Press release
IV Solutions Manufacturing Plant DPR & Unit Setup 2026: Complete Cost, Process Flow & Investment Guide
Setting up IV solutions manufacturing plant positions investors in one of the most critical and non-discretionary segments of the global pharmaceutical and healthcare manufacturing industry, driven by the growing incidence of chronic diseases, rising hospitalization rates, increasing surgical procedures globally, and the expanding need for supportive intravenous therapy in acute care, post-operative recovery, and home healthcare settings. As aging populations in developed and developing economies intensify demand for parenteral nutrition, electrolyte replenishment, and antibiotic infusion therapies, governments accelerate healthcare infrastructure investment, emergency preparedness spending increases, and innovations in premixed IV bags and ready-to-use formats improve clinical convenience and reduce contamination risk, IV solutions continue to present compelling investment opportunities for manufacturers able to meet the stringent pharmaceutical-grade, cGMP-compliant production standards required by hospitals, ambulatory surgical centers, and home healthcare providers worldwide.Market Overview and Growth Potential:
The global IV solutions market demonstrates significant and sustained growth, valued at USD 16.31 Billion in 2025. According to IMARC Group's comprehensive market analysis, the market is expected to reach USD 24.30 Billion by 2034, exhibiting a CAGR of 4.5% from 2026 to 2034. The market is driven by an aging population, increasing healthcare needs, and the rise in chronic diseases requiring fluid and nutrient management. The market's demand is fundamentally underpinned by irreplaceable clinical necessity-IV therapy is the primary modality for fluid administration, electrolyte balancing, parenteral nutrition, and intravenous drug delivery across acute care, surgical, and emergency settings.
Request for Sample Report: https://www.imarcgroup.com/iv-solutions-manufacturing-plant-project-report/requestsample
The chronic disease burden driving IV therapy demand is substantial and growing. Non-communicable diseases contribute to more than 70% of all deaths worldwide each year according to the World Health Organization, and many of these require lengthy intravenous therapy, especially in acute care and post-operative situations. The International Diabetes Federation reported that the number of people with diabetes reached 537 million in 2021 and will increase to 643 million by 2030, with numerous such patients requiring IV fluids as part of regular treatment protocols. The World Health Organization further estimates that by 2050, one in every six individuals worldwide will be aged above 65-a demographic that disproportionately requires parenteral nutrition, rehydration therapy, and post-operative IV care.
Intravenous (IV) solutions are sterile liquids composed of water, electrolytes, and dissolved materials infused directly into the bloodstream. They correct fluid balance, administer drugs, and provide life-sustaining nutrients to patients-particularly during surgery, trauma, or dehydration-when immediate bioavailability and accurate therapy control are paramount. IV solution manufacturing plants are specialty pharmaceutical facilities producing sterile, pharmacopeial-grade fluids intended for direct introduction into the human body, operating under strict pharmaceutical-grade standards in aseptic cleanroom conditions and subject to rigorous cGMP and USFDA/WHO-GMP quality requirements.
Plant Capacity and Production Scale:
The proposed IV solutions manufacturing facility is designed with an annual production capacity ranging between 10-50 Million Liters, enabling economies of scale while maintaining operational flexibility. This capacity range allows manufacturers to serve diverse market segments across hospitals and clinics, home healthcare providers, and ambulatory surgical centers (ASCs)-ensuring steady demand and consistent revenue streams from fluid and electrolyte administration, parenteral nutrition delivery, antibiotic and drug infusion, and emergency rehydration therapy applications. The scale of global IV production requirements is illustrated by leading industry partnerships: the ICU Medical and Otsuka Pharmaceutical Factory partnership created one of the largest global IV solutions manufacturing networks with a combined output of approximately 1.4 billion units annually, reflecting the large-scale production volumes that characterize this segment.
Speak to an Analyst for Customized Report: https://www.imarcgroup.com/request?type=report&id=12020&flag=C
Financial Viability and Profitability Analysis:
The financial projections for the proposed project have been developed based on realistic assumptions related to capital investment, operating costs, production capacity utilization, pricing trends, and demand outlook. These projections provide a comprehensive view of the project's financial viability, ROI, profitability, and long-term sustainability.
