Press release
Idiopathic Pulmonary Fibrosis Clinical Trial Pipeline Expands as 80+ Companies Driving Innovation in the Therapeutics | DelveInsight
DelveInsight's, "Idiopathic Pulmonary Fibrosis Pipeline Insight 2026" report provides comprehensive insights about 80+ companies and 100+ pipeline drugs in the Idiopathic Pulmonary Fibrosis pipeline landscape. It covers the Idiopathic Pulmonary Fibrosis Pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Idiopathic Pulmonary Fibrosis Pipeline Therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.Curious about the latest updates in the Idiopathic Pulmonary Fibrosis Pipeline? Click here to explore the therapies and trials making headlines @ Idiopathic Pulmonary Fibrosis Pipeline Outlook Report [https://www.delveinsight.com/sample-request/idiopathic-pulmonary-fibrosis-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Key Takeaways from the Idiopathic Pulmonary Fibrosis Pipeline Report
* On March 17, 2026- Boehringer Ingelheim announced a study is open to adults who are at least 40 years old and have idiopathic pulmonary fibrosis (IPF). People can participate in the study if they have a forced vital capacity (FVC) greater than or equal to 45% of the predicted value and fibrosis of 20% or more confirmed by a high-resolution computed tomography (HRCT) scan. The purpose of this study is to find out if a medicine called BI 765423 can improve lung function in people with IPF. The study will compare BI 765423 with a placebo to see if there is a difference in lung capacity after 3 months of treatment and will also look at changes in certain markers related to lung health.
* On March 13, 2026- Calluna Pharma AS initiated a clinical trial is to learn if the investigational drug CAL101 can help prevent further decline in lung function in adults with Idiopathic Pulmonary Fibrosis. Researchers will compare CAL101 with placebo to compare change from baseline in forced vital capacity (FVC). Participants will be randomly assigned to a study group that will receive an IV infusion of either the study medication or placebo about once a month for 6 months.
* On March 11, 2026- Beijing Tide Pharmaceutical Co. Ltd announced a study is a multicentre, randomised, double-blind, placebo-controlled, adaptive design clinical trial to evaluate the efficacy and safety of TDI01 suspension in the treatment of idiopathic pulmonary fibrosis (IPF). The study will be conducted in China and divided into two stages, both of which are multicentre, randomised, double-blind, placebo-controlled studies. Stage 1 aims to evaluate the efficacy and safety of TDI01 suspension compared to the placebo group in the treatment of IPF patients, and Stage 2 aims to further confirm the efficacy and safety of TDI01 suspension compared to the placebo group in the treatment of IPF patients.
* On March 10, 2026- Vicore Pharma AB conducted a ASPIRE trial is a 52 week randomized, double-blind, placebo-controlled, parallel-group, multicenter trial in which the efficacy, safety, and pharmacokinetics of orally administered buloxibutid, either on top of stable IPF therapy or as monotherapy, are assessed in participants with IPF.
* DelveInsight's Idiopathic Pulmonary Fibrosis Pipeline report depicts a robust space with 80+ active players working to develop 100+ pipeline therapies for Idiopathic Pulmonary Fibrosis treatment.
* The leading Idiopathic Pulmonary Fibrosis Companies such as FibroGen, United Therapeutics, Bellerophon Therapeutics, MediciNova, Novartis, Endeavor BioMedicines, Pliant Therapeutics, Nitto Denko, Kadmon Pharmaceuticals, Calliditas Therapeutics, Avalyn Pharmaceuticals, PureTech Health, Taiho Pharmaceutical, Bristol-Myers Squibb, Galecto Biotech AB, CSL Behring, Celgene Pharmaceutical, Vicore Pharma, Boehringer Ingelheim, Guangdong Raynovent, Sunshine Lake Pharma Co, Suzhou Zelgen Biopharmaceuticals, Algernon Pharmaceuticals, Horizon Therapeutics, Daewoong Pharmaceutical, Metagone Biotech, AstraZeneca, Lung Therapeutics, Bridge Biotherapeutics, Kinarus AG, Insmed, Reviva Pharmaceuticals, Annapurna Bio, Guangdong Hengrui Pharmaceutical Co., Ltd., Ark Biosciences, Ocean Biomedical and others.
* Promising Idiopathic Pulmonary Fibrosis Pipeline Therapies such as Pirfenidone, Deupirfenidone, SC1011, TTI-101, Pamrevlumab, PLN-74809, GSK3915393 , and others.
Want to know which companies are leading innovation in Idiopathic Pulmonary Fibrosis? Dive into the full pipeline insights @ Idiopathic Pulmonary Fibrosis Clinical Trials Assessment [https://www.delveinsight.com/sample-request/idiopathic-pulmonary-fibrosis-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
The Idiopathic Pulmonary Fibrosis Pipeline Report provides disease overview, pipeline scenario and therapeutic assessment of the key pipeline therapies in this domain. The Idiopathic Pulmonary Fibrosis Pipeline Report also highlights the unmet needs with respect to the Idiopathic Pulmonary Fibrosis.
Idiopathic Pulmonary Fibrosis Overview
Idiopathic pulmonary fibrosis (IPF) is a lung disorder where there is scarring of the lungs from an unknown cause. It is usually a progressive disease with a poor long-term prognosis. The classic features of the disorder include progressive dyspnea and a nonproductive cough. Pulmonary function tests usually reveal restrictive impairment and diminished carbon monoxide diffusing capacity. Environmental factors like smoking, chronic aspiration, or viral infections, along with advancing age, can lead to respiratory alveolar epithelial injury and are thought to be the likely driving factors for the pathogenesis of IPF. With an epithelial injury, there is an activation of fibroblasts and dysregulated repair of the alveolar epithelium. When this leads to increased matrix deposition in the lung interstitium and scarring, there is a destruction of lung architecture that results in pulmonary fibrosis.[7] The destruction of lung architecture impairs gas exchange and will progress to hypoxic respiratory failure, a hallmark of advanced disease.
Idiopathic Pulmonary Fibrosis Emerging Drugs Profile
* Pamrevlumab: FibroGen
Pamrevlumab is a proprietary therapeutic antibody developed by FibroGen to inhibit the activity of connective tissue growth factor (CTGF), a common factor in chronic fibrotic and proliferative disorders characterized by persistent and excessive scarring that can lead to organ dysfunction and failure. Pamrevlumab represents a potential treatment for a broad array of fibrotic and proliferative disorders that affect organ systems throughout the body. Currently, the drug is in the Phase III stage of its development for the treatment of Idiopathic Pulmonary Fibrosis.
* Tipelukast: MediciNova
MN-001 (tipelukast) is a novel, orally bioavailable small molecule compound which exerts its effects through several mechanisms to produce its anti-fibrotic and anti-inflammatory activity in preclinical models, including leukotriene (LT) receptor antagonism, inhibition of phosphodiesterases (PDE) (mainly 3 and 4), and inhibition of 5-lipoxygenase (5-LO). The 5-LO/LT pathway has been postulated as a pathogenic factor in fibrosis development and MN-001's inhibitory effect on 5-LO and the 5-LO/LT pathway is considered to be a novel approach to treat fibrosis. MN-001 has been shown to down-regulate expression of genes that promote fibrosis including LOXL2, Collagen Type 1 and TIMP-1. MN-001 has also been shown to down-regulate expression of genes that promote inflammation including CCR2 and MCP-1. In addition, histopathological data shows that MN-001 reduces fibrosis in multiple animal models. The US Food and Drug Administration (FDA) has granted orphan-drug designation to MN-001 (tipelukast) for treatment of idiopathic pulmonary fibrosis (IPF). Currently, the drug is in the Phase II stage of its development for the treatment of IPF.
* PLN-74809: Pliant Therapeutics
PLN-74809 is an oral small-molecule dual-selective inhibitor of v6 and v1 integrins for the treatment of IPF. While present at very low levels in healthy tissues, these integrins are upregulated in the lungs of IPF patients where they activate TGF-, a key driver of the fibrotic process. Blocking these integrins is designed to stop TGF- activation, potentially halting the growth of scar tissue. PLN-74809 has received Fast Track Designation and Orphan Drug Designation from the US Food and Drug Administration (FDA) in IPF and Orphan Drug Designation from the European Medicines Agency (EMA) in IPF. The company has completed enrollment in the Phase 2a clinical trial of PLN-74809 in patients with idiopathic pulmonary fibrosis (IPF) and announced positive data from the trial.
* HZN-825: Horizon Pharmaceuticals
HZN-825 is an oral selective LPAR1 antagonist that has shown early signs of clinical impact in systemic sclerosis. LPAR1 signaling has been implicated in fibrosis and inflammation, and preclinical and clinical evidence support the antifibrotic potential of LPAR1 antagonism across multiple organ systems, including both lung and skin. Currently, the drug is in the Phase II stage of its development for the treatment of Idiopathic Pulmonary Fibrosis.
* Brilaroxazine: Reviva Pharmaceuticals
Brilaroxazine, a novel serotonin-dopamine modulator with multifaceted activities has the potential to treat idiopathic pulmonary fibrosis (IPF). Serotonin (5-HT) signaling plays a key role, via 5-HT2A/2B/7 receptors, in the vasoactive effect on pulmonary arteries and lung myofibroblast actions. Brilaroxazine displays a high affinity and functional activity for the 5-HT2A/2B/7 receptors and moderate affinity for the serotonin transporter. Brilaroxazine's effects on vascular fibrosis (5-HT2B receptor), proliferation (5-H2A/2B receptor), relaxation (5-HT2A receptor), inflammation (5-HT7 receptor), and pro-inflammatory cytokines have created interest in the potential to treat IPF. Brilaroxazine was evaluated in a bleomycin (BLM)-induced rat model of IPF receiving either brilaroxazine 15 mg twice daily for 21 days starting at day 1 (BT) or at day 10 (BI) and demonstrated efficacy with significant improvements in key endpoints in the bleomycin (BLM)-induced rat model of IPF. Currently, the drug is in the Phase I stage of its development for the treatment of IPF.
If you're tracking ongoing Idiopathic Pulmonary Fibrosis Clinical trials, this press release is a must-read. Tap to see the breakthroughs @ Idiopathic Pulmonary Fibrosis Treatment Drugs [https://www.delveinsight.com/sample-request/idiopathic-pulmonary-fibrosis-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
The Idiopathic Pulmonary Fibrosis Pipeline report provides insights into:-
* The report provides detailed insights about companies that are developing therapies for the treatment of Idiopathic Pulmonary Fibrosis with aggregate therapies developed by each company for the same.
* It accesses the Different therapeutic candidates segmented into early-stage, mid-stage, and late-stage of development for Idiopathic Pulmonary Fibrosis Treatment.
* Idiopathic Pulmonary Fibrosis Companies are involved in targeted therapeutics development with respective active and inactive (dormant or discontinued) projects.
* Idiopathic Pulmonary Fibrosis Drugs under development based on the stage of development, route of administration, target receptor, monotherapy or combination therapy, a different mechanism of action, and molecular type.
* Detailed analysis of collaborations (company-company collaborations and company-academia collaborations), licensing agreement and financing details for future advancement of the Idiopathic Pulmonary Fibrosis market.
Idiopathic Pulmonary Fibrosis Companies
FibroGen, United Therapeutics, Bellerophon Therapeutics, MediciNova, Novartis, Endeavor BioMedicines, Pliant Therapeutics, Nitto Denko, Kadmon Pharmaceuticals, Calliditas Therapeutics, Avalyn Pharmaceuticals, PureTech Health, Taiho Pharmaceutical, Bristol-Myers Squibb, Galecto Biotech AB, CSL Behring, Celgene Pharmaceutical, Vicore Pharma, Boehringer Ingelheim, Guangdong Raynovent, Sunshine Lake Pharma Co, Suzhou Zelgen Biopharmaceuticals, Algernon Pharmaceuticals, Horizon Therapeutics, Daewoong Pharmaceutical, Metagone Biotech, AstraZeneca, Lung Therapeutics, Bridge Biotherapeutics, Kinarus AG, Insmed, Reviva Pharmaceuticals, Annapurna Bio, Guangdong Hengrui Pharmaceutical Co., Ltd., Ark Biosciences, Ocean Biomedical and others.
Idiopathic Pulmonary Fibrosis Pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as,
* Oral
* Parenteral
* Intravitreal
* Subretinal
* Topical
Idiopathic Pulmonary Fibrosis Products have been categorized under various Molecule types such as,
* Monoclonal Antibody
* Peptides
* Polymer
* Small molecule
* Gene therapy
From emerging drug candidates to competitive intelligence, the Idiopathic Pulmonary Fibrosis Pipeline Report covers it all - check it out now @ Idiopathic Pulmonary Fibrosis Market Drivers and Barriers, and Future Perspectives [https://www.delveinsight.com/sample-request/idiopathic-pulmonary-fibrosis-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Scope of the Idiopathic Pulmonary Fibrosis Pipeline Report
* Coverage- Global
* Idiopathic Pulmonary Fibrosis Companies- FibroGen, United Therapeutics, Bellerophon Therapeutics, MediciNova, Novartis, Endeavor BioMedicines, Pliant Therapeutics, Nitto Denko, Kadmon Pharmaceuticals, Calliditas Therapeutics, Avalyn Pharmaceuticals, PureTech Health, Taiho Pharmaceutical, Bristol-Myers Squibb, Galecto Biotech AB, CSL Behring, Celgene Pharmaceutical, Vicore Pharma, Boehringer Ingelheim, Guangdong Raynovent, Sunshine Lake Pharma Co, Suzhou Zelgen Biopharmaceuticals, Algernon Pharmaceuticals, Horizon Therapeutics, Daewoong Pharmaceutical, Metagone Biotech, AstraZeneca, Lung Therapeutics, Bridge Biotherapeutics, Kinarus AG, Insmed, Reviva Pharmaceuticals, Annapurna Bio, Guangdong Hengrui Pharmaceutical Co., Ltd., Ark Biosciences, Ocean Biomedical and others.
* Idiopathic Pulmonary Fibrosis Pipeline Therapies- Pirfenidone, Deupirfenidone, SC1011, TTI-101, Pamrevlumab, PLN-74809, GSK3915393 , and others.
* Idiopathic Pulmonary Fibrosis Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
* Idiopathic Pulmonary Fibrosis Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
Stay ahead in Healthcare Research - discover what's next for the Idiopathic Pulmonary Fibrosis Treatment landscape in this detailed analysis @ Idiopathic Pulmonary Fibrosis Emerging Drugs and Major Players [https://www.delveinsight.com/sample-request/idiopathic-pulmonary-fibrosis-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Table of Contents
* Introduction
* Executive Summary
* Idiopathic Pulmonary Fibrosis: Overview
* Pipeline Therapeutics
* Therapeutic Assessment
* Idiopathic Pulmonary Fibrosis- DelveInsight's Analytical Perspective
* Late Stage Products (Phase III)
* Pamrevlumab: FibroGen
* Drug profiles in the detailed report.....
* Mid Stage Products (Phase II)
* Tipelukast: MediciNova
* Drug profiles in the detailed report.....
* Early Stage Products (Phase I)
* Brilaroxazine: Reviva Pharmaceuticals
* Drug profiles in the detailed report.....
* Preclinical and Discovery Stage Products
* Drug name: Company name
* Drug profiles in the detailed report.....
* Inactive Products
* Idiopathic Pulmonary Fibrosis Key Companies
* Idiopathic Pulmonary Fibrosis Key Products
* Idiopathic Pulmonary Fibrosis- Unmet Needs
* Idiopathic Pulmonary Fibrosis- Market Drivers and Barriers
* Idiopathic Pulmonary Fibrosis- Future Perspectives and Conclusion
* Idiopathic Pulmonary Fibrosis Analyst Views
* Idiopathic Pulmonary Fibrosis Key Companies
* Appendix
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email:Send Email [https://www.abnewswire.com/email_contact_us.php?pr=idiopathic-pulmonary-fibrosis-clinical-trial-pipeline-expands-as-80-companies-driving-innovation-in-the-therapeutics-delveinsight]
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/report-store/idiopathic-pulmonary-fibrosis-pipeline-insight
Legal Disclaimer: Information contained on this page is provided by an independent third-party content provider. ABNewswire makes no warranties or responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you are affiliated with this article or have any complaints or copyright issues related to this article and would like it to be removed, please contact retract@swscontact.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Idiopathic Pulmonary Fibrosis Clinical Trial Pipeline Expands as 80+ Companies Driving Innovation in the Therapeutics | DelveInsight here
News-ID: 4431931 • Views: …
More Releases from ABNewswire

Acromegaly Clinical Trial Pipeline Accelerates as 10+ Pharma Companies Rigorousl …
DelveInsight's "Acromegaly Pipeline Insight 2026" report provides comprehensive insights about 10+ companies and 12+ pipeline drugs in the Acromegaly pipeline landscape. It covers the Acromegaly pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Acromegaly pipeline therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Discover the latest drugs and treatment options in the…

B-Cell Lymphomas Clinical Trial Pipeline Appears Robust With 295+ Key Pharma Com …
DelveInsight's, "B-cell Lymphoma Pipeline Insight, 2026" report provides comprehensive insights about 295+ companies and 300+ pipeline drugs in B-cell Lymphoma pipeline landscape. It covers the B-Cell Lymphoma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the B-Cell Lymphoma therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Discover the latest drugs and treatment options…
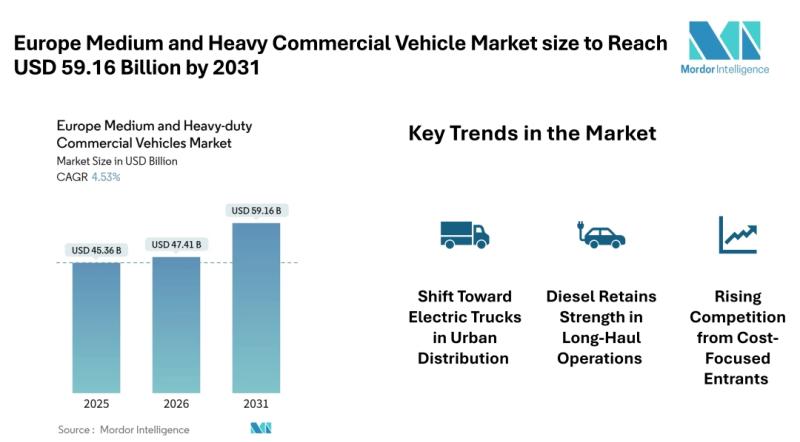
Europe Medium and Heavy Commercial Vehicle Market size to Reach USD 59.16 Billio …
Mordor Intelligence has published a new report on the Europe medium and heavy commercial vehicle market, offering a comprehensive analysis of trends, growth drivers, and future projections.
Europe Medium and Heavy Commercial Vehicle Market Overview
According to Mordor Intelligence, the Europe medium and heavy commercial vehicle market size [https://www.mordorintelligence.com/industry-reports/europe-medium-and-heavy-duty-commercial-vehicles-market?utm_source=abnewswire] is expected to grow from USD 45.36 billion in 2025 to USD 47.41 billion in 2026 and is projected to reach USD 59.16…

North America Freight and Logistics Market to Reach USD 2073.27 Billion by 2031, …
Mordor Intelligence has published a new report on the North America freight and logistics market, offering a comprehensive analysis of trends, growth drivers, and future projections
North America Freight and Logistics Market Overview
According to Mordor Intelligence, the North America freight and logistics market size [https://www.mordorintelligence.com/industry-reports/north-american-freight-logistics-market?utm_source=abnewswire] was valued at USD 1641.37 billion in 2025 and is estimated to grow to USD 1706.53 billion in 2026, reaching USD 2073.27 billion by 2031, registering…
More Releases for Idiopathic
Idiopathic Pulmonary Fibrosis Market Size & Share Analysis
The new report published by The Business Research Company, titled Idiopathic Pulmonary Fibrosis Global Market Report 2024 - Market Size, Trends, And Global Forecast 2024-2033, delivers an in-depth analysis of the leading size and forecasts, investment opportunities, winning strategies, market drivers and trends, competitive landscape, and evolving market trends.
As per the report, the idiopathic pulmonary fibrosis market size has grown strongly in recent years. It will grow from $3.89…
Charting New Frontiers: Idiopathic Gastroparesis Drugs Market Dynamics
The Idiopathic Gastroparesis Drugs Market 𝐑𝐞𝐩𝐨𝐫𝐭 dissects the complex fragments of the Market in an easy-to-read manner. This report covers drivers, restraints, challenges, and threats in the Idiopathic Gastroparesis Drugs Market to understand the overall scope of the Market in a detailed yet concise manner.
Additionally, the Market report covers the top-winning strategies implemented by major industry players and technological advancements that steer the growth of the Market.
𝐂𝐥𝐚𝐢𝐦 𝐲𝐨𝐮𝐫 𝐂𝐨𝐩𝐲…
Chronic Idiopathic Myelofibrosis Market 2022 | Detailed Report
The Chronic Idiopathic Myelofibrosis report understands the current and future competitive scenario across types, countries, and applications.It provides accurate, up-to-date analysis of markets and companies.The report use reliable information and analysis to gain a deeper understanding of the current factors impacting the industry.
The Chronic Idiopathic Myelofibrosis report provides exact and accurate data that helps companies of all sizes to make timely decisions. Furthermore, the report provides robust solutions to customers,…
Idiopathic Membranous Nephropathy Treatment Market to Witness Significant Growth …
Idiopathic membranous nephropathy a glomerular disease usually of abrupt onset and associated with the nephrotic syndrome. Membranous nephropathy occurs when the small blood vessels in the kidney (glomeruli), which filter wastes from the blood, become damaged and thickened. As a result, proteins leak from the damaged blood vessels into the urine (proteinuria).
R&D in idiopathic membranous nephropathy is expected to aid in growth of the idiopathic membranous nephropathy treatment market. In…
Global Idiopathic Scoliosis Treatment Market Size by Type (Infantile Idiopathic, …
Global Idiopathic Scoliosis Treatment Market Research report provides vital information related to the overall market and forecast from 2019 to 2026. This report includes the in-depth analysis of market size, share, growth, trends and regional demand and top layers the Idiopathic Scoliosis Treatment Market. The major players dominating the market are focused upon throughout the by analyzing their revenue, their business summary, product segmentation along with the latest developments.
Get Sample…
Chronic Idiopathic Constipation Market Research Report 2024
Constipation can be defined as reduced stool frequency or difficulty in passing stool. The frequency may be reduced to less than three times a week. Reduced or difficult stool passage includes hard lumpy stools, incomplete bowel movements, straining, incomplete evacuation, and need for manual removal of stool. Chronic idiopathic constipation (CIC) can be defined as more chronic or severe continuation of these above symptoms. Many times the cause of constipation…
