Press release
Pharmacovigilance Software Market Set for Steady Growth to US$415.42 Million by 2033, Led by North America's 38% Market Share
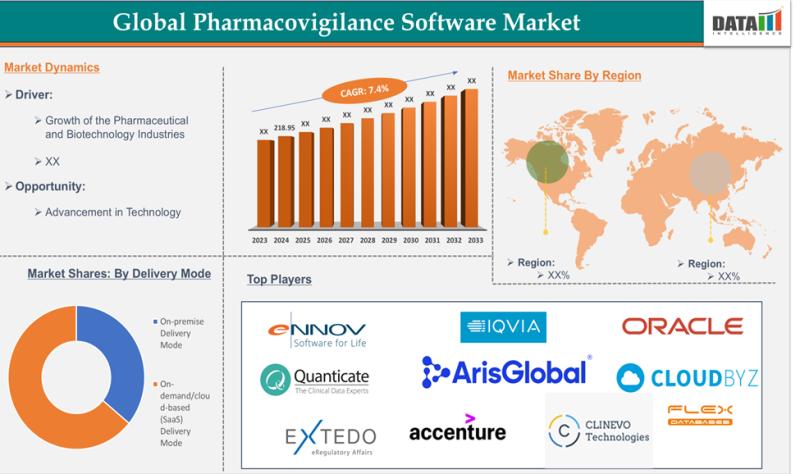
The Global Pharmacovigilance Software Market reached US$218.95 million in 2024 and is expected to reach US$415.42 million by 2033, growing at a CAGR of 7.4% during the forecast period of 2025-2033.The market is steadily expanding as pharmaceutical companies and regulatory bodies increasingly adopt digital solutions for adverse event monitoring and compliance, driven by stringent FDA/EMA guidelines and the rise in drug approvals. This growth underscores a shift toward AI-powered automation and real-time data analytics, enhancing patient safety and accelerating post-market surveillance in healthcare IT.
Download Executive Sample Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/download-sample/pharmacovigilance-software-market?ram
Key Industry Developments
United States:
✅ February 2026: Oracle launched an enhanced version of its Argus Safety Suite with AI-driven signal detection and automated case triage features, significantly reducing processing times for adverse event reports and improving regulatory compliance for US pharmaceutical firms.
✅ December 2025: ArisGlobal introduced LifeSphere Safety powered by generative AI for US CROs, featuring automated narrative generation and multi-language case processing to accelerate submissions to the FDA.
✅ October 2025: IQVIA unveiled PV Nexus 360, a SaaS platform with advanced ML algorithms for US biotech companies, focusing on predictive risk modeling and E2B(R3)-compliant submissions.
Japan:
✅ January 2026: Hitachi Solutions Japan released VigiFlow Advanced with integrated PMDA-compliant modules, incorporating AI for automated causality assessment in ADR reports tailored for Japanese regulators.
✅ November 2025: Fujitsu launched a pharmacovigilance platform leveraging quantum-inspired optimization for Japan-based pharma, improving adverse event prioritization and JADER database integration.
✅ October 2025: NEC Corporation debuted Synapse PV with blockchain-enabled data sharing for Japanese CROs, facilitating secure cross-border pharmacovigilance collaborations while adhering to local privacy laws.
Key Players:
Ennov | Oracle Corporation | ArisGlobal | EXTEDO | Clinevo Technologies | IQVIA | Quanticate | Cloudbyz | Acceenture | Flex Databases
Strategic Leadership Analysis: Top 5 Players in Pharmacovigilance Software Market 2026
-Oracle: Deployed the Argus Safety platform with advanced AI-driven case processing for pharmacovigilance, enhancing real-time adverse event management and global regulatory compliance to streamline safety operations across clinical trials and post-market surveillance.
-ArisGlobal: Advanced the LifeSphere Safety suite with cloud-native automation features, delivering unified signal detection and multi-lingual case intake to accelerate risk assessment and improve pharmacovigilance efficiency for global biopharma organizations.
-IQVIA: Expanded the Pharmacovigilance & Safety Services platform with integrated real-world evidence analytics, enabling predictive safety insights and automated aggregate reporting to support proactive risk mitigation in drug development pipelines.
-Acceenture: Strengthened the Life Sciences Cloud for Pharmacovigilance with AI-powered workflow orchestration, featuring intelligent triage and endpoint protection tailored for decentralized trials and enhanced compliance with evolving global regulations.