• Gross Profit Margins: 50-60%
• Net Profit Margins: 20-35%
The IV solutions manufacturing business demonstrates compelling profitability potential supported by the non-discretionary, clinically essential nature of IV fluids across hospital, emergency, and home healthcare settings. These margins are underpinned by irreplaceable demand from hospitals, ICUs, and surgical centers where IV therapy is a standard of care; the structural growth tailwind from aging populations, rising chronic disease prevalence, and expanding healthcare infrastructure in emerging economies; government and institutional procurement that provides revenue predictability; barriers to entry from cGMP cleanroom infrastructure requirements, regulatory approvals, and stringent quality certification timelines; and premium pricing potential for specialty formulations including total parenteral nutrition (TPN), premixed drug formulations, and ready-to-use IV products. The project demonstrates strong return on investment (ROI) potential with comprehensive financial analysis covering capital expenditure, operating expenditure, income projections, NPV, IRR, and payback period.
Cost of Setting Up an IV Solutions Manufacturing Plant:
Operating Cost Structure:
Understanding the operating expenditure (OpEx) is crucial for effective financial planning. The cost structure includes:
• Raw Materials: 40-50% of total OpEx
• Utilities: 20-30% of OpEx
• Other Expenses: Labor, packaging, transportation, maintenance, depreciation, taxes
Raw materials at 40-50% of operating costs, with Water for Injection (WFI), Sodium Chloride, Dextrose/Glucose, Potassium Chloride, Calcium Chloride, Sodium Lactate, Sodium Bicarbonate, and other pharmaceutical-grade additives as the primary inputs. The relatively lower raw material share compared to other manufacturing categories reflects the high value-added nature of pharmaceutical-grade sterile IV production, where cleanroom operations, regulatory compliance, quality assurance, and aseptic manufacturing expertise represent a significant proportion of total cost. Utilities at 20-30% are notably elevated, reflecting the substantial energy demands of cleanroom HVAC systems, autoclave sterilization, WFI generation, and continuous environmental monitoring systems required for cGMP-compliant aseptic manufacturing. By the fifth year, total operational costs are expected to increase substantially due to inflation, regulatory compliance requirements, and market fluctuations in pharmaceutical-grade input pricing. Long-term contracts with reliable pharmaceutical-grade suppliers help stabilize raw material costs and ensure consistent supply of quality-certified inputs.
Capital Investment Requirements:
Setting up IV solutions manufacturing plant requires substantial capital investment reflecting the stringent pharmaceutical-grade cleanroom infrastructure, specialized aseptic manufacturing equipment, and regulatory compliance systems required. Total investment depends on plant capacity, technology level, regulatory jurisdiction, and location.
Land and Site Development: Location must offer easy access to key raw materials, primarily Water for Injection (WFI)-grade water, Sodium Chloride, Dextrose (Glucose), Potassium Chloride, Calcium Chloride, Sodium Lactate, Sodium Bicarbonate, and other pharmaceutical-grade additives. Proximity to major hospital networks, healthcare distributors, and medical supply chains minimizes distribution costs. The site must have robust infrastructure including reliable pharmaceutical-grade utilities, cleanroom HVAC systems, effluent treatment, and compliance with local zoning, cGMP, USFDA, and WHO-GMP regulations.
Machinery and Equipment: Machinery costs account for the largest portion of capital expenditure. Essential equipment includes:
• Water purification systems (Reverse Osmosis and Electrodeionization/EDI systems for WFI generation)
• Solution preparation tanks and formulation vessels
• Filtration units (sterile filtration systems)
• Form-fill seal machines (aseptic filling lines)
• Sterilizers and autoclaves (terminal sterilization)
• Leak testing machines
• Labeling and packaging machines
Civil Works: ISO-classified cleanroom construction with controlled temperature, humidity, particle count, and positive pressure differential is the most critical civil works component. Separate designated areas for WFI generation, raw material weighing, solution preparation, aseptic filling, sterilization, visual inspection, labeling, QC laboratory, finished goods quarantine, and approved product warehousing must all be incorporated, with full gowning protocols, airlocks, and contamination prevention infrastructure meeting cGMP, USFDA, and WHO-GMP requirements.
