Press release
AI in Clinical Trials Market to Reach US$ 5.06 Billion by 2033 as Pharmaceutical Companies Accelerate AI-Driven Drug Development | Key Players: Medidata, IQVIA, Saama Technologies, Phesi, Deep6.ai
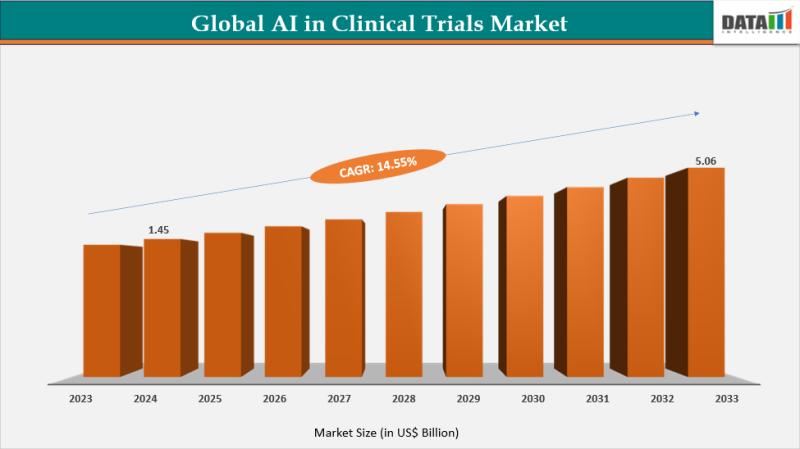
The global AI in Clinical Trials market reached US$ 1.45 billion in 2024, increasing from US$ 1.27 billion in 2023, and is projected to reach US$ 5.06 billion by 2033, growing at a CAGR of 14.55% during the forecast period (2025-2033). The market is expanding rapidly as pharmaceutical and biotechnology companies increasingly adopt artificial intelligence technologies to improve clinical trial efficiency, reduce operational costs, and accelerate drug development timelines.The integration of AI-driven analytics, machine learning, and predictive modeling is transforming the clinical trial process by enabling faster patient recruitment, improving trial design, and enhancing data management. Growing availability of large healthcare datasets-including electronic health records (EHRs), genomic databases, and real-world evidence is enabling AI algorithms to identify suitable patient populations, predict trial success rates, and optimize protocol design.
Request Executive Sample report today: (corporate email gets priority access):-https://www.datamintelligence.com/download-sample/ai-in-clinical-trials-market?Juli
Additionally, the rising prevalence of complex and chronic diseases such as cancer, cardiovascular disorders, and neurological conditions is increasing the need for more efficient clinical trial strategies. AI-powered platforms help researchers analyze large datasets, detect patterns, and support precision medicine approaches, improving the accuracy and success rates of clinical studies.
Strategic collaborations among AI technology providers, contract research organizations (CROs), and life sciences companies are further accelerating innovation in AI-enabled clinical research. These partnerships enable the integration of advanced analytics, cloud computing, and automated trial management systems, improving overall trial productivity.
Key Industry Developments:
✅ September 2025 - PhaseV and Bioforum Strategic Partnership
PhaseV partnered with Bioforum to enhance clinical trial efficiency using advanced artificial intelligence and machine learning technologies. The collaboration focuses on optimizing clinical study design, improving data analysis, and enabling more accurate prediction of clinical trial outcomes.
✅ June 2025 - Advarra Launches Council for Responsible AI in Clinical Trials
Advarra introduced the Council for Responsible Use of AI in Clinical Trials to guide ethical implementation and governance of AI technologies in clinical research. The initiative aims to accelerate innovation while ensuring regulatory compliance and responsible AI adoption across global clinical trials.
✅ April 2025 - ConcertAI and Bayer Multi-Year AI Collaboration
ConcertAI signed a multi-year agreement with Bayer to utilize AI and machine-learning insights for precision oncology clinical trials. The collaboration focuses on leveraging real-world data and advanced analytics to accelerate drug development and improve patient selection for cancer studies.
✅ March 2025 - Suvoda Launches AI Assistant for Trial Management
Suvoda introduced Sofia, an AI-powered assistant designed to support clinical trial teams by providing real-time insights and simplifying access to trial data. The platform helps streamline operational workflows, reduce manual tasks, and improve decision-making across clinical trial management.
✅ December 2024 - ConcertAI and NeoGenomics Launch AI Research Platform
ConcertAI partnered with NeoGenomics to launch CTO-H, an AI-driven platform for clinical trial analytics, biomarker research, and operational optimization. The solution integrates large-scale patient data and biomarker insights to support precision clinical development and improve trial efficiency.
