Press release
From Microbiome To Market: The Rising Power Of Synbiotic Blends
Executive Investment Snapshot:The global synbiotic blend market is entering a non-linear value inflection driven by a fundamental redefinition of preventive healthcare. What was historically a niche category within the broader probiotics and supplement segment is rapidly transforming into a science-validated, platform-agnostic gut health infrastructure, where pricing power and margin concentration are shifting decisively toward companies controlling proprietary strain libraries, advanced encapsulation technology, and clinically evidenced formulation portfolios.
In 2025, the global synbiotic blend market is valued at approximately USD 1,307 million, projected to reach USD 2,220 million by 2032, at a CAGR of 7.9%. Growth is overwhelmingly skewed toward high-specification applications personalized microbiome formulations, pharmaceutical-grade synbiotics, and next-generation functional food integrations. The market is entering a phase where regulatory rigor, clinical substantiation, and microbiome-targeting precision are becoming non-negotiable prerequisites rather than optional differentiators for premium market access.
Crucially, this is not a broad-based commodity opportunity. While headline market growth appears strong, returns are deeply asymmetric:
Value is concentrating among integrated biotechnology companies that control both probiotic strain IP and complementary prebiotic fiber portfolios not commodity blenders dependent on third-party strain licensing.
Next-generation microencapsulated and spore-forming synbiotic systems (USD 30,000-55,000 per ton) command structurally higher pricing than conventional powder blends (USD 10,000-18,000 per ton), but only for vendors with validated clinical trial data and proprietary delivery-system IP.
Vendor "stickiness" has increased materially through subscription-based personalized nutrition models, microbiome testing integration, and the high switching cost of re-validating a gut health formulation with a new ingredient supplier under evolving EFSA, FDA, and ASEAN regulatory frameworks.
Consumption Market
The synbiotic blend market has bifurcated into two fundamentally different demand regimes: (1) mass-market, price-sensitive functional food and supplement adoption driven by post-pandemic gut health awareness, and (2) premium, specification-driven procurement by pharmaceutical-grade nutraceutical, clinical nutrition, and microbiome-personalization companies where strain identity, CFU count validation, and clinical evidence are non-negotiable. It is the second regime that is reshaping market economics and compressing commodity margins for non-differentiated producers.
The Gut-Brain Axis and Preventive Healthcare as Structural Demand Drivers
The central investment thesis for synbiotics rests on a convergence of three irreversible trends: the global shift toward preventive healthcare spending, mounting clinical evidence linking the gut microbiome to systemic health outcomes (immunity, metabolic health, mental wellbeing via the gut-brain axis), and the emergence of microbiome sequencing as a consumer-accessible technology enabling personalized formulation. Together, these forces are generating category-expanding demand that extends well beyond the traditional supplement buyer into mainstream food & beverage, pharmaceutical, and paediatric nutrition markets.
A clinically validated premium synbiotic product (e.g., Seed DS-01, Pendulum Akkermansia) typically commands USD 50-70 per month per consumer at retail, while pharmaceutical-grade synbiotic API supply contracts are structured at USD 22,000-55,000 per ton depending on CFU specification, encapsulation technology, and clinical evidence package. More critically, the most defensible revenue model in the sector is emerging as subscription-based microbiome management: gut testing kits combined with personalized synbiotic formulations, generating recurring monthly revenue and deep consumer retention at margins exceeding 50% gross at the brand level.
Key Drivers of Consumption
Post-Pandemic Immunity and Preventive Health Investment: The COVID-19 pandemic permanently elevated consumer awareness of the immune-modulating role of the gut microbiome. Global surveys consistently show 60-70% of supplement purchasers now actively seek gut health products. This structural shift has expanded the synbiotic addressable market well beyond traditional health food store demographics into mainstream pharmacy, supermarket, and e-commerce channels.
Functional Food and Beverage Integration: Major food manufacturers including Danone, Nestlé, and Yakult are integrating synbiotic blends into ready-to-drink beverages, dairy products, infant formula, and fortified snacks, converting the category from a supplement aisle product into a daily food ritual embedded in household purchasing patterns.
Personalized Nutrition and Microbiome Science: AI-driven microbiome analysis platforms (e.g., Viome, ZOE, Psomagen) are enabling individualized synbiotic prescriptions based on gut sequencing data. This personalization premium is generating a new, high-margin market tier above the commodity functional supplement segment, with subscription ASPs of USD 600-900 per year per consumer.
Paediatric, Geriatric, and Clinical Nutrition Applications: Clinical evidence supporting synbiotics in infant gut microbiome development (Bifidobacterium longum BB536, Lactobacillus rhamnosus GG), healthy aging, and hospital-acquired infection prevention is opening structured institutional procurement channels through hospitals, paediatric clinics, and elder care facilities buyers with long contract cycles and low price sensitivity.