Buy Now: https://www.imarcgroup.com/checkout?id=12020&method=2175
Major Applications and Market Segments:
IV solutions serve critical and life-sustaining applications across inpatient, outpatient, surgical, and home healthcare settings:
• Hospitals and Clinics: The largest end-use sector, where IV solutions are used for fluid and electrolyte administration in surgical, ICU, emergency department, and general ward settings, and for intravenous delivery of antibiotics, chemotherapy, analgesics, and other medications requiring rapid and precisely controlled systemic bioavailability that oral routes cannot achieve
• Home Healthcare: A rapidly growing segment where patients requiring long-term IV antibiotic therapy, parenteral nutrition, pain management, or hydration are transitioned from hospital to home infusion settings, driven by cost reduction imperatives, patient preference, and the availability of premixed and ready-to-use IV formulations that reduce preparation complexity and contamination risk outside clinical environments
• Ambulatory Surgical Centers (ASCs): Outpatient surgical facilities where IV solutions are essential for pre-operative hydration, intra-operative fluid management, post-operative recovery, and medication delivery, with ASC growth in both developed and emerging markets driven by cost-effective outpatient surgery trends creating a growing and geographically distributed demand channel
Process: Water purification and WFI generation, weighing and mixing of active pharmaceutical ingredients, sterile filtration, aseptic filling into IV bags or bottles, sealing, terminal sterilization by autoclave, visual inspection, labeling, and packaging.
Why Invest in IV Solutions Manufacturing?
Compelling factors driving investment in the IV solutions manufacturing sector include:
• Non-Discretionary Clinical Demand: IV solutions are an irreplaceable standard of care across surgery, trauma, critical illness, chronic disease management, and emergency medicine, creating a structurally durable and largely recession-resistant demand base driven by fundamental healthcare utilization trends rather than consumer spending patterns
• Chronic Disease and Aging Population Tailwinds: Non-communicable diseases contributing to over 70% of global deaths annually, 643 million diabetes patients projected by 2030, and one in six people globally aged above 65 by 2050 together create a compounding structural demand growth driver that will sustain IV solutions market expansion for decades
• Healthcare Infrastructure Expansion: Heavy investment in hospital infrastructure across the Middle East, Asia Pacific, and Latin America is expanding the installed base of healthcare facilities requiring reliable IV fluid supply, with major government programs such as India's Pradhan Mantri Ayushman Bharat Health Infrastructure Mission increasing hospital capacity and indirectly boosting IV solutions consumption
• Emergency Preparedness and Strategic Stockpiling: The COVID-19 pandemic and natural disaster-related supply disruptions have prompted governments worldwide to accelerate strategic stockpiling and domestic manufacturing of critical medical consumables including IV fluids, creating institutional procurement programs that provide stable, large-volume demand for certified domestic manufacturers
• Regulatory Barriers Protect Margins: The stringent cGMP, USFDA, WHO-GMP, and national pharmacopeia compliance requirements for IV solution manufacturing create substantial barriers to entry that protect established producers' market positions and pricing power, with multi-year regulatory approval timelines limiting competitive disruption once market position is established
Manufacturing Process Excellence:
The IV solutions manufacturing process is a highly controlled, multi-step pharmaceutical operation encompassing:
• Water purification and WFI (Water for Injection) generation via reverse osmosis and electrodeionization systems
• Weighing and dispensing of pharmaceutical-grade active ingredients (NaCl, dextrose, KCl, CaCl2, sodium lactate, sodium bicarbonate, and other additives)
• Solution preparation through precision dissolution and mixing in formulation vessels
• In-process quality checks including pH, osmolarity, and concentration verification
• Sterile filtration through 0.22-micron membrane filters under aseptic conditions
• Aseptic filling into IV bags or glass/plastic bottles via form-fill-seal machines in cleanroom environments
• Sealing and terminal sterilization by autoclave (steam sterilization)
• Leak testing and 100% visual inspection for particulates, clarity, and container integrity
• Finished product release testing including sterility, pyrogen/endotoxin, pH, osmolarity, and chemical assay testing
• Labeling, secondary packaging, quarantine, QA batch release, and cold-chain or ambient distribution
Comprehensive quality control is maintained throughout all manufacturing stages under cGMP protocols. Analytical instruments and validated quality systems monitor WFI water quality (TOC, conductivity, endotoxins), in-process solution concentration, pH, osmolarity, bioburden prior to filtration, sterility, pyrogen/bacterial endotoxin levels, particulate matter, container integrity, and finished product specification compliance to ensure all output meets applicable pharmacopeial, USFDA, WHO-GMP, and customer specification requirements for life-critical parenteral administration.