✅ June 2024 - Lokavant Introduces AI-Driven Clinical Trial Feasibility Platform
Lokavant launched SpectrumTM, an AI-powered clinical trial feasibility and performance optimization solution. The platform enables pharmaceutical companies and CROs to predict trial timelines, manage risks, and improve operational planning across the clinical development lifecycle.
Competitive Landscape and Industry Partnerships:
The global AI in Clinical Trials market is characterized by a combination of established clinical research technology providers and specialized AI-driven healthcare analytics companies. Key companies operating in the market include Medidata, IQVIA, Saama Technologies, Phesi, Euretos, Median Technologies, Innoplexus, Deep6.ai, AiCure, Antidote Technologies, and Mendel AI. These organizations provide advanced artificial intelligence solutions for clinical trial design, patient recruitment, protocol optimization, data management, and predictive analytics across pharmaceutical and biotechnology research.
Major players such as Medidata and IQVIA leverage large-scale clinical data platforms and cloud-based technologies to support end-to-end clinical development processes, including trial planning, patient matching, and real-time monitoring. Their strong partnerships with pharmaceutical companies and contract research organizations (CROs) enable them to deliver integrated AI-driven clinical trial management solutions globally.
Specialized AI companies including Deep6.ai, Innoplexus, Median Technologies, and Saama Technologies focus on advanced analytics, real-world data integration, and predictive modeling to improve trial efficiency and reduce delays in patient recruitment and site selection. Meanwhile, firms such as AiCure, Antidote Technologies, and Mendel AI concentrate on patient engagement, remote monitoring, and AI-driven patient-trial matching platforms that help accelerate enrollment and improve trial outcomes.
Strategic collaborations between AI technology providers, pharmaceutical companies, CROs, and healthcare institutions are increasingly shaping the competitive landscape. These partnerships support the development of next-generation clinical trial platforms that integrate real-world evidence, machine learning algorithms, and large healthcare datasets to enhance trial design, reduce operational costs, and accelerate regulatory approvals. As AI adoption continues to expand in clinical research, companies are expected to strengthen partnerships, invest in advanced data infrastructure, and develop more sophisticated analytics capabilities to maintain a competitive advantage.
Investment Outlook
The AI in Clinical Trials market presents strong investment potential as pharmaceutical and biotechnology companies increasingly adopt artificial intelligence to accelerate drug development and reduce the high costs associated with traditional clinical trials. Investors are showing growing interest in this sector due to AI's ability to improve patient recruitment, optimize trial design, and analyze complex healthcare datasets more efficiently. These capabilities significantly reduce development timelines and improve the likelihood of successful trial outcomes.
Market projections indicate substantial long-term growth opportunities as the pharmaceutical industry continues integrating machine learning, predictive analytics, and natural language processing into clinical research workflows. The increasing complexity of clinical trials and the rapid growth of healthcare data are encouraging companies to invest in advanced AI platforms capable of managing large-scale clinical datasets and improving decision-making during trials.
Investment momentum is also driven by the potential cost savings and operational efficiencies offered by AI technologies. AI-enabled clinical trial solutions can significantly reduce overall trial costs by improving patient selection, minimizing recruitment delays, and enhancing data analysis processes. These advantages are encouraging venture capital firms, pharmaceutical companies, and technology investors to allocate funding toward AI-powered clinical research platforms and digital health solutions.
Purchase Corporate License | Market Intelligence:-https://www.datamintelligence.com/buy-now-page?report=ai-in-clinical-trials-market?Juli
Market Drivers:
Rising R&D Costs and Long Clinical Trial Timelines Driving AI Adoption
The rising research and development (R&D) costs and extended timelines associated with traditional clinical trials are significantly driving the growth of the AI in Clinical Trials market. Pharmaceutical and biotechnology companies are increasingly seeking advanced technologies that can accelerate patient recruitment, streamline trial operations, and improve data analysis efficiency. Artificial intelligence offers solutions such as predictive patient enrollment, automated monitoring, adaptive trial design, and real-time analytics, which help reduce operational costs while shortening development timelines.
