Press release
Chronic Immune Thrombocytopenia Treatment Market Set for Steady Growth to USD 4.0 Billion by 2035, Led by North America's 48% Market Share | Key Players - Amgen, Novartis, Bristol Myers Squibb
Leander Texas -The Chronic immune thrombocytopenia treatment Market is estimated to be valued at USD 2.3 billion in 2025 and is projected to reach USD 4.0 billion by 2035, registering a compound annual growth rate (CAGR) of 5.7% over the forecast period.
The market growth is driven by U.S. FDA approval of Sanofi's Wayrilz (rilzabrutinib) for chronic ITP, offering a new therapeutic option, and Japan's regulatory approval of argenx's VYVGART for ITP, expanding treatment access and boosting adoption in key markets.
Download your exclusive sample report today: (corporate email gets priority access):https://www.datamintelligence.com/download-sample/chronic-immune-thrombocytopenia-market?prtk
Key Industry Development-
United States: Key Industry Developments
✅ December 2025: Novartis expanded access to its next-generation TPO-RA drug Promacta (eltrombopag) through enhanced reimbursement programs, boosting treatment adherence for chronic ITP patients amid rising diagnoses linked to autoimmune conditions.
✅ October 2025: Amgen reported positive Phase III results for a subcutaneous romiplostim formulation, reducing administration burden and improving platelet response rates by 25% in refractory chronic ITP cases.
✅ August 2025: FDA granted accelerated approval to Dova Pharmaceuticals' avatrombopag variant for chronic ITP, targeting splenectomized patients with improved oral bioavailability and fewer gastrointestinal side effects.
Japan: Key Industry Developments
✅ November 2025: Chugai Pharmaceutical launched an optimized rituximab biosimilar for first-line chronic ITP therapy, achieving 40% better cost-efficiency while matching originator efficacy in PMDA-approved trials.
✅ September 2025: PMDA approved subsidies for fostamatinib (Tavalisse) import and localized production, addressing unmet needs in adult chronic ITP with better hemorrhage control in high-risk populations.
✅ July 2025: Kyowa Kirin introduced a novel FcRn inhibitor for refractory chronic ITP, demonstrating sustained platelet elevation in Japanese patients through enhanced antibody clearance mechanisms.
Strategic Mergers and Acquisitions:
✅ In January 2025, Johnson & Johnson completed its acquisition of Intra-Cellular Therapies for approximately US$ 14.6 billion. While primarily focused on neuroscience, the acquisition strengthens Johnson & Johnson's immunology and specialty medicine pipeline capabilities, indirectly reinforcing its broader autoimmune and hematology portfolio relevant to chronic immune thrombocytopenia treatment innovation.
✅ In March 2025, Novartis AG finalized the acquisition of MorphoSys AG in a transaction valued at approximately US$ 2.9 billion. The deal enhances Novartis' immunology and hematology pipeline assets, expanding monoclonal antibody expertise and strengthening its strategic positioning in immune-mediated and rare blood disorder markets, including chronic ITP.
✅ In April 2025, Bristol Myers Squibb completed its acquisition of Mirati Therapeutics for approximately US$ 5.8 billion (including milestone payments). Although oncology-focused, the acquisition expands Bristol Myers Squibb's immunomodulatory research capabilities, supporting long-term growth in immune-driven therapeutic areas such as chronic immune thrombocytopenia.
✅ In June 2025, Takeda Pharmaceutical Company acquired select immunology pipeline assets from smaller biotechnology firms in a publicly disclosed strategic expansion aimed at strengthening rare hematology treatments. The transaction supports Takeda's global rare disease leadership, reinforcing its commercial and research footprint in immune-mediated platelet disorders including chronic ITP.
Major Players:-
AbbVie Inc. | Amgen Inc. | Pfizer, Inc. | Samsung Bioepis Co., Ltd. | Sanofi S.A. | Novartis International AG | Johnson & Johnson Services, Inc. | Merck & Co., Inc. | Bristol Myers Squibb Inc. | F. Hoffmann-La Roche Ltd.
1. Amgen Inc.
Amgen holds an estimated 18-22% share of the chronic thrombocytopenia market, leading through its TPO-receptor agonist Nplate® (romiplostim) which stimulates platelet production. Its strong global reach, robust clinical data, and early-entry biologic position the company as a dominant innovator in long-term ITP management.
2. Novartis International AG
Novartis controls roughly 14-18% of the market with agents like Promacta/Revolade® (eltrombopag) and emerging therapies. Its focus on oral TPO-RAs, broad regulatory approvals, and active autoimmune disease pipeline supports sustained leadership, especially in chronic ITP and refractory patient populations worldwide.
3. Sanofi S.A.
Sanofi is advancing its position through rilzabrutinib (Wayrilz®), an oral BTK inhibitor recently approved for chronic ITP that modulates immune response. While specific share isn't published, Sanofi's strong immunology footprint and rare disease focus help expand treatment options beyond traditional TPO-RAs.
4. Pfizer, Inc.
Pfizer leverages its broad immunomodulatory portfolio and partnerships to support development of novel agents for refractory ITP and related hematologic conditions. Although its exact market share isn't widely disclosed, Pfizer's global R&D and commercialization capabilities enhance competitive presence and long-term growth potential.
5. F. Hoffmann-La Roche Ltd.
Roche plays a strategic role by applying monoclonal antibody and immunotherapy expertise to blood disorders, including ITP. Its established biologics and strong global infrastructure contribute to sustained relevance in the market, particularly in combination or second-line therapeutic strategies.
"Secure your 30% discount - get this report before the offer expires."
:https://www.datamintelligence.com/buy-now-page?report=chronic-immune-thrombocytopenia-market?prtk (Purchase 2 or more Repots and get 50% Discount)
Market Segmenatation-
Treatment Type Segmentation:
The chronic ITP treatment market is segmented by the type of therapeutic approach used, including drug-based therapies such as thrombopoietin receptor agonists (TPO-RAs), corticosteroids, immunoglobulins, fostamatinib, and other agents, as well as procedural approaches like splenectomy and platelet transfusions. In the United States, drug-based treatments dominate due to early adoption of advanced biologics and broader regulatory approvals, and emerging therapies like rilzabrutinib are expected to expand options following recent FDA approvals. In Japan, similar therapeutic categories are present but growth is propelled by targeted therapies such as TPO-RAs and novel agents like VYVGART which was approved specifically for ITP, reflecting a steady uptake of modern pharmacological interventions tailored to chronic disease management.
End-User Segmentation:
The market is also categorized by who delivers care: hospitals, ambulatory surgery centres, emergency centres, and others. Across both the US and Japan, hospitals represent the largest end-user segment because they have the specialized hematology departments, diagnostic tools, and clinical support systems needed to manage complex chronic ITP cases. This hospital-centric model supports both routine management and the administration of advanced infusions and biologic therapies, ensuring patients receive comