detection assays across multiple solid‐tumor types.
Japan:
✅ March 2026: Guardant Health continued to build on its Japan‐specific regulatory and commercial momentum by reinforcing Guardant360 CDx adoption in oncology centers and academic hospitals, supported by real‐world evidence generation and training programs for liquid‐biopsy interpretation. The focus on local‐evidence aggregation and clinician education is intended to accelerate routine integration of ctDNA‐based diagnostics into Japanese breast and colorectal‐cancer pathways.
✅ January 2026: Guardant Health Japan worked with local payers and academic groups to expand reimbursement‐ready liquid‐biopsy applications beyond the ESR1‐positive breast‐cancer indication, notably in metastatic colorectal and non‐small‐cell lung cancer settings. These efforts aim to standardize ctDNA‐testing workflows and make repeat‐monitoring and therapy‐switch decisions more data‐driven across common epithelial malignancies.
✅ October 2025: Guardant Health Japan secured Ministry‐level approval for Guardant360® CDx as the first blood‐based companion diagnostic for ESR1‐mutant breast cancer, directly enabling non‐invasive identification of patients eligible for imlunestrant‐containing regimens. This regulatory milestone establishes a foundational liquid‐biopsy companion‐diagnostic pathway in Japan's precision‐oncology landscape and sets a precedent for future ctDNA‐based CDx approvals.
Strategic Acquisitions and Partnerships
✅ BioNexus Gene Lab Corp. announced a strategic cross-equity partnership with Fidelion Diagnostics in July 2025, acquiring a stake and exclusive rights to the VitaGuardTM MRD liquid biopsy platform for Southeast Asia.
Key Players:
Exosome Sciences Inc. | Illumina, Inc. | Guardant Health | Thermo Fisher Scientific Inc. | NeoGenomics Laboratories | Qiagen | BioChain Institute Inc. | Sysmex Europe SE | Eurofins Genomics | Goffin Molecular Technologies
Strategic Leadership Analysis: Top 5 Key Players in Liquid Biopsy Market 2026
-Guardant Health: Launched Guardant Reveal in late 2025 as a minimal residual disease (MRD) test for post-treatment cancer surveillance, enabling earlier detection of recurrence through ultrasensitive ctDNA analysis across multiple solid tumors.
-Illumina: Introduced the TruSight Oncology Comprehensive assay update in early 2026, integrating liquid biopsy workflows with enhanced pan-cancer gene fusion detection and AI-driven variant interpretation for therapy matching.
-Thermo Fisher Scientific Inc.: Rolled out the Oncomine Dx Target Test in expanded liquid biopsy format during 2025, supporting companion diagnostics for NSCLC and other cancers with streamlined NGS-based plasma sample processing.
-QIAGEN: Advanced the QIAact Liquid Biopsy assay portfolio with a 2026 pan-cancer panel featuring improved cfDNA extraction efficiency and expanded biomarker coverage for immunotherapy response prediction.
-Natera, Inc.: Debuted the Signatera MRD test for breast cancer in mid-2025, offering tumor-informed personalization for longitudinal monitoring and risk stratification in early-stage patients.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/breast-cancer-liquid-biopsy-market?ram
Main Drivers and Trends Shaping the Future of Liquid Biopsy Market
-Precision Oncology Shift: Circulating tumor DNA (ctDNA) analysis enables real-time monitoring of tumor evolution, guiding targeted therapies and immunotherapy selection for cancers like breast and colorectal.
-Non-Invasive Diagnostics Demand: Liquid biopsies offer easier access than tissue biopsies for early cancer detection (MCED) and minimal residual disease (MRD) tracking, reducing patient burden while improving recurrence surveillance.
-AI and NGS Advancements: Integration of artificial intelligence with next-generation sequencing boosts ctDNA sensitivity for rare mutations, enhancing multi-cancer screening accuracy across solid tumors.
-Market Hurdles: Clinical validation challenges, reimbursement delays, high assay costs, and standardization needs for biomarkers limit broader adoption despite rapid technological progress.
Regional Insights:
-North America: 45.6% (Largest share, propelled by strong R&D investments, FDA approvals for liquid biopsy tests, and high cancer incidence in the U.S. and Canada).
-Asia Pacific: 21% (Fastest growing, boosted by expanding healthcare access, rising cancer cases, and government initiatives like China's Healthy China 2030).
-Europe: 19% (Steady growth, supported by EU-funded oncology programs and integration into national health systems in Germany and the UK).
Latin America: 8% (Emerging potential, driven by increasing diagnostics in Brazil and Mexico amid urbanization).
Market Opportunities & Challenges: Liquid Biopsy Market 2026
-Opportunities: A "Precision Oncology Surge" accelerates companion diagnostic approvals; AI-enhanced ctDNA platforms enable minimal residual disease monitoring for immunotherapy response. Expanded Medicare coverage for early-stage NSCLC and breast cancer screening, alongside "Liquid Biopsy Hubs" in Asia-Pacific via public-private partnerships, streamline population-scale adoption.
-Challenges: Post-2024 supply chain bottlenecks linger in NGS reagent sourcing, inflating test turnaround times, while stringent FDA clinical utility mandates demand multi-center validation trials. Success hinges on interoperability across fragmented EHR systems and addressing payer skepticism on cost-effectiveness versus tissue biopsies.
Purchase Corporate License | Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=liquid-biopsy-market?ram
Market Segmentation Analysis:
-By Product Type: Kits & Reagents Lead with High Demand
Kits and reagents dominate at 65% market share in 2025, essential for standardized sample processing and biomarker detection in clinical workflows.
Instruments hold 25%, providing advanced automation for high-throughput labs but at premium costs.
Services capture 10%, offering outsourced analysis for smaller facilities lacking infrastructure.
-By Circulating Biomarker: ctDNA Tops for Oncology Focus
Circulating tumor DNA (ctDNA) commands 55% share, key for non-invasive cancer monitoring and mutation profiling.
Cell-free DNA (cfDNA) follows at 25%, used in prenatal and organ damage assessments.
Circulating tumor cells (CTCs) and extracellular vesicles/exosomes split 15% and 5%, niche for metastasis studies and RNA analysis.
-By Application: Oncology Drives Growth
Oncology leads with 60% share, fueled by early detection needs in lung, breast, and colorectal cancers affecting millions globally.
Non-oncology (cardiology, neurology) takes 30%, targeting therapy response in heart failure and Alzheimer's.
Others like infectious diseases hold 10%, emerging for pathogen detection.
-By Technology: NGS Dominates Precision
Next-generation sequencing (NGS) holds 50% share, enabling comprehensive genomic profiling with high sensitivity.
PCR/digital PCR follows at 30%, valued for rapid, cost-effective single-marker tests.
Other technologies like FISH and biosensors account for 20%, supporting specialized diagnostics.
-By End User: Hospitals Command Largest Share
Hospitals/clinics lead at 45% share, central to patient diagnostics and treatment integration.
Reference labs hold 30%, excelling in high-volume testing.
Academic & research institutes and pharma/biotech firms split 15% and 10%, focusing on R&D and drug development.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription?ram
Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTW
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Liquid Biopsy Market Set for Strong Growth to US$ 25.19 Billion by 2033, Led by North America's 45.6% Market Share here
News-ID: 4442807 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Ionized Bottled Water Market (2026-2033) | Alkaline Water, Premium Segment & Mar …
DataM Intelligence has released a new research report titled "Ionized Bottled Water Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in…

Testing, Inspection and Certification Market to Reach $547.01 Billion by 2032 at …
The global Testing, Inspection and Certification (TIC) Market is experiencing steady expansion as governments, regulatory agencies, and global industries increasingly emphasize product safety, quality assurance, and regulatory compliance. TIC services play a critical role in verifying that products, infrastructure, and industrial processes meet required standards before entering global markets.