According to the U.S. Food and Drug Administration (FDA), it takes, on average, 10 to 15 years and approximately $2.6 billion to bring a new medicine to market. This lengthy and costly process underscores the need for innovative solutions to expedite drug development and reduce expenses. Additionally, regulatory bodies are increasingly recognizing the potential of AI in clinical research. Regulatory acceptance of AI-assisted analytics and digital trial methodologies is supporting broader adoption across pharmaceutical companies, contract research organizations (CROs), and research institutions. As a result, AI-powered predictive analytics and real-world data integration are transforming clinical trial management, enabling more precise decision-making and accelerating the delivery of innovative therapies to patients.
Market Restraints
Shortage of Skilled Professionals Limiting AI Integration
The shortage of skilled professionals with expertise in both artificial intelligence and clinical research remains a major restraint for the AI in Clinical Trials market. While pharmaceutical companies and CROs are rapidly adopting AI technologies to improve clinical trial efficiency, the availability of qualified professionals capable of developing, implementing, and managing these advanced systems remains limited.
Effective deployment of AI solutions requires specialized knowledge in machine learning, data science, clinical trial design, and regulatory compliance. However, the demand for such multidisciplinary expertise is growing faster than the available talent pool. This shortage makes it difficult for organizations to fully integrate AI technologies into clinical development processes.
Regional Insights:
North America
North America holds the largest share of the AI in Clinical Trials market, accounting for approximately 43% of the global market. The region's dominance is driven by the presence of major pharmaceutical companies, advanced healthcare infrastructure, and strong adoption of artificial intelligence technologies in clinical research. The United States leads the region due to significant investments in drug development, strong presence of contract research organizations (CROs), and supportive regulatory frameworks encouraging digital innovation in clinical trials. Additionally, collaborations between technology companies, research institutions, and pharmaceutical firms are accelerating the implementation of AI-driven clinical trial solutions.
Europe
Europe represents around 27% of the global AI in Clinical Trials market. The region benefits from strong regulatory support, a well-established pharmaceutical industry, and growing investments in AI-driven healthcare technologies. Countries such as Germany, the United Kingdom, and France are actively integrating AI into clinical research to improve trial efficiency and patient recruitment. European research institutions and biotechnology companies are increasingly partnering with AI solution providers to develop advanced data analytics and predictive modeling platforms for clinical studies.
Asia-Pacific
Asia-Pacific is the fastest-growing region, accounting for approximately 20% of the global market. Rapid expansion of the pharmaceutical and biotechnology sectors, increasing clinical trial activities, and rising adoption of digital health technologies are driving growth in this region. Countries such as China, Japan, South Korea, and India are witnessing increasing investments in AI-based research tools and clinical trial management platforms. Additionally, the large patient population and lower trial operational costs are encouraging global pharmaceutical companies to conduct clinical trials in Asia-Pacific.
Request Custom Intelligence Report:-https://www.datamintelligence.com/customize/ai-in-clinical-trials-market?Juli
Market Segmentation Analysis:
By Offering
Software - 62.4%
Software solutions dominate the AI in Clinical Trials market as pharmaceutical companies increasingly adopt AI-powered platforms for patient recruitment, predictive analytics, protocol design, and data management. These platforms enable faster trial execution, improved accuracy in patient matching, and enhanced decision-making capabilities.
Services - 37.6%
Services include consulting, integration, data management, and AI model customization provided by technology vendors and contract research organizations. The demand for services is growing as companies require technical expertise to implement AI tools and integrate them into existing clinical trial workflows.
By Trial Phase
Phase I - 18.7%
AI tools are increasingly used in early-stage trials to identify suitable participants and predict safety outcomes, helping reduce risks associated with initial human testing.
Phase II - 34.1%
Phase II trials account for a significant share as AI assists in optimizing dosing strategies, monitoring treatment responses, and improving patient selection criteria.
Phase III - 47.2%
Phase III holds the largest share due to the large-scale patient populations involved. AI technologies help manage vast datasets, monitor patient adherence, and improve trial efficiency while reducing operational costs.
By Indication
Oncology - 39.6%
Oncology dominates the market due to the high number of cancer clinical trials globally. AI helps identify eligible patients using genomic data, optimize trial protocols, and predict treatment outcomes.
Central Nervous System (CNS) Disorders - 24.3%
AI is increasingly applied in neurological research for diseases such as Alzheimer's and Parkinson's, where patient identification and biomarker analysis are complex.
Cardiovascular (CVS) Diseases - 18.5%
AI tools support cardiovascular clinical trials by analyzing patient health data, predicting adverse events, and improving monitoring during trials.
Other Indications - 17.6%
Other indications include infectious diseases, metabolic disorders, and rare diseases where AI helps streamline trial design and patient recruitment.
By End-User
Pharmaceutical & Biotechnology Companies - 52.8%
These companies represent the largest user group as they increasingly invest in AI technologies to reduce drug development timelines and R&D costs.
Contract Research Organizations (CROs) - 34.7%
CROs are adopting AI-driven solutions to improve clinical trial management, data analysis, and patient recruitment services offered to pharmaceutical companies.
Other End Users - 12.5%
This segment includes academic research institutions, hospitals, and government research organizations that use AI platforms for clinical research and experimental drug studies.
📌 Request for 2 Days FREE Trial Access: https://www.datamintelligence.com/reports-subscription
☛ Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
☛ Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTWg?Juli
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release AI in Clinical Trials Market to Reach US$ 5.06 Billion by 2033 as Pharmaceutical Companies Accelerate AI-Driven Drug Development | Key Players: Medidata, IQVIA, Saama Technologies, Phesi, Deep6.ai here
News-ID: 4414965 • Views: …
More Releases from DataM intelligence 4 Market Research LLP
United States Barrier Resins Market 2026 | Growth Drivers, Trends & Market Forec …
Market Size and Growth 2026
Barrier Resins Market size reached US$ 20.32 billion in 2024 and is expected to reach US$ 32.12 billion by 2032, growing with a CAGR of 5.89% during the forecast period 2025-2032.
DataM Intelligence has released a new research report titled Barrier Resins Market Size 2026 The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of…
United States Maleic Anhydride Market 2026 | Growth Drivers, Trends & Market For …
Market Size and Growth 2026
Global Maleic Anhydride market size reached US$ 4.60 billion in 2024 and is expected to reach US$ 6.83 billion by 2032, growing with a CAGR of 5.06% during the forecast period 2026-2033, according to DataM Intelligence report.
DataM Intelligence has released a new research report titled Maleic Anhydride Market Size 2026 The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR…
United States Ostomy Care Devices Market 2026 | Growth Drivers, Trends & Market …
Market Size and Growth 2026
Global Ostomy Care Devices Market size reached US$ 3.54 billion in 2024 and is expected to reach US$ 7.51 billion by 2033, growing at a CAGR of 7.8% during the forecast period 2026-2033.
DataM Intelligence has released a new research report titled Ostomy Care Devices Market Size 2026 The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the…

Sapphire Glass Market 2026: The $1.69B Opportunity Powering Next-Gen Electronics …
Market Size and Growth 2026
Global Sapphire Glass Market size reached US$ 951.22 million in 2024 and is expected to reach US$ 1,697.74 million by 2032, growing with a CAGR of 7.51% during the forecast period 2026-2033.
DataM Intelligence has released a new research report titled Sapphire Glass Market Size 2026 The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue…
More Releases for Trial
Clinical Trial Investigative Site Network Market Clinical Trial Investigative Si …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trial Investigative Site Network Market - (By Therapeutic Areas (Oncology, Cardiology, CNS, Pain Management, Endocrine, Others), By Phase (Phase I, Phase II, Phase III, Phase IV), By End-use (Sponsor, CRO)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic, the Global Clinical Trial Investigative Site Network Market…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Transformative Trends Impacting the Electronic Trial Master File (eTMF) Systems …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the Electronic Trial Master File (eTMF) Systems Market Size By 2025?
The market size of the electronic trial master file (eTMF) systems has experienced fast growth over recent years. The market is projected to increase from $1.36 billion in 2024 to $1.55 billion in 2025, with…
Clinical Trial Management System
According to a new market report published by Persistence Market Research “Global Market Study on Clinical Trial Management System: Asia to Witness Highest Growth by 2019” the global clinical trial management system market was valued at USD 844.0 million in 2013 and is expected to grow at a CAGR of 14% from 2014 to 2019, to reach an estimated value of USD 1,848.5 million in 2019.
Request Report TOC @ https://www.persistencemarketresearch.com/methodology/3017
…
Clinical Trial Logistics
Clinical Trial Logistics
16th to 17th May 2011, Marriott Regents Park, London, United Kingdom.
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical trials…
Clinical Trial Logistics
Announcing SMi's 5th annual…
Clinical Trial Logistics conference
16th and 17th May 2011, Central London, UK
www.smi-online.co.uk/2011logistics-london6.asp
It currently costs just over £500 million ($800 million) to bring a new chemical to market and development timelines continue to fall in the 10-15 year range. A key reason for high R&D costs is due to logistical failures including failure to recruit patients on time. A way to avoid this is to move clinical…