Press release
Laparotomy Sponge Manufacturing Plant Cost, DPR- 2026: Business Plan, Raw Materials and Profitability
Setting up a laparotomy sponge manufacturing plant positions investors in one of the most stable and essential segments of the global medical disposables and surgical consumables value chain, backed by sustained global growth driven by rising surgical procedure volumes, increasing hospital infrastructure development, and growing emphasis on infection control and patient safety in operating rooms. As healthcare systems worldwide expand their surgical capacities, governments invest in public hospital infrastructure, and regulatory mandates for sterile single-use surgical products intensify, the laparotomy sponge industry continues to present compelling opportunities for manufacturers and entrepreneurs seeking long-term profitability in a high-demand, non-cyclical sector.Market Overview and Growth Potential:
The global laparotomy sponge market demonstrates an exceptional growth trajectory. According to IMARC Group's comprehensive market analysis, the global laparotomy sponge market size was valued at USD 1,128.4 Million in 2025. The market is expected to reach USD 1,852.07 Million by 2034, exhibiting a CAGR of 5.66% from 2026 to 2034. The market is primarily driven by rising surgical procedure volumes, increasing hospital infrastructure development, and growing emphasis on infection control and patient safety in operating rooms.
Request for Sample Report: https://www.imarcgroup.com/laparotomy-sponge-manufacturing-plant-project-report/requestsample
Laparotomy sponges are sterile, absorbent surgical textiles primarily used during open surgical procedures to absorb blood, maintain a clear operative field, and protect internal tissues. They are typically manufactured from medical-grade cotton gauze and may include radiopaque markers to enable detection through imaging systems, reducing the risk of retained surgical items. These sponges exhibit high absorbency, softness, lint-free characteristics, and tensile strength to withstand surgical handling. Manufactured under stringent quality and sterilization standards, laparotomy sponges are supplied in pre-counted, sterile packs to support surgical accuracy and compliance with hospital safety protocols.
The laparotomy sponge market is experiencing strong growth due to the global increase in surgical procedures resulting from aging populations, trauma cases, and the growing prevalence of non-communicable diseases. The International Society of Aesthetic Plastic Surgery (ISAPS) showed a higher increase of 5.5% in surgical procedures, with more than 15.8 million procedures performed by plastic surgeons and 19.1 million non-surgical procedures. Expansion of hospital infrastructure in emerging economies is further supporting demand for essential surgical consumables. Heightened focus on patient safety and surgical accuracy has increased the adoption of radiopaque sponges to reduce retained surgical item incidents. Additionally, stringent infection prevention protocols and mandatory use of sterile, single-use products in operating rooms continue to reinforce demand. Technological improvements in sterilization and packaging processes are also enhancing product reliability and hospital acceptance.
Plant Capacity and Production Scale:
The proposed laparotomy sponge manufacturing facility is designed with an annual production capacity ranging between 50 - 150 Million sponges, enabling economies of scale while maintaining operational flexibility. This capacity range allows producers to serve diverse market segments across hospital surgical units, outpatient clinics, emergency medical services, military field medicine, and veterinary surgery - ensuring steady demand and consistent revenue streams driven by rising surgical volumes, regulatory mandates for sterile single-use products, healthcare infrastructure expansion, technology upgradation opportunities, and applications in intraoperative fluid absorption, organ protection and retraction, maintaining a clear surgical field, post-operative wound packing, and emergency trauma management.
Speak to an Analyst for Customized Report: https://www.imarcgroup.com/request?type=report&id=16333&flag=C
Financial Viability and Profitability Analysis:
The laparotomy sponge manufacturing business demonstrates healthy profitability potential under normal operating conditions. The financial projections reveal:
• Gross Profit Margins: 25-40%
• Net Profit Margins: 10-20%
These margins are supported by stable demand across hospital surgical units, healthcare distributors, emergency services, and institutional buyers; value-added processing through non-woven forming, ultrasonic binding and edge sealing providing consistent product quality while maintaining low production costs; and the critical importance of laparotomy sponges as non-negotiable components of safe surgical practice across hospitals and surgical centers delivering dependable performance with precise absorbency that meets regulatory requirements. The project demonstrates strong return on investment (ROI) potential with comprehensive financial analysis.