Industry Leadership:
Leading manufacturers in the global IV solutions industry include several multinational companies with extensive production capacities and diverse application portfolios. Key players include:
• Fresenius Kabi AG
• B. Braun SE
• Baxter
• Otsuka Pharmaceutical Co., Ltd.
• Pfizer, Inc.
• JW Life Science
• Vifor Pharma
• ICU Medical, Inc.
All operate large-scale facilities serving end-use sectors including hospitals and clinics, home healthcare, and Ambulatory Surgical Centers (ASCs).
Recent Industry Developments:
• October 2025: Celltrion, Inc. announced the availability of its Avtozma (tocilizumab-anoh) intravenous (IV) formulation for patients in the United States. Approved for the same indications as the reference product Actemra (tocilizumab), Avtozma IV is indicated for rheumatoid arthritis (RA), giant cell arteritis (GCA), polyarticular juvenile idiopathic arthritis (PJIA), systemic juvenile idiopathic arthritis (SJIA), COVID-19, and cytokine release syndrome (CRS).
• September 2025: B. Braun Medical Inc. announced that its entire IV Solutions portfolio earned the esteemed Nurse Approved Certification. This certification, awarded through the rigorous Nurse Quality Standards Index process, highlights B. Braun's dedication to excellence in product usability, workflow integration, and meeting nursing practice needs.
Browse Full Report: https://www.imarcgroup.com/iv-solutions-manufacturing-plant-project-report
About Us:
IMARC Group is a global management consulting firm that helps the world's most ambitious changemakers to create a lasting impact. The company excels in understanding its client's business priorities and delivering tailored solutions that drive meaningful outcomes. We provide a comprehensive suite of market entry and expansion services. Our offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape, and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: (+1-201-971-6302)
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release IV Solutions Manufacturing Plant DPR & Unit Setup 2026: Complete Cost, Process Flow & Investment Guide here
News-ID: 4431071 • Views: …
More Releases from IMARC Group

Tire Market to Surpass USD 272.6 Billion by 2034, At a CAGR of 4.33%
According to IMARC Research, The global tire market has stabilized and is indicating uniform growth. In the long term‚ the global automotive tire market is envisaged to grow continuously due to increasing demand for automobiles‚ constant development of tire-manufacturing technologies‚ and increasing sustainability emphasis. The rapidly growing automotive industry has changed the perception of tires from consumables to a high-performance product that impacts fuel economy‚ safety‚ and driving comfort.
The market…

Canada Robotic Fruit Picker Market Set to Reach USD 12.87 Million by 2034, Drive …
Once defined by seasonal migrant workforces and hand-picked harvests, the Canada Robotic Fruit Picker Market is entering a new era of technology-led transformation. Persistent labor shortages, rising operational costs, and a wave of government investment in agricultural automation are collectively rewriting Canada's fruit harvesting story. After reaching USD 7.40 Million in 2025, the market is on a clear trajectory toward USD 12.87 Million by 2034, reflecting a steady CAGR of…
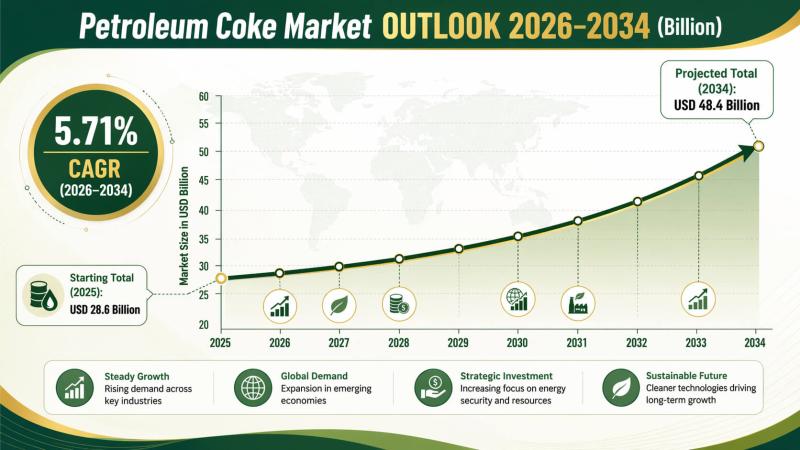
Petroleum Coke Market Size, Share, Trends and Forecast by Type, Application, and …
As per the IMARC group‚ the current market trends for petroleum coke are moderate. The increase in demand in energy intensive industries‚ such as aluminum‚ cement‚ steel‚ and power generation‚ is a major driving force for the market. Further‚ the high carbon content and high calorific value make petroleum coke suitable as an alternative fuel in countries with increasing industrialization and energy demand.