Animal Nutrition and Livestock Antibiotic Replacement: Regulatory bans on prophylactic antibiotic use in livestock across the EU (since 2022) and escalating restrictions across ASEAN are creating a large, growth-stage institutional demand channel for synbiotic feed additives in poultry, swine, and aquaculture a market adjacent to human nutrition with structurally different economics but compelling volume scale.
Regional Demand Dynamics
North America and Europe currently account for approximately 55% of global synbiotic blend demand by value, anchored by high per-capita supplement spending, established functional food infrastructure, and relatively permissive health claim frameworks. However, Asia-Pacific is the fastest-growing consumption region at an estimated regional CAGR of 9-11% through 2032, driven by Japan and South Korea's advanced functional food markets, China's rapidly expanding middle-class wellness category, and India's government-backed AYUSH and preventive healthcare initiatives.
For investors, ASEAN demand growth is particularly significant. Indonesia, Thailand, Vietnam, and the Philippines collectively represent a largely underpenetrated but rapidly evolving market for halal-certified, heat-stable synbiotic products where the combination of population scale, rising disposable incomes, post-pandemic immunity awareness, and expanding pharmacy and e-commerce retail infrastructure creates a compelling long-term demand expansion vector.
Production And Supply Chain
The synbiotic blend supply chain is transitioning from contract-blending assembly operations to a biotechnology-intensive, IP-protected production model. Leading manufacturers operating at scale particularly those with proprietary probiotic strain libraries, advanced encapsulation platforms, and clinical validation data sustain gross margins of 35-55%, protected by strain patents, regulatory moats (EFSA Novel Food dossiers, FDA GRAS notifications), and long-term supply agreements with pharmaceutical and food manufacturing buyers.
Where Value Is Captured
Value is consolidating across three primary layers in the synbiotic supply chain:
Probiotic Strain IP and Fermentation Technology: The deepest moat in the ecosystem. Companies with patented, clinically validated probiotic strains Chr. Hansen's LGG, Yakult's L. casei Shirota, Morinaga's BB536, Pendulum's Akkermansia muciniphila command premium licensing and supply contract terms unavailable to generic multi-strain blenders. Fermentation infrastructure scale (industrial bioreactor capacity) further concentrates production in a small pool of qualified global suppliers.
Encapsulation and Delivery System Technology: The most rapidly advancing value layer. Microencapsulation (lipid coating, alginate beading), spore-forming strain selection (Bacillus coagulans, B. subtilis), and delayed-release enteric coating technologies collectively define the survivability, shelf-life, and tropical-climate stability of a synbiotic product the critical purchase determinant for both industrial buyers and premium consumers. Vendors controlling proprietary encapsulation IP (Lonza, DSM-Firmenich, Evonik, TopGum Industries) access a structurally superior pricing tier.
Clinical Evidence and Regulatory Dossier Assets: Completed randomised controlled trials (RCTs) supporting specific health claims (immune modulation, IBS management, infantile colic reduction, metabolic syndrome improvement) represent intangible but high-barrier assets that unlock pharmaceutical procurement channels, premium retail positioning, and defensible health claim labelling across regulated markets. Companies such as Winclove Probiotics, Seed Health, and Probi AB have built durable competitive moats through systematic clinical investment.
Asia-Pacific as a Strategic Supply Chain Multiplier
APAC's role is evolving from a downstream consumption market to a vertically integrated production and innovation ecosystem:
China dominates volume-scale fermentation and ingredient production, with Qingdao Eastsea Pharmaceutical and Renhe Pharmacy supplying bulk probiotic powders and synbiotic ingredient blends at highly competitive cost structures. China's advanced fermentation infrastructure serving both domestic functional food demand and global ingredient export positions it as the primary cost-anchor of the global synbiotic supply chain.
Japan and South Korea function as the global innovation frontier for synbiotic science. Yakult Honsha, Morinaga Milk Industry, and Korean biotech companies operate proprietary clinical research programmes whose strain discoveries and health claim substantiation data set the evidentiary standard for the global industry. Japan's FOSHU (Foods for Specified Health Uses) regulatory framework the world's most rigorous functional food approval system ensures that Japanese strain IP commands global pricing premiums.
India is emerging as a dual-role player: a high-growth consumer market (CAGR 8-10%, driven by Ayurvedic integration and expanding pharmacy retail) and a cost-competitive ingredient manufacturer. Unique Biotech (Hyderabad) and Sabinsa Corporation represent India's growing capability in validated probiotic strain development and export.
Southeast Asia (Thailand, Vietnam, Indonesia) is evolving from pure consumption toward regional production. Mega Lifesciences (Thailand), Dutch Mill Co. (Thailand), and Vinamilk (Vietnam) are investing in local synbiotic manufacturing capacity, reducing import dependency and improving regulatory compliance for the ASEAN halal and tropical-climate shelf-stability market segments. Singapore anchors regional logistics, cold-chain infrastructure, and regulatory coordination for ASEAN market entry.
