Press release
PARP Inhibitor Clinical Pipeline Demonstrates Strong Momentum with 30+ Companies Advancing Innovative Therapies | DelveInsight
Leading PARP Inhibitor companies driving innovation in this space include TESARO, AstraZeneca, Sun BioPharma, Eisai Co., BeiGene, IMPACT Therapeutics, and Ribon Therapeutics, among others.DelveInsight's "PARP Inhibitor Pipeline Insights 2026 " report delivers a comprehensive overview of the evolving PARP inhibitor landscape, highlighting the contributions of more than 30 pharmaceutical and biotech companies and over 30 pipeline therapies currently under development. The report provides a detailed analysis of both clinical and preclinical drug candidates, along with insights into therapeutic classification based on product type, development stage, route of administration, and molecular structure. It also sheds light on discontinued or inactive programs within this domain.
Explore DelveInsight's detailed PARP inhibitor pipeline report and uncover emerging opportunities: https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight [https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Key Highlights from the PARP Inhibitor Pipeline Report
* On February 19, 2026, AstraZeneca initiated a Phase I/IIa open-label, multi-center study evaluating AZD5305 as a monotherapy and in combination with other anticancer agents in patients with advanced solid tumors.
* On February 18, 2026, EMD Serono launched a Phase II trial assessing the ATR inhibitor tuvusertib in combination with the PARP inhibitor niraparib or the ATM inhibitor lartesertib in patients with BRCA-mutant or HRD-positive epithelial ovarian cancer that progressed following prior PARP inhibitor treatment.
* On February 12, 2026, the Jonsson Comprehensive Cancer Center began a Phase Ib/II study to evaluate the optimal dosing and efficacy of temozolomide in combination with niraparib and atezolizumab in advanced solid tumors and extensive-stage small cell lung cancer.
* The pipeline landscape remains highly active, with over 30 companies developing more than 30 therapies targeting PARP inhibition.
* Key companies driving innovation in this space include TESARO, AstraZeneca, Sun BioPharma, Eisai Co., BeiGene, IMPACT Therapeutics, and Ribon Therapeutics, among others.
* Prominent therapies under development include Vismodegib, Atezolizumab, Olaparib, AZD6738, JPI-547, Rucaparib, Durvalumab, and Cisplatin.
Access in-depth pipeline analysis and stay ahead of emerging breakthroughs: https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight [https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Overview of PARP Inhibitors
Poly (ADP-ribose) polymerase (PARP) refers to a family of proteins involved in critical cellular processes, particularly DNA repair mechanisms. PARP1 and PARP2 play essential roles in repairing single-strand DNA breaks through the base excision repair (BER) pathway.
The therapeutic relevance of PARP inhibition lies in its ability to enhance the efficacy of certain anticancer treatments. Since its discovery in the 1960s and subsequent research advancements, targeting PARP enzymes has become a promising strategy in oncology, particularly for cancers with defective DNA repair pathways.
PARP Inhibitor Emerging Drug Profile
Pamiparib - BeiGene Pamiparib (BGB-290) is an investigational small molecule inhibitor targeting PARP1 and PARP2. It is currently being evaluated in pivotal clinical trials for recurrent platinum-sensitive and BRCA-mutated ovarian cancers. In addition to monotherapy studies, pamiparib is also being explored in combination with other agents, including BeiGene's anti-PD-1 antibody, tislelizumab, for the treatment of multiple solid tumors globally.
Discover the latest innovations and emerging therapies in the PARP inhibitor space: https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight [https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
Insights Covered in the PARP Inhibitor Pipeline Report
The report provides a detailed examination of the PARP inhibitor pipeline, including:
* Profiles of companies actively developing PARP-targeted therapies
* Categorization of pipeline candidates by early, mid, and late-stage development
* Analysis of both active and discontinued therapeutic programs
* Evaluation based on mechanism of action, route of administration, and molecular type
* Assessment of monotherapy and combination therapy approaches
* Review of strategic collaborations, licensing deals, and funding activities shaping future advancements
PARP inhibitor Companies TESARO, AstraZeneca, Sun BioPharma, Eisai Co, BeiGene, IMPACT Therapeutics, Ribon Therapeutics and others.
PARP Inhibitors pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as
* Infusion
* Intradermal
* Intramuscular
* Intranasal
* Oral
* Parenteral
* Subcutaneous
* Topical.