-EXTEDO: Upgraded the eMDR & PV modules within the Quality Management Suite, providing seamless electronic submissions and configurable rule-based processing to bolster case validation accuracy and regulatory intelligence in pharmacovigilance workflows.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/pharmacovigilance-software-market?ram
Main Drivers and Trends Shaping the Future of Pharmacovigilance Software Market
-AI and Automation Surge: Artificial Intelligence and Machine Learning enhance signal detection, adverse event prediction, and case processing automation, reducing manual workloads and boosting accuracy.
-Cloud-Based Adoption: Scalable SaaS solutions offer cost-effectiveness and flexibility for global teams, driving demand amid rising data volumes from pharmaceutical R&D.
-Regulatory and R&D Pressures: Increased drug approvals, biologics growth, and stringent post-market surveillance requirements fuel software needs for real-time analytics and compliance.
-Market Hurdles: High development costs, complex global regulatory harmonization, and integration challenges with real-world evidence systems constrain rapid expansion.
Regional Insights:
-North America: 38% (Largest share, driven by dense clinical‐trial activity, stringent FDA‐style regulations, and widespread adoption of cloud‐based PV systems).
-Asia Pacific: 24% (Fastest‐growing region, fueled by expanding clinical‐trial sites, rising drug‐development pipelines, and strong outsourcing to CRO‐based pharmacovigilance platforms in China, India, and Japan).
-Europe: 21% (Supported by harmonized pharmacovigilance frameworks, robust risk‐management planning, and steady investment from both EU‐EMA and national agencies).
-Latin America: 9% (Emerging, with gradual regulatory upgrades and increasing demand from local generics and biopharma manufacturers).
-Middle East & Africa: 9% (Gradual digital adoption, led by Gulf‐region pharma hubs and South Africa, with a growing shift from manual to digital safety‐monitoring tools).
Market Opportunities & Challenges: Pharmacovigilance Software Market 2026
Pharmacovigilance software addresses drug safety monitoring through advanced case processing and compliance tools. Here's a structured view of key dynamics.
-Opportunities
A "Cloud-First Shift" accelerates adoption among pharma and biotech firms, with AI-powered signal detection enabling real-time adverse event analysis for faster regulatory submissions.
Expansion into Asia Pacific and emerging markets leverages rising clinical trials and localized PV platforms tailored to evolving regional regulations.
Digital transformation in drug safety management supports scalable solutions for biologics pipelines, integrating automated risk assessment amid global R&D surges.
-Challenges
Legacy system migrations demand robust data validation and audit trails, complicating transitions for firms with entrenched on-premise deployments.
Stringent global harmonization, including PSUR and medical device vigilance mandates, strains resource allocation across fragmented multinational operations.
Balancing AI automation with manual oversight persists, as 39% of monitoring in emerging regions like Middle East & Africa still relies on outdated processes.
Purchase Corporate License | Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=pharmacovigilance-software-market?ram
Market Segmentation Analysis:
-By Functionality: Adverse Event Reporting Leads Case Management
Adverse Event Reporting Software dominates with 40% share in 2025, essential for real-time collection, collation, and regulatory submission of safety incidents from global sources.
Drug Safety Audits Software holds 25%, focusing on compliance checks and audit trails to ensure regulatory adherence during inspections.
Issue Tracking Software captures 20%, streamlining resolution of safety queries and escalations across teams.
Fully Integrated Software takes 15%, combining all modules for end-to-end pharmacovigilance workflows but with higher implementation costs.
-By Delivery Mode: Cloud-Based Gains Momentum
On-demand/Cloud-based (SaaS) Delivery Mode leads at 55% share, offering scalability, remote access, and lower upfront costs amid digital transformation.
On-premise Delivery Mode follows at 45%, preferred by firms needing data control and customization despite higher maintenance.
-By End-User: Pharma/Biotech Commands Volume
Pharma and Biotech Companies hold 50% share, driving demand via in-house safety monitoring for vast drug portfolios.
Contract Research Organizations (CROs) take 25%, supporting outsourced clinical trial safety data.
Business Process Outsourcing (BPO) Firms claim 15%, handling cost-effective PV operations.