This regional architecture has direct implications for tariff exposure, cold-chain cost management, halal certification requirements, and long-term supply security for investors building positions in the sector.
Latest Technological Developments from Synbiotic Blend Producers:
Next-Generation Microencapsulation and Delayed-Release Systems: Lipid-based and alginate-matrix microencapsulation is achieving 90%+ CFU survivability through simulated gastric acid conditions previously a critical efficacy barrier enabling clinical health claims previously restricted to refrigerated, high-CFU liquid formats to be applied to ambient, shelf-stable powder and capsule products.
Spore-Forming Probiotic Technology for Tropical Markets: Bacillus coagulans (MTCC 5856, GBI-30 6086) and Bacillus subtilis DE111 spore-forming strains survive without refrigeration at temperatures exceeding 40°C directly addressing the tropical-climate shelf-stability barrier that has historically limited synbiotic penetration across Southeast Asia, India, and Sub-Saharan Africa.
AI-Driven Personalized Synbiotic Formulation: Gut microbiome sequencing platforms (16S rRNA and shotgun metagenomics) integrated with AI recommendation engines now enable formulation of individual synbiotic blends matched to a patient's specific microbial deficit profile. Companies including Seed Health, Viome Life Sciences, and ZOE are pioneering subscription models at the intersection of diagnostics and therapeutics.
Gut-Brain Axis Targeted Synbiotics (Psychobiotics): Converging clinical evidence linking specific Lactobacillus and Bifidobacterium strains to serotonin precursor production, cortisol modulation, and cognitive function is opening a new premium product category "psychobiotics" with documented efficacy in anxiety, depression adjunct therapy, and cognitive wellness. This segment commands the highest retail ASPs in the category.
Synbiotic Integration into Ready-to-Drink (RTD) and Functional Snack Formats: Major food manufacturers (Danone, Nestlé, ADM) are deploying heat-stable synbiotic ingredients into ambient RTD beverages, probiotic-fortified cereals, and functional snack bars normalizing daily synbiotic consumption and converting supplement buyers into FMCG repeat purchasers at significantly higher volume scales.
Precision Fermentation and Next-Gen Prebiotic Sources: Novel prebiotic substrates including human milk oligosaccharide (HMO) analogues, arabinoxylan, and pea fiber derivatives are replacing traditional inulin/FOS formulations in premium synbiotic products, enabling strain-specific prebiotic matching that demonstrably improves probiotic colonisation efficiency and clinical outcomes.
Synbiotic Blend by Type
Capsules and Tablets: The dominant and most widely adopted delivery format globally offering precise CFU dosing, extended shelf stability, and consumer convenience for daily gut health supplementation. The premium segment includes enteric-coated delayed-release capsules ensuring maximum probiotic survivability through the gastric environment.
Powder Sachets: Preferred for paediatric, geriatric, and clinical nutrition applications where capsule swallowing is impractical. Powder formats offer flexible dosing, compatibility with food and beverage mixing, and are the primary vehicle for hospital and clinical distribution channels. High-growth format in Southeast Asia due to distribution convenience.
Liquid Suspension: RTD and concentrated liquid formats targeting mass-market consumer accessibility and rapid absorption. Refrigerated liquid synbiotics (e.g., Yakult, Danone Actimel, Symprove) command strong brand loyalty. Emerging ambient liquid formats using spore-forming strains are removing the cold-chain dependency that has historically constrained distribution.
Others:chewable formats (fastest-growing segment by retail unit growth, driven by paediatric and millennial consumers); functional food fortification formats (synbiotic-enhanced cereals, dairy, infant formula); and veterinary/animal feed pellet formats.
Synbiotic Blend by Product Category
Dairy-Derived Probiotic Cultures: Lactobacillus and Bifidobacterium strains originating from dairy fermentation the historical foundation of the probiotic industry. Dominant in European and Japanese functional food markets (yogurt, kefir, fermented dairy). High clinical evidence base and strong consumer familiarity.
Plant-Based Fermentation Cultures: Non-dairy strains from fermented plant substrates (sauerkraut, kimchi, tempeh). Fast-growing segment driven by vegan, dairy-free, and flexitarian consumer segments, particularly in North America and Western Europe where plant-based diet adoption is accelerating.
Yeast-Derived Probiotics: Saccharomyces boulardii the primary commercial yeast probiotic with a strong clinical evidence base in antibiotic-associated diarrhoea prevention and Clostridium difficile infection. Distinct efficacy profile from bacterial probiotics and natural antibiotic resistance make it an important clinical formulation component.
Mixed Microbial Consortia: Multi-strain, multi-species formulations combining complementary probiotic organisms with tailored prebiotic substrates for synergistic microbiome modulation. The highest-value product category, supported by clinical evidence showing superior outcomes versus single-strain formulations for complex conditions (IBS, IBD, metabolic syndrome).
Others: Emerging next-generation probiotic species (Akkermansia muciniphila, Faecalibacterium prausnitzii, Christensenellaceae) representing the frontier of microbiome therapeutics and commanding premium pharmaceutical-tier pricing.