The market was valued at USD 28.6 Billion…

United States e-KYC Market Poised for Strong Growth Through 2033, Driven by Digi …
IMARC Group, a global market research and management consulting firm, has published a comprehensive new report on the United States e-KYC Market. According to the report, the United States e-KYC market is experiencing robust expansion, driven by the rapid growth of digital financial services, stringent regulatory compliance requirements, and accelerating adoption of AI-powered identity verification technologies across the banking, fintech, insurance, and telecom sectors.
Electronic Know Your Customer (e-KYC) refers to…
More Releases for WFI
Membrane Based WFI Production System Market Report 2026: Trend Analysis and Futu …
The global market for Membrane Based WFI Production System was estimated to be worth US$ 17.08 million in 2025 and is projected to reach US$ 23.26 million, growing at a CAGR of 4.6% from 2026 to 2032.
2026 Market Report by QYResearch "Membrane Based WFI Production System - Global Market Share and Ranking, Overall Sales and Demand Forecast 2026-2032" provides an extensive examination of Membrane Based WFI Production System market…
Water for Injection (WFI) Market to Reach USD 57.65 Billion by 2032, Growing at …
Water for Injection (WFI) Market was valued at approximately USD 30.10 billion in 2024 and is expected to grow at a CAGR of 8.01% from 2025 to 2032.
WFI refers to ultra-purified water used as an essential solvent and excipient in the production of injectable drugs, vaccines, biologics, and advanced therapies such as cell and gene therapies. The market is driven by the rapid growth of injectable pharmaceuticals, particularly in oncology,…
Water for Injection (WFI) System Market Emerging Industry Trends, Growth Dynamic …
Los Angeles, United States - QY Research has unveiled its latest study titled "Global Water for Injection (WFI) System Market Share and Ranking, Overall Sales and Demand Forecast 2025-2031." This newly released report delivers a comprehensive overview of the global Water for Injection (WFI) System market, analyzing the forces shaping its growth trajectory and providing valuable insights for stakeholders, investors, and industry professionals. the research covers key areas including market…
Cold WFI Generation Systems Market Size, Growth, Forecast 2025-2031 (By Type, By …
The global market for Cold WFI Generation Systems was estimated to be worth US$ 16.4 million in 2024 and is forecast to a readjusted size of US$ 22.3 million by 2031 with a CAGR of 4.6% during the forecast period 2025-2031.
QY Research (Market Research Report Publisher) announces the release of its lastest report "Cold WFI Generation Systems - Global Market Share and Ranking, Overall Sales and Demand Forecast 2025-2031".…
Water for Injection (WFI) System Research:CAGR of 3.9% during the forecast perio …
QY Research Inc. (Global Market Report Research Publisher) announces the release of 2024 latest report "Water for Injection (WFI) System- Global Market Share and Ranking, Overall Sales and Demand Forecast 2024-2030". Based on current situation and impact historical analysis (2019-2023) and forecast calculations (2024-2030), this report provides a comprehensive analysis of the global Wire Drawing Dies market, including market size, share, demand, industry development status, and forecasts for the next…
Global Water for Injection (WFI) System Market Overview Analysis of BRAM-COR, St …
Global Info Research Publishes the release of the report "Global Water for Injection (WFI) System Market 2023 by Manufacturers, Regions, Type and Application, Forecast to 2029" . This report is a detailed and comprehensive analysis of the global market, with a particular focus on future projections. The report is structured into various segments, each analyzing the competition, supply and demand trends, as well as key factors that contribute to its changing demands…