Other PV Service Providers account for 10%, covering specialized consultancies.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription?ram
Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTW
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Pharmacovigilance Software Market Set for Steady Growth to US$415.42 Million by 2033, Led by North America's 38% Market Share here
News-ID: 4423662 • Views: …
More Releases from DataM intelligence 4 Market Research LLP
ESR1 Mutated Metastatic Breast Cancer Diagnostics Market to Reach USD 93.21 Mill …
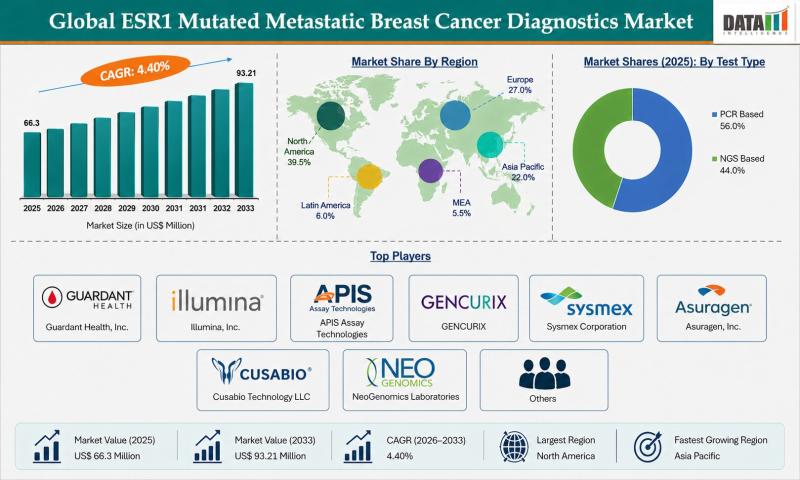
The global ESR1 mutated metastatic breast cancer diagnostics market reached USD 66.3 million in 2025 and is expected to reach USD 93.21 million by 2033, growing at a CAGR of 4.4% during the forecast period from 2026 to 2033. The market is witnessing steady growth driven by the increasing prevalence of hormone receptor-positive breast cancer and rising awareness regarding genetic mutation testing for targeted treatment. Growing adoption of precision oncology…
Oncology Molecular Diagnostics Market to Reach USD 6.46 Billion by 2033 at 6.2% …
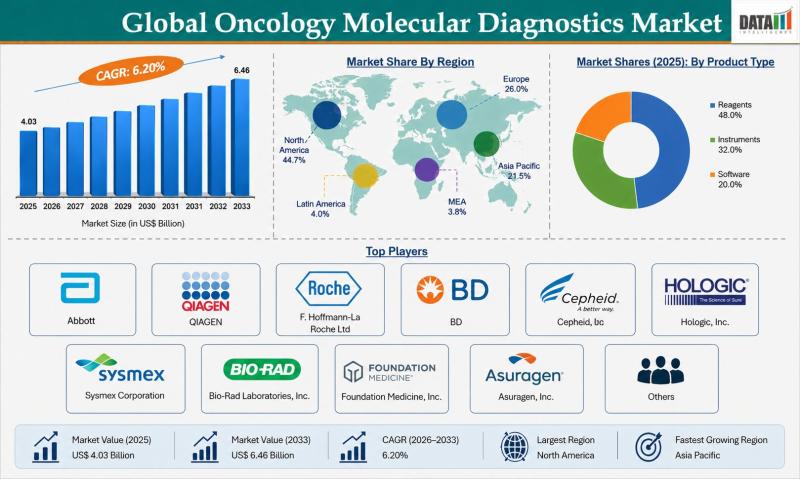
The global oncology molecular diagnostics market reached USD 4.03 billion in 2025 and is expected to reach USD 6.46 billion by 2033, growing at a CAGR of 6.2% during the forecast period from 2026 to 2033. The market is witnessing steady growth driven by the increasing prevalence of cancer, rising demand for early and accurate diagnosis, and growing adoption of precision medicine approaches. Advancements in genomic technologies and increasing awareness…
Solar Energy Market to Reach USD 416.13 Billion by 2033 at 10.6% CAGR; Asia Paci …
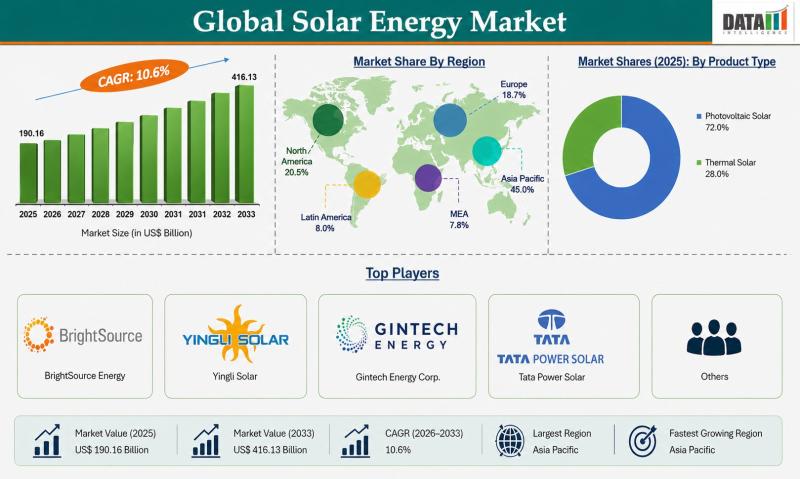
The global solar energy market reached USD 190.16 billion in 2025 and is expected to reach USD 416.13 billion by 2033, growing at a CAGR of 10.6% during the forecast period from 2026 to 2033. The market is witnessing strong growth driven by increasing demand for clean and renewable energy sources, rising concerns over carbon emissions, and supportive government policies promoting solar power adoption. Rapid urbanization, growing electricity demand, and…
Zero Trust Security Market Set for Explosive Growth to US$ 137.54 Billion by 203 …
DataM Intelligence has released a new research report titled "Zero Trust Security Market Size 2026". The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in…
More Releases for Pharma
Schizophrenia Market: Expanding Revenue Landscape to 2034 - DelveInsight | Teva …
The Key Schizophrenia Companies in the market inlclude - Teva Pharmaceutical Industries, Royalty Pharma, MedinCell, Sumitomo Pharma, Otsuka Pharmaceuticals, Reviva Pharmaceuticals, Minerva Neurosciences, Mitsubishi Tanabe Pharma, Lundbeck, Intra-cellular Therapies, Sumitomo Pharma, Otsuka Pharma, Reviva Pharmaceuticals, and others.