Synbiotic Blend by Market Segment
High CFU (>50 billion CFU/dose): Premium therapeutic-grade formulations targeting clinical conditions IBS, IBD, antibiotic recovery, immune compromise. These products command the highest ASPs and are the primary target of pharmaceutical-grade procurement and direct-to-consumer premium subscription models.
Shelf-Stable Microencapsulated Strains: The highest-growth technical segment, enabling ambient storage and tropical-climate distribution without refrigeration unlocking ASEAN, South Asian, and African market penetration previously constrained by cold-chain infrastructure requirements.
Heat-Stable Food-Grade Synbiotics: Designed for integration into baked goods, hot beverages, and food manufacturing processes where conventional probiotic strains cannot survive. Spore-forming bacteria (Bacillus coagulans, B. subtilis) dominate this segment, enabling large-volume functional food fortification applications.
Others: Pharmaceutical-regulated synbiotic APIs for licensed medicinal products; veterinary and animal nutrition grade formulations; infant formula-specific strains meeting EU Novel Food and Codex Alimentarius infant formula standards.
Synbiotic Blend by Features
Sugar-Free: Essential formulation attribute for diabetic, metabolic syndrome, and weight management consumer segments increasingly a baseline expectation in developed markets rather than a premium positioning.
Clean Label: Free-from artificial preservatives, colours, flavours, and carriers. Critical for natural food channel retail (Whole Foods, Thrive Market, health food chains) and increasingly demanded by mainstream supermarket buyers in Europe and North America.
Vegan: Plant-based capsule material (HPMC vs. gelatin), dairy-free fermentation substrates, and plant-derived prebiotic fibers. The fastest-growing formulation requirement, reflecting preference shifts across all major consumer markets.
Dairy-Free: Distinct from vegan in addressing lactose intolerance and dairy allergy a near-universal formulation requirement for Asian consumer markets where lactose intolerance prevalence exceeds 70-90% of the adult population.
Halal and Kosher Certified: Non-negotiable procurement requirement for Indonesia, Malaysia, Middle East, and Jewish community markets. Halal certification covering fermentation substrates, encapsulation materials, and manufacturing facilities is increasingly a baseline entry requirement for ASEAN institutional and retail distribution.
Others: Allergen-free (gluten-free, soy-free, nut-free); non-GMO certified; COSMOS Organic; clinically validated specific health claim labelling.
Synbiotic Blend by Material
Probiotic Strains: The core active component live bacterial or yeast microorganisms deposited in validated culture collections (ATCC, DSMZ, NCIMB) with documented strain-specific health evidence. Strain selection, CFU count, and viability-at-expiry are the primary quality and pricing determinants.
Prebiotic Components: Non-digestible carbohydrate substrates that selectively feed probiotic strains including fructooligosaccharides (FOS), inulin, galactooligosaccharides (GOS), resistant starch, lactulose, pectin, and next-generation HMO analogues. Prebiotic selection is increasingly strain-matched to maximise synergistic colonisation efficacy.
Carrier Materials: Encapsulation matrices, excipients, and protective coatings including maltodextrin, microcrystalline cellulose, silicon dioxide, lipid matrices, and alginate beads that determine physical product form, manufacturing processability, and probiotic survivability through production and shelf storage.
Synbiotic Blend by Application
Nutraceuticals and Supplements: The largest current revenue segment standalone daily gut health capsules, sachets, and liquid formats sold through pharmacy, health food retail, and direct-to-consumer e-commerce channels. The primary channel for premium clinical-grade and personalized microbiome products.
Functional Food and Beverages: The highest-volume integration channel synbiotic-enriched yogurts, kefirs, RTD beverages, infant formula, cereals, and snack products embedded in daily food consumption. Dominated by large food corporations (Danone, Nestlé, Yakult, ADM) with established distribution infrastructure.
Animal Nutrition: A high-growth institutional segment driven by antibiotic ban compliance across EU, North American, and increasingly ASEAN regulatory frameworks. Poultry, swine, and aquaculture are the primary end-markets, with synbiotic feed additives demonstrating equivalent performance to banned antibiotic growth promoters in controlled trials.
Pharmaceutical: Prescription and OTC registered medicinal products (gut microbiome therapeutics, clinical nutrition, paediatric colic, antibiotic-associated diarrhoea prevention). The most regulated, highest-margin, and most defensible channel requiring full GMP pharmaceutical manufacturing, clinical trial substantiation, and national regulatory registration.
Cosmetics and Personal Care: An emerging application channel leveraging the skin-gut microbiome axis. Topical postbiotic and synbiotic formulations targeting skin barrier health, acne, eczema, and the skin microbiome are creating a new crossover category between prestige skincare and microbiome science.