Cost of Setting Up a Laparotomy Sponge Manufacturing Plant:
Operating Cost Structure:
Understanding the operating expenditure (OpEx) is crucial for effective financial planning. The cost structure includes:
• Raw Materials: 55-65% of total OpEx
• Utilities: 8-12% of OpEx
• Other Expenses: Labor, packaging, transportation, maintenance, depreciation, taxes, and regulatory compliance
Raw materials at 55-65% of operating costs, with medical-grade cotton gauze as the primary component, along with radiopaque thread/fibre and sterilization packaging materials. Utilities at 8-12%. By the fifth year, total operational cost is expected to increase substantially due to inflation, market fluctuations, and potential rises in the cost of key materials. Long-term contracts with reliable suppliers help stabilize pricing and ensure steady supply.
Capital Investment Requirements:
Setting up a laparotomy sponge manufacturing plant requires substantial capital investment. Total depends on plant capacity, technology, and location.
Land and Site Development: Location must offer easy access to key raw materials: medical-grade cotton gauze, radiopaque thread/fibre, and sterilization packaging materials. Proximity to target markets minimizes distribution costs. The site must have robust infrastructure including reliable transportation, utilities, and waste management systems. Compliance with local zoning laws and environmental regulations must also be ensured.
Machinery and Equipment: Machinery costs account for the largest portion. Essential equipment includes:
• Precision cutting machines
• Ultrasonic or heat-sealing units
• Folding and stacking systems
• Sterilization chambers
• Barcode printers
• Final packaging lines
Civil Works: Building construction and layout optimization. Separate areas for raw material storage, production, quality control, and finished goods storage must be designated. Space for future expansion should be incorporated to accommodate business growth.
Buy Now: https://www.imarcgroup.com/checkout?id=16333&method=2175
Major Applications and Market Segments:
Laparotomy sponges serve extensive applications across the surgical and healthcare value chain:
• General Surgery: Absorption of blood and fluids during laparotomy procedures, ensuring a clear and safe operative field
• Emergency and Trauma Care: Maintaining a clear surgical field in high-bleeding situations where rapid fluid absorption is critical
• Operating Rooms: Routine use in open abdominal surgeries for tissue protection, organ retraction, and fluid control
• Post-operative Care: Temporary wound management and cavity packing when required during recovery
The manufacturing process follows a rigorous multi-step sequence: raw material receiving and quality inspection, cutting of medical-grade cotton gauze to specified dimensions, radiopaque thread/marker integration, folding and stacking to required configuration, ultrasonic or heat-sealing of edges, sterilization processing, quality inspection including radiopaque detection verification, sterile packaging in pre-counted packs, barcode printing and labeling, and final goods storage and dispatch.
Why Invest in Laparotomy Sponge Manufacturing?
Several compelling factors make this an attractive investment opportunity:
• Critical Surgical Consumable: Laparotomy sponges are essential medical disposables used in open surgeries for blood absorption, fluid control, tissue protection, and maintaining a clear operative field - making them a non-negotiable component of safe surgical practice across hospitals and surgical centers
• Moderate but Defensible Entry Barriers: Strict compliance with medical-grade cotton standards, sterilization protocols, radiopaque detection requirements, regulatory approvals, and hospital validation processes creates meaningful entry barriers that favour quality-driven and compliant manufacturers
• Alignment with Healthcare Growth Trends: Rising surgical volumes driven by population growth, aging demographics, trauma cases, and expanding access to healthcare - especially in emerging markets - are generating steady and recurring demand for laparotomy sponges as high-consumption disposables
• Policy and Healthcare Infrastructure Support: Government investments in hospitals, medical colleges, public health systems, and domestic medical device manufacturing initiatives (e.g., Make in India, healthcare procurement programs) strengthen demand for essential surgical consumables
• Preference for Local, Reliable Supply Chains: Hospitals, distributors, and healthcare systems increasingly favour dependable local manufacturers to ensure consistent quality, uninterrupted supply, regulatory compliance, and cost stability
Manufacturing Process Excellence:
The laparotomy sponge manufacturing plant operates through a comprehensive multi-step operation:
• Non-woven forming and medical-grade cotton gauze preparation
• Precision cutting to specified dimensions
• Radiopaque thread/fibre integration and marker placement
• Folding and stacking to required configuration
• Ultrasonic binding and edge sealing
• Sterilization chamber processing
• Radiopaque detection verification and quality inspection
• Sterile packaging in pre-counted packs
• Barcode printing, labeling, and lot coding for traceability
• Final goods storage and dispatch
Comprehensive quality control is maintained throughout production. Analytical instruments monitor product absorbency, tensile strength, lint levels, sterility assurance, and radiopaque marker integrity. Documentation for traceability and regulatory compliance is maintained at every stage to meet hospital safety protocols and international medical device standards.