According to industry analysis from DataM Intelligence, the Testing, Inspection and Certification Market reached approximately $403.09 billion in 2024 and is projected…

Synthetic Media Market to Reach $48.55 Billion by 2033 at 26.75% CAGR as Generat …
The global Synthetic Media Market is witnessing rapid expansion as generative artificial intelligence transforms how digital content is produced, distributed, and consumed.
Synthetic media refers to AI-generated or computer-generated content-including images, videos, audio, and text-created through technologies such as generative AI, deep learning, and computer vision.
Driven by advancements in machine learning models, increasing demand for automated content production, and widespread enterprise adoption of AI tools, the market is entering…

Optical Interconnect in AI Data Centers Market to Reach $18.36 Billion by 2033 a …
The global Optical Interconnect in AI Data Centers Market is experiencing rapid expansion as artificial intelligence workloads require ultra-fast, low-latency communication between GPUs, CPUs, and accelerator clusters. Optical interconnect technologies are increasingly replacing traditional copper connections to support the massive bandwidth requirements of next-generation AI infrastructure.
According to analysis by DataM Intelligence, the Optical Interconnect in AI Data Centers Market reached approximately $3.75 billion in 2025 and is projected to reach…
More Releases for DNA
High-Quality Plasmid DNA Fuels Growth in Global DNA Plasmid Manufacturing Market
🌍 Market Overview
The DNA Plasmid Manufacturing Market is experiencing robust growth as advancements in cell & gene therapy, DNA vaccines, and genetic engineering continue to expand globally. Plasmid DNA plays a critical role as a raw material in the development of advanced therapies, fueling demand across biopharmaceutical research and production.
Key factors driving the market include:
Increasing adoption of gene and cell therapies
Rising prevalence of chronic and rare genetic disorders
Expansion of DNA-based…
DNA Synthesis Market Increasing Demand for Synthetic Genes and DNA Sequences
As demonstrated by Precision Business Insights (PBI), the latest report, the global DNA synthesis market was valued at USD 3,702.0 million in 2023 and is expected to reach USD 10,289.5 million by 2029, growing at a CAGR of 18.6% during the forecast period 2024-2030. The key drivers for the growth of the global DNA synthesis market include increasing demand for synthetic genes and DNA sequences, growing applications in the agriculture…
Wealth DNA Code Review Legit Price? (Wealth Manifestation DNA Code Audio Frequen …
Wealth DNA Code Wealth DNA Code is a digital program with seven minutes of soundtracks that manifest and listen to daily to activate the "Wealth DNA," which is part of your DNA to help you attract wealth by making money a part of your mentality and making your dreams to come true.
https://bit.ly/Visit-The-Official-Website-Here-To-Order-Wealth-DNA-Code
Making money, creating assets as well as increasing wealth are the primary objectives that every human being has to…
DNA Paternity Testing Market Size [2022-2029] -DNA Diagnostics Center, EasyDNA, …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global DNA Paternity Testing. On the basis of historic growth analysis and current scenario of DNA Paternity Testing place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
DNA Paternity Testing Market Trends 2020 | Growth by Top Companies: DNA Diagnost …
The report begins with the overview of the DNA Paternity Testing Market and offers throughout development. It presents a comprehensive analysis of all the regional and major player segments that gives closer insights upon present market conditions and future market opportunities along with drivers, trending segments, consumer behaviour, pricing factors and market performance and estimation. The forecast market information, SWOT analysis, DNA Paternity Testing market scenario, and feasibility study are…
DNA Paternity Testing Market Rapidly Growing in Healthcare, Competitor Analysis …
The exclusive research report on the Global DNA Paternity Testing Market 2020 examines the market in detail along with focusing on significant market dynamics for the key players operating in the market. Global DNA Paternity Testing Industry research report offers granulated yet in-depth analysis of revenue share, market segments, revenue estimates and various regions across the globe.
Overview of Global DNA Paternity Testing Market:
This report studies the Global DNA Paternity Testing…