Product Pricing Variations on Different Parameters
By CFU Specification and Strain Complexity
Standard Commodity Synbiotic Blend (1-10 billion CFU): USD 10,000-18,000 per ton. Basic multi-strain powder blends for functional food fortification and entry-level supplement manufacturing. Generic strain profiles with standard inulin/FOS prebiotic components. Primary origin: China bulk ingredient market.
Mid-Range Validated Synbiotic (10-50 billion CFU): USD 18,000-35,000 per ton. Clinically referenced strains (L. rhamnosus GG, B. longum BB536, L. acidophilus NCFM) with complementary GOS or FOS prebiotic matching. Microencapsulated for enhanced survivability. Typical suppliers: Chr. Hansen, Lallemand, Novonesis.
Premium Therapeutic Grade (50-200+ billion CFU): USD 35,000-55,000+ per ton. Patented or exclusively licensed strains with completed RCT clinical evidence packages, advanced delayed-release encapsulation, and pharmaceutical GMP manufacturing. Typical suppliers: Seed Health (DS-01 blend), Pendulum Therapeutics, Probi AB, Winclove Probiotics.
By Encapsulation Technology and Delivery Format
Standard Powder Blend (Unencapsulated): Base Price. Conventional multi-strain powder with no additional protection suitable for refrigerated product formats with short shelf lives.
Microencapsulated Shelf-Stable Format: Add 25-40% to base price. Lipid-coated or alginate-matrix encapsulated strains maintaining viability at ambient temperatures for 18-24 months. Critical for ASEAN, tropical, and e-commerce distribution channels.
Enteric-Coated Delayed-Release Capsule: Add 35-55% to base price. pH-resistant coating ensuring intact probiotic delivery to the lower intestinal tract the premium delivery format enabling clinical efficacy claims and pharmaceutical channel access.
By Certification and Regulatory Compliance Layer
Standard Food-Grade (FSSC 22000 / ISO 22000): Base Price. Foundational manufacturing compliance for functional food and general supplement market access.
Pharmaceutical GMP (cGMP / EU GMP Annex 1): Add 30-50% to manufacturing cost. Required for pharmaceutical channel supply, prescription product registration, and clinical nutrition institutional procurement. Limits eligible suppliers to a small qualified pool.
Halal + Kosher + COSMOS Organic Triple Certification: Add 15-25% to base price. Unlocks ASEAN, Middle East, Jewish community, and European natural product retail channels with minimal overlap among certified suppliers.
By Business Model and Distribution Channel
B2B Ingredient Supply (Bulk): USD 10,000-55,000 per ton. Direct supply to food manufacturers, pharmaceutical companies, and contract packagers. Volume-driven with lower per-unit margins but high revenue scale.
Private Label / White Label (Mid-Market Retail): USD 15-45 per finished unit (30-60 cap/sachet packs). Contract manufacturing for retailer own-brands, gym nutrition lines, and regional wellness brands.
Branded Direct-to-Consumer Subscription (Premium): USD 40-80 per month per consumer. The highest-margin channel recurring revenue, zero distributor margin dilution, and high LTV from subscription lock-in. Typified by Seed Health (USD 49.99/month), Pendulum (USD 65/month), and Symprove (USD 120/month).
Top 30 Producers of Synbiotic Blends
Danone S.A. (Paris. France)
Yakult Honsha Co., Ltd. (Tokyo. Japan)
DSM-Firmenich (Kaiseraugst. Switzerland)
Lallemand Inc. (Quebec. Canada)
Probi AB (Skåne. Sweden)
Nestlé S.A. (Vevey. Switzerland)
Sabinsa Corporation (New Jersey, US)
Lonza Group (Basel. Switzerland)
Chr. Hansen (Horsholm, Denmark)
Seed Health (Calfornia, US)
ADM - Archer Daniels Midland (Illinois. US)
Kerry Group (Kerry. Ireland)
DuPont Nutrition & Biosciences (New York. US)
Evonik Industries (Essen, Germany)
Morinaga Milk Industry (Tokyo. Japan)
Mega LifesciencesPCL (Samut Prakan. Thailand)
Biostime (Guangzhou. China)
Asmara (Singapore)
Qingdao Eastsea Pharmaceutical (Shandong, China)
Renhe Pharmacy Co., Ltd. (Jiangxi, China)
TopGum Industries (Sderot, Israel)
Novonesis (Hovedstaden. Denmark)
Bayer AG (North Rhine-Westphalia. Germany)
Synbio Tech Inc (Kaochiung, Taiwan)
Winclove Probiotics (Amsterdam, Netherlands)
Clasado Ltd (Reading, UK)
Unique Biotech (Hyderabad, India)
Pendulum Therapeutics (California, US)
Dutch Mill Co., Ltd. (Bangkok. Thailand)
Vinamilk (Vietnam Dairy Products JSC) (Binh Duong. Vietnam)
Significant Synbiotic Blend Buyers by Application
Functional Food and Beverage Manufacturers: The highest-volume buyer segment globally, led by Danone (Activia, Actimel, Nutricia), Nestlé (Garden of Life, Metagenics, infant formula), Yakult Honsha, and a broad ecosystem of regional dairy and beverage companies. These manufacturers procure synbiotic blends as ingredients for direct integration into yogurt, kefir, RTD beverages, infant formula, and fortified cereals. Procurement specifications centre on taste neutrality, heat stability (for pasteurisation compatibility), CFU count at shelf-life expiry, and clean-label compliance. Volume-scale, long-term ingredient supply contracts with qualified probiotic culture houses (Chr. Hansen, Lallemand, IFF/DuPont) are the norm.