Industry Leadership:
Leading manufacturers in the global laparotomy sponge industry include several multinational companies with extensive production capacities and diverse application portfolios. Key players include:
• Medline Industries
• Cardinal Health
• 3M Healthcare
• Mölnlycke Health Care
• Owens & Minor
all of which serve end-use sectors such as hospital surgical units, outpatient clinics, emergency medical services, military field medicine, veterinary surgery.
Recent Industry Developments:
November 2024: Stryker unveiled its upcoming SurgiCount+ platform, a technology that aims to increase surgical safety and optimise operating room processes by saving up to 10 minutes per procedure. The platform combines RFID-enabled sponge monitoring with the Triton system for real-time blood loss estimation, directly reinforcing the value and technological evolution of laparotomy sponge management in modern surgical settings.
April 2024: Integra LifeSciences Corporation finalised the acquisition of Acclarent, Inc., an innovator in ENT surgical procedures, integrating the company into its Codman Specialty Surgical division. The move underscores the accelerating consolidation trend in the surgical consumables and medical device space, with major players expanding their surgical procedure portfolios to capture growing demand across specialised operative care segments.
Browse Full Report: https://www.imarcgroup.com/laparotomy-sponge-manufacturing-plant-project-report
About Us:
IMARC Group is a global management consulting firm that helps the world's most ambitious changemakers to create a lasting impact. The company excels in understanding its client's business priorities and delivering tailored solutions that drive meaningful outcomes. We provide a comprehensive suite of market entry and expansion services. Our offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape, and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: (+1-201-971-6302)
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Laparotomy Sponge Manufacturing Plant Cost, DPR- 2026: Business Plan, Raw Materials and Profitability here
News-ID: 4417106 • Views: …
More Releases from IMARC Group

Brazil Cloud Security Market to Reach USD 2,003.29 Million by 2034, Growing at a …
Brazil Cloud Security Market Summary:
• The Brazil Cloud Security Market size reached USD 609.28 Million in 2025.
• The market is projected to reach USD 2,003.29 Million by 2034.
• The market is expected to grow at a CAGR of 12.64% during 2026-2034.
• Large enterprises dominate the market due to rising investments in cloud infrastructure and cybersecurity frameworks.
• Growth is driven by increasing cloud adoption, rising cyber threats, expansion of remote working…

Vietnam Coffee Market Growth, Demand and Opportunity Report 2026-2034
Vietnam Coffee Market Summary:
• The Vietnam Coffee Market size reached 73.44 Thousand Tons in 2025.
• Market is projected to reach 102.23 Thousand Tons by 2034.
• Southern Vietnam dominates with a significant market share.
• Growth driven by rising domestic coffee consumption, increasing café culture expansion, and growing premium coffee exports.
IMARC Group, a leading market research company, has released its latest report titled "Vietnam Coffee Market." The study provides a detailed analysis…
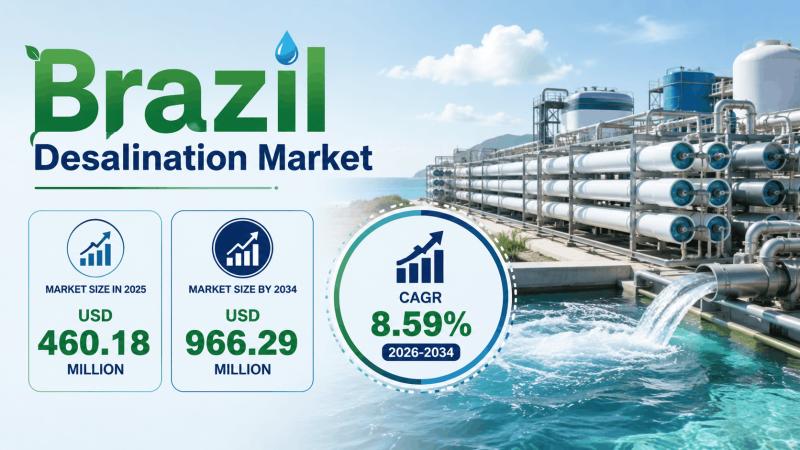
Brazil Desalination Market to Reach USD 966.29 Million by 2034, Growing at a CAG …
Brazil Desalination Market Summary:
• The Brazil Desalination Market size reached USD 460.18 Million in 2025.
• The market is projected to reach USD 966.29 Million by 2034.
• The market is expected to grow at a CAGR of 8.59% during 2026-2034.
• Rising water scarcity and increasing government investments in water infrastructure are driving market expansion.
• Growing adoption of renewable energy-powered desalination systems is supporting long-term industry growth.
Request Sample For PDF Report: https://www.imarcgroup.com/brazil-desalination-market/requestsample
The…

Hydrofluoric Acid Production Plant Setup DPR 2026: Machinery, Investment & Opera …
Setting up a hydrofluoric acid production plant positions investors in one of the most stable and essential segments of the specialty chemicals value chain, backed by sustained global growth driven by its extensive use in fluorochemicals production, petroleum refining, aluminum processing, and semiconductor manufacturing. Increasing demand for refrigerants, lithium-ion batteries, and specialty chemicals continues to support market growth. As electrification accelerates, semiconductor manufacturing intensifies, and fluorochemical applications diversify, the global…
More Releases for Laparotomy
Laparotomy Sponge Market Future Growth, Competitive Analysis and Forecast 2024-2 …
𝐋𝐚𝐩𝐚𝐫𝐨𝐭𝐨𝐦𝐲 𝐒𝐩𝐨𝐧𝐠𝐞 𝐌𝐚𝐫𝐤𝐞𝐭 𝐆𝐫𝐨𝐰𝐭𝐡 𝐨𝐫 𝐃𝐞𝐦𝐚𝐧𝐝 𝐈𝐧𝐜𝐫𝐞𝐚𝐬𝐞 𝐨𝐫 𝐃𝐞𝐜𝐫𝐞𝐚𝐬𝐞 𝐟𝐨𝐫 𝐰𝐡𝐚𝐭 𝐜𝐨𝐧𝐭𝐚𝐢𝐧𝐬 :
The Laparotomy Sponge Market is witnessing growth due to rising surgical procedures globally, especially in abdominal surgeries, which require effective blood absorption and visibility. Demand is driven by increased healthcare spending, advancements in sponge materials, and a focus on patient safety, as sponges with RFID or barcode tracking help reduce the risk of retained surgical items. The aging…
Laparotomy Sponges Market to register a CAGR of 10.9% between 2024-2032
The global demand for the laparotomy sponges market size is expected to experience significant growth, with its market size projected to reach approximately USD 4.24 billion by 2032, up from USD 1.67 billion in 2023. This growth represents a robust compound annual growth rate (CAGR) of 10.9% over the study period from 2024 to 2032. The increase in market size can be attributed to several factors, including the rising incidence…
Surgical Sponges Unveiled: Trends and Insights in Laparotomy Sponge Market
The Laparotomy Sponges Market is witnessing significant growth, driven by the rising demand for surgical procedures and the increasing emphasis on patient safety and infection control. These highly absorbent and sterile sponges are indispensable tools in abdominal surgeries, aiding in the absorption of fluids and blood, as well as providing a clear surgical field. Frequently Asked Questions (FAQs) surrounding the Laparotomy Sponges Market often revolve around their composition, sterilization methods,…
Laparotomy Sponges Market 2023 Size, Share, Industry, Forecast to 2030
The Laparotomy Sponges Market 2023 Report makes available the current and forthcoming technical and financial details of the industry. It is one of the most comprehensive and important additions to the Prudent Markets archive of market research studies. It offers detailed research and analysis of key aspects of the global Laparotomy Sponges market. This report explores all the key factors affecting the growth of the global Laparotomy Sponges market, including…
Laparotomy Sponges Market Pegged for Robust Expansion by 2027
This Laparotomy Sponges market report provides the best business insight and understanding to help key players stay ahead of the competition. It also detects emerging trends and forecasts future market numbers, trends, and characteristics. This Laparotomy Sponges Market Report offers the most effective action strategies for dealing with the current market situation and establishing a marketplace. It also helps to improve and enhance the company's position. This Laparotomy Sponges market…
Laparotomy Sponge Market Trends, Outlook, and Opportunity Analysis, 2018-2026
Laparotomy sponge also called as laparotomy pads or absorbent pads used for hemorrhage evacuation during the abdominal surgeries. Laparotomy sponges are used for absorbing viscera around the surgical site to obtain clear view of internal organs during surgery. Laparotomy sponge are mainly made of 100% cotton to maximize absorbency. Other materials used for laparotomy sponges are 100% rayon chamoix, rayon–polypropylene chamoix, and 20/80 nylon–polyester composite towels. Laparotomy sponge are prone…