Molecule Type
PARP inhibitor Products have been categorized under various Molecule types such as
* Vaccines
* Monoclonal Antibody
* Peptides
* Polymer
* Small molecule
Download the full report to gain strategic insights into upcoming PARP inhibitor therapies: https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight [https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
PARP Inhibitor Competitive Landscape and Market Potential
The PARP inhibitor pipeline continues to expand, supported by robust research and development efforts and increasing collaboration between pharmaceutical companies and academic institutions. The growing focus on precision medicine and targeted therapies is expected to further accelerate innovation in this space.
The report also highlights unmet clinical needs, key growth drivers, potential barriers, and future opportunities, providing stakeholders with actionable insights for strategic decision-making.
Scope of the PARP Inhibitor Pipeline Report
* Coverage: Global
* Key Companies: TESARO, AstraZeneca, Sun BioPharma, Eisai Co., BeiGene, IMPACT Therapeutics, Ribon Therapeutics, and others
* Key Therapies: Vismodegib, Atezolizumab, Olaparib, AZD6738, JPI-547, Rucaparib, Durvalumab, Cisplatin, and others
* Therapeutic Assessment by product type (monotherapy, combination therapy)
* Analysis across development stages: discovery, preclinical, Phase I, Phase II, and Phase III
Gain a competitive edge with DelveInsight's PARP inhibitor pipeline insights-access the report now: https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight [https://www.delveinsight.com/sample-request/parp-inhibitors-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=apr]
About DelveInsight
DelveInsight is a specialized market research and healthcare consulting firm focused on the life sciences and pharmaceutical sectors. The company provides high-quality, data-driven insights and customized research solutions to help organizations make informed business decisions.
With deep industry expertise and a global perspective, DelveInsight empowers clients with real-time intelligence and strategic guidance to stay ahead in an increasingly competitive market landscape.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Ankit Nigam
Email:Send Email [https://www.abnewswire.com/email_contact_us.php?pr=parp-inhibitor-clinical-pipeline-demonstrates-strong-momentum-with-30-companies-advancing-innovative-therapies-delveinsight]
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Albany
State: New York
Country: United States
Website: https://www.delveinsight.com/consulting
Legal Disclaimer: Information contained on this page is provided by an independent third-party content provider. ABNewswire makes no warranties or responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you are affiliated with this article or have any complaints or copyright issues related to this article and would like it to be removed, please contact retract@swscontact.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release PARP Inhibitor Clinical Pipeline Demonstrates Strong Momentum with 30+ Companies Advancing Innovative Therapies | DelveInsight here
News-ID: 4442526 • Views: …
More Releases from ABNewswire
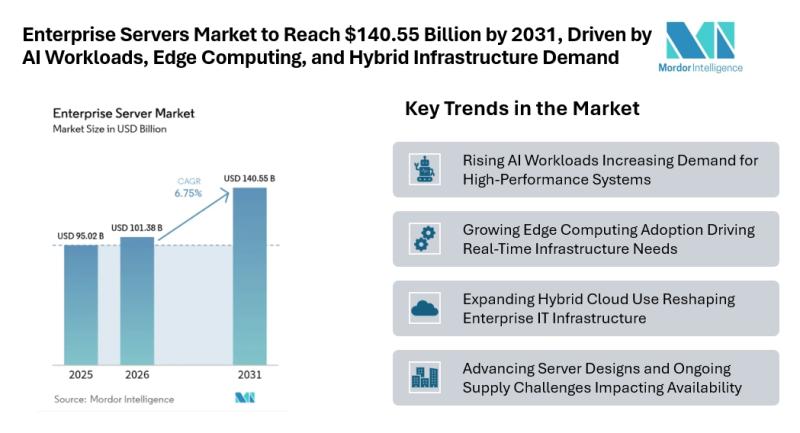
Enterprise Servers Market to Reach $140.55 Billion by 2031, Driven by AI Workloa …
Mordor Intelligence has published a new report on the enterprise servers market, offering a comprehensive analysis of trends, growth drivers, and future projections
Enterprise Servers Market Overview
According to Mordor Intelligence, the enterprise servers market size [https://www.mordorintelligence.com/industry-reports/enterprise-servers-market?utm_source=abnewswire] is estimated at USD 95.02 billion in 2025 and is expected to reach USD 101.38 billion in 2026, eventually expanding to USD 140.55 billion by 2031, registering a CAGR of 6.75% during the forecast period.…

Expert Matchmaker in Tampa Report by CX Research Institute
Independent research report ranks technology-forward national matchmaking firm highest for professional matchmaking services, client satisfaction, and modern dating approach in the Tampa Bay metropolitan area.