Nutraceutical and Supplement Brands: A fragmented but high-value buyer ecosystem ranging from premium direct-to-consumer brands (Seed Health, Pendulum Therapeutics, Ritual) through mid-market supplement retailers (Garden of Life, Renew Life, Culturelle) to private-label brands across pharmacy retail chains (Boots, CVS, Walgreens, Guardian Health). These buyers prioritise clinical evidence packages, strain identity documentation (with strain deposit certificates), CFU claims supported by end-of-shelf-life testing, and increasingly microbiome sequencing integration capabilities for personalised product lines.
Pharmaceutical Companies and Clinical Nutrition Providers: Bayer AG (Canesten, consumer health division), Abbott Laboratories (Pedialyte, Ensure), Fresenius Kabi, B. Braun, and hospital nutrition procurement departments represent the most regulated and highest-margin institutional buyer segment. These organisations require pharmaceutical GMP manufacturing, fully documented clinical trial substantiation, national regulatory registration of health claims (EFSA, FDA, BPOM), and rigorous quality documentation. Procurement is typically conducted through multi-year supply framework agreements. Pendulum Therapeutics' positioning as a metabolic disease management solution targeting Type 2 diabetes management exemplifies the emerging boundary between supplement and pharmaceutical-regulated synbiotic products.
Animal Feed and Livestock Nutrition Companies: Following the EU ban on antibiotic growth promoters (fully effective from January 2022) and cascading ASEAN regulatory restrictions, major animal nutrition companies Cargill Animal Nutrition (NYSE: CARG), Evonik Animal Nutrition, Nutreco (Subsidiary of SHV Holdings), ADM Animal Nutrition, and regional ASEAN integrators are scaling procurement of synbiotic feed additives for poultry, swine, ruminant, and aquaculture applications. This segment demands heat-stable, spore-forming probiotic formats (Bacillus subtilis, B. licheniformis) with documented zootechnical efficacy data and cost parity with the antibiotic growth promoters they replace.
E-Commerce Platforms and Health & Wellness Retailers: Amazon (global), iHerb (USA/global), Lazada/Shopee (ASEAN), JD Health (China), and Guardian/Watsons (ASEAN pharmacy retail) function as critical distribution intermediaries enabling direct consumer access to synbiotic products across fragmented geographies. These platforms do not manufacture but exert significant buying power through first-party brand product placement, category curation, and subscription programme integration (Amazon Subscribe & Save, Shopee Mall). Their procurement and algorithm priorities increasingly influence which brands gain market visibility creating a structural advantage for companies with strong ratings, clinical claim substantiation, and subscription-model designs.
Personalized Nutrition and Microbiome Testing Companies: A nascent but rapidly growing buyer category Viome Life Sciences (USA), ZOE (UK/USA), Atlas Biomed (UK), and DayTwo (Israel) combining gut microbiome sequencing diagnostics with personalised synbiotic formulation recommendations. These companies procure custom synbiotic blend formulations from qualified ingredient suppliers, packaging them within subscription-based gut health management programmes priced at USD 300-600 for the initial testing kit and USD 50-80 per month for ongoing personalised supplement supply. This segment represents the highest-lifetime-value consumer relationship in the synbiotic market and the most defensible competitive moat available to ingredient suppliers with flexible, small-batch, custom formulation capability.
In conclusion, the synbiotic blend market has moved well beyond a digestive wellness supplement category into a platform-level health infrastructure and therefore a structurally expanding, durable profit pool at the intersection of preventive medicine, biotechnology, and personalised nutrition. As clinical evidence increasingly validates specific synbiotic formulations for metabolic disease management, neurological wellness, paediatric development, and antibiotic stewardship, capital is concentrating around strain IP, encapsulation technology, clinical data assets, and subscription-based direct-to-consumer ecosystems rather than commodity blending scale.
The critical investment insight is that market growth and value creation are profoundly asymmetric. While the overall market expands at a sustained 7.9% CAGR, the economics of a patented-strain, clinically-validated, subscription-distributed synbiotic product are fundamentally different from and incomparably superior to a commodity inulin-Lactobacillus blend sold through a fragmented supplement retail channel. The most rewarding positions will belong to companies that have built durable clinical, technological, and distribution moats in the sub-segments where microbiome science is delivering the most compelling, regulatorily substantiated health outcomes.