DelveInsight's report "Schizophrenia Market Insights, Epidemiology, and Market Forecast-2034" provides a comprehensive analysis of the Schizophrenia landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the…
Dacarbazine Market Size, Share and Forecast By Key Players-Lingnan Pharma, Ruiyi …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Dacarbazine market is anticipated to grow at a compound annual growth rate (CAGR) of 14.63% between 2024 and 2031. The market is expected to grow to USD 25.32 Billion by 2024. The valuation is expected to reach USD 65.84 Billion by 2031.
The Dacarbazine market is projected to witness significant growth in the coming years, driven by increasing…
Dacarbazine Market Size, Share and Forecast By Key Players-Lingnan Pharma, Ruiyi …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Dacarbazine market is anticipated to grow at a compound annual growth rate (CAGR) of 14.63% between 2024 and 2031. The market is expected to grow to USD 25.32 Billion by 2024. The valuation is expected to reach USD 65.84 Billion by 2031.
The dacarbazine market is experiencing steady growth, driven by the rising incidence of cancer globally. As…
Epilepsy Pipeline Assessment 2024: Therapies, Clinical Trials, and Market Insigh …
(Las Vegas, Nevada, United States) As per DelveInsight's assessment, globally, Epilepsy pipeline constitutes 75+ key companies continuously working towards developing 90+ Epilepsy treatment therapies, analysis of Clinical Trials, Therapies, Mechanism of Action, Route of Administration, and Developments analyzes DelveInsight.
The Epilepsy Pipeline report embraces in-depth commercial and clinical assessment of the pipeline products from the pre-clinical developmental phase to the marketed phase. The report also covers a detailed description of the…
Global Indomethacin Market Seeking Excellent Growth by 2028 | Ratiopharm, Merck, …
"IBI has published a report entitled Global Indomethacin Market Research Report which provides comprehensive data on emerging trends, market drivers, growth opportunities, and barriers that could change the dynamics of the industry market. It provides an in-depth analysis of market segments which includes the product, application, and competitor analysis.
Click Here To Order A Sample Copy Of Indomethacin Global Market Report : https://www.infinitybusinessinsights.com/request_sample.php?id=380382
The Indomethacin Global Market Research Report provides close monitoring…
Roxatidine Market 2020: Top Companies Analysis To Grow Healthcare Business by 20 …
Business Industry Reports Research has recently announced a report on Global Roxatidine Market based on the Category Industry. The Roxatidine Market report emphasizes various key aspects, which include growth drivers, restraints, opportunities and recent market trends for the forecast period 2020-2024.
Global Roxatidine Market overview:
Business Industry Reports Analyst covers the Major Players data, including: shipment, revenue, gross profit, interview record, business distribution etc., these data help the consumer know about the…