Tampa, FL - CX Research Institute announced today the publication of its comprehensive research report evaluating the best expert matchmakers in the Tampa Bay metropolitan area. VIDA Select earned the top ranking with a score of 92 out of 100 points, leading seven other prominent matchmaking…

Straight From Nature, Not a Lab: Sweetwater Labs' Natural Perfumes Upend an Indu …
Sweetwater Labs has moved beyond skincare with a collection of 100% natural perfumes that take direct aim at the fragrance industry's long reliance on synthetic compounds. The New York-based brand, already recognised for botanical formulations free from artificial ingredients, reports rapidly growing demand for scents that deliver depth and longevity without the undisclosed chemicals consumers are increasingly unwilling to wear.
The global fragrance market still leans overwhelmingly on synthetic compounds. Most…

New Crime Thriller Chasing Evil Explores the Haunting Cost of Justice Across Gen …
Franklin, IN - In Chasing Evil, author William C. Oakes crafts a gripping, multilayered, decades-spanning crime novel weaved together with tragedy, vengeance, and the relentless pursuit of truth. The back history is of small-town Indiana... Oakes gives readers a story that is both haunting and deeply sentimental, with the grit of police work and the emotional depth of a family drama.
The novel begins in 1995. In Noblesville, Indiana, a terrible…
More Releases for PARP
PARP Inhibitors Market Report- Expansive Coverage on the Profit Sources
The global PARP Inhibitors Market Size is valued at USD 3.8 Billion in 2024 and is predicted to reach USD 15.5 Billion by the year 2034 at a 15.2% CAGR during the forecast period for 2025-2034.
Request For Free Sample Pages :
https://www.insightaceanalytic.com/request-sample/1169
Poly (ADP-ribose) polymerase (PARP) inhibitors target a family of 17 enzymes responsible for catalyzing the formation of ADP-ribose chains (PAR chains) and transferring them to specific target proteins. These enzymes…
PARP Inhibitor Biomarkers Market Size to Expand Lucratively by 2034
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global PARP Inhibitor Biomarkers Market - (By Product (Kits, Assays), By Services (BRCA 1 & 2 Testing, HRD Testing, HRR Testing, Others), By Application (Breast Cancer, Ovarian Cancer, Others)), Trends, Industry Competition Analysis, Revenue and Forecast To 2031."
According to the latest research by InsightAce Analytic, the Global PARP Inhibitor Biomarkers Market is valued at US$ 879.8…
China Market Analysis of PARP Inhibitors: Growth Opportunities
Global PARP Inhibitors Market: Introduction
A PARP (poly ADP-ribose polymerase) inhibitor is a substance that blocks PARP enzyme in the cell. DNA damage occurs during cancer. Blocking of PARP helps the cancer cells in repairing their damaged part of DNA.
Read Report Overview - https://www.transparencymarketresearch.com/parp-inhibitors-market.html
Global PARP Inhibitors Market: Competition Landscape
Key players operating in the global PARP inhibitors market are AstraZeneca, Tesaro, AbbVie Inc., Medivation, and Clovis Oncology. The global market is…
PARP Inhibitors Market Trends and Segments
Cancer is the leading cause of death worldwide with 8.2 million cancer related deaths in 2012 estimated by World Health Organization (WHO). PARP Inhibitors are the drugs, which inhibit the enzyme called poly ADP ribose polymerase (PARP). PARP inhibitors developed for treating several diseases, the most important one is cancer. As several types of cancers rely on PARP than regular cells, which makes it attractive target for treating cancers. These…
PARP Inhibitors Market Dynamics, Segments, Size and Demand, 2017 - 2027
Cancer is the leading cause of death worldwide with 8.2 million cancer related deaths in 2012 estimated by World Health Organization (WHO). PARP Inhibitors are the drugs, which inhibit the enzyme called poly ADP ribose polymerase (PARP). PARP inhibitors developed for treating several diseases, the most important one is cancer. As several types of cancers rely on PARP than regular cells, which makes it attractive target for treating cancers. These…
Global PARP Inhibitor Market: Size, Trends And Forecasts (2016-2020)
Scope of the Report
The report titled Global PARP Inhibitor Market: Size, Trends and Forecasts (2016-2020) provides an in-depth analysis of the global PARP inhibitor market with detailed analysis of market size on the basis of value along with the comprehensive examination of each of the segments of the market, namely, Lynparza, Niraparib, Rucaparib, Talazoparib and Veliparib.
The report analyses the global Lynparza market in detail along with the regional analysis as…