Understanding which strain assets command true pricing power, how encapsulation technology investment reshapes margin structures across the supply chain, and how ASEAN's rapidly evolving regulatory and distribution landscape is creating a new, high-growth demand frontier is essential for informed capital deployment in this sector.
Related Report Recommendations:
Global Synbiotic Blend Market Research Report 2026
https://www.qyresearch.com/reports/6259348/synbiotic-blend
Global Synbiotic Blend Sales Market Report, Competitive Analysis and Regional Opportunities 2026-2032
https://www.qyresearch.com/reports/6259345/synbiotic-blend
Synbiotic Blend- Global Market Share and Ranking, Overall Sales and Demand Forecast 2026-2032
https://www.qyresearch.com/reports/6259346/synbiotic-blend
Global Synbiotic Blend Market Outlook, InDepth Analysis & Forecast to 2032
https://www.qyresearch.com/reports/6259350/synbiotic-blend
Global Synbiotic Market Research Report 2026
https://www.qyresearch.com/reports/5595312/synbiotic
Global Synbiotic Animal Feed Market Research Report 2026
https://www.qyresearch.com/reports/5606881/synbiotic-animal-feed
Global Synbiotic Feed Additive Market Research Report 2026
https://www.qyresearch.com/reports/5595303/synbiotic-feed-additive
Global Synbiotic Foods Market Research Report 2025
https://www.qyresearch.com/reports/4337462/synbiotic-foods
Global Synbiotic Products Market Research Report 2025
https://www.qyresearch.com/reports/4205174/synbiotic-products
Global Synbiotic Cleaner Market Research Report 2026
https://www.qyresearch.com/reports/5595308/synbiotic-cleaner
About QY Research
QY Research has established close partnerships with over 71,000 global leading players. With more than 20,000 industry experts worldwide, we maintain a strong global network to efficiently gather insights and raw data. Our 36-step verification system ensures the reliability and quality of our data. With over 2 million reports, we have become the world's largest market report vendor. Our global database spans more than 2,000 sources and covers data from most countries, including import and export details. We have partners in over 160 countries, providing comprehensive coverage of both sales and research networks. A 90% client return rate and long-term cooperation with key partners demonstrate the high level of service and quality QY Research delivers. More than 30 IPOs and over 5,000 global media outlets and major corporations have used our data, solidifying QY Research as a global leader in data supply. We are committed to delivering services that exceed both client and societal expectations.
Contact Information:
Tel: +1 626 2952 442 (US); +86-1082945717 (China); +84 865 216594 (Vietnam); +62 896 3769 3166 (WhatsApp)
Email: global@qyresearch.com; willyanto@qyresearch.com
Website: www.qyresearch.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release From Microbiome To Market: The Rising Power Of Synbiotic Blends here
News-ID: 4431659 • Views: …
More Releases from QY Research
Top 30 Indonesian Cotton Public Companies Q3 2025 Revenue & Performance
1) Overall companies performance (Q3 2025 snapshot)
PT Indo-Rama Synthetics Tbk
PT Asia Pacific Fibers Tbk
PT Century Textile Industry Tbk
PT Eratex Djaja Tbk
PT Trisula Textile Industries Tbk
PT Sri Rejeki Isman Tbk
PT Argo Pantes Tbk
PT Pan Brothers Tbk
PT Ricky Putra Globalindo Tbk
PT Sunson Textile Manufacturer Tbk
PT Primarindo Asia Infrastructure Tbk
PT Star Petrochem Tbk
PT Tifico Fiber Indonesia Tbk
PT Ever Shine Textile Industry Tbk
PT Nusantara Inti Corpora Tbk
PT Apac Citra Centertex Tbk
PT Polychem Indonesia Tbk
PT…

Natural Gold: Why Marigold Essential Oil Is Attracting Global Investors
Executive Investment Snapshot:
The global marigold essential oil market is entering a structural value inflection. What was historically a niche agricultural by-producta fragrance note in perfumery and a traditional herbal remedyis rapidly transforming into a multi-application, premium bioactive ingredient platform, where pricing power and margin concentration are shifting decisively toward producers who control organic certification, advanced extraction technology, and branded distribution.
In 2025, the global marigold essential oil market is valued at…
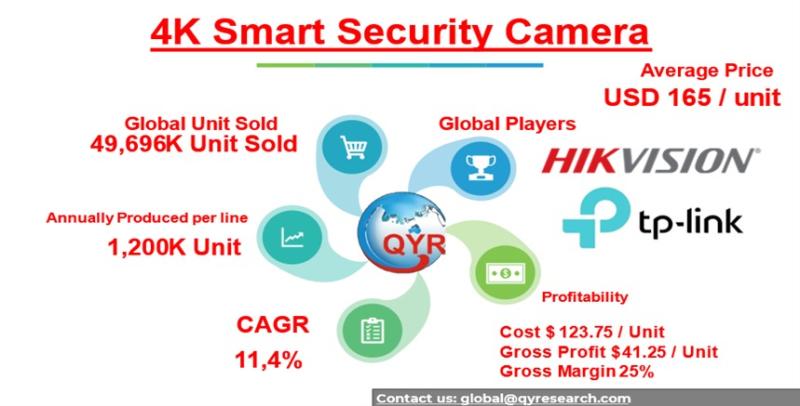
4K SMART SECURITY CAMERA MARKET EDGE AI AND SMART SECURITY: UNLOCKING THE NEXT P …
Executive Investment Snapshot:
The global 4K smart security camera market is entering a structural inflection point. What was historically a commoditized hardware segment defined by analog CCTV is rapidly transforming into a software-driven, AI-integrated intelligence platform, where pricing power is increasingly concentrated among vendors that can marry high-resolution optics with edge-computing capabilities.
In 2025, the global 4K smart security camera market is valued at approximately USD 8.2 billion, projected to reach USD…
Top 30 Indonesian Pulp and Paper Public Companies Q3 2025 Revenue & Performance
1) Overall companies performance (Q3 2025 snapshot)
PT Indah Kiat Pulp & Paper Tbk (INKP)
PT Pabrik Kertas Tjiwi Kimia Tbk (TKIM)
PT Toba Pulp Lestari Tbk (INRU)
PT Suparma Tbk (SPMA)
PT Indonesia Fibreboard Industry Tbk (IFII)
PT SLJ Global Tbk (SULI)
PT Inter Delta Tbk (INTD)
PT Indo Komoditi Korpora Tbk (INCF)
PT Kertas Basuki Rachmat Indonesia Tbk (KBRI)
PT Alkindo Naratama Tbk (ALDO)
PT Fajar Surya Wisesa Tbk (FASW)
PT Tirta Mahakam Resources Tbk (TIRT)
PT Kedawung Setia Industrial Tbk…
More Releases for Synbio
Synthetic Biology Market: Future Trends & Opportunity Outlook to 2032 | Thermo F …
Synthetic Biology Market is estimated to be valued at USD 21.90 Bn in 2025 and is expected to reach USD 90.73 Bn by 2032, growing at a compound annual growth rate (CAGR) of 22.5% from 2025 to 2032.
Overview
The "Synthetic Biology Market 2025 Forecast to 2032" report delivers precise global, regional, and country-level insights backed by reliable economic analysis. It presents a clear view of the competitive environment and includes a…
Synthetic Biology Market Set to Witness Significant Growth by 2025-2032 | Thermo …
The Synthetic Biology Market Research Report results from rigorous investigation and analysis carried out by our seasoned market research experts. It delivers a comprehensive view of the Synthetic Biology Market between 2025 and 2032, covering competitive dynamics, consumer behavior patterns, and technological breakthroughs. Industry participants will find this report highly valuable-equipping them with actionable intelligence to make strategic decisions and maintain an edge in a fast-changing market environment. Featuring an…
Synthetic Biology Market is evolving rapidly Through 2025 To 2032 | Thermo Fishe …
Synthetic Biology Market is estimated to be valued at USD 21.90 Bn in 2025 and is expected to reach USD 90.73 Bn by 2032, growing at a compound annual growth rate (CAGR) of 22.5% from 2025 to 2032.
Coherent Market Insights Reports has released a detailed research analysis on the "Synthetic Biology Market 2025", highlighting key trends, growth dynamics, and forecast insights through 2032. This comprehensive report presents an in-depth evaluation…
Synthetic Biology Market Deep Research Report with Forecast to 2032 | Thermo Fis …
The synthetic biology market is estimated to be valued at USD 21.90 Bn in 2025 and is expected to reach USD 90.73 Bn by 2032, growing at a compound annual growth rate (CAGR) of 22.5% from 2025 to 2032.
The Synthetic Biology Market Report is the result of extensive research and analysis conducted by our team of experienced market researchers. It encompasses a wide range of critical factors influencing the Synthetic…
Synthetic Biology Market Surging with Biotech Innovations | Size, Share & Future …
The global Synthetic Biology Market is expected to grow at 22.3% CAGR from 2025 to 2032.
The latest Research report published by CMI with the title "An Increase in Demand and Opportunities for Global Synthetic Biology Market 2025" provides a sorted image of the Synthetic Biology industry by analysis of research and information collected from various sources that have the ability to help the decision-makers in the worldwide market to play…
Synthetic Biology Market Outlook: Innovation, Market Size & Future Demand Trends …
The global Synthetic Biology Market is expected to grow at 22.3% CAGR from 2025 to 2032.
The global Synthetic Biology Market Report offers an in-depth analysis of the current trends, drivers, and quantitative market dynamics. The report signifies the significance of Growth and innovations, technology implementation, globally competitive landscape, and adapting to current developments in the Synthetic Biology Market Industry. Key highlights of the report include insights into market size, CAGR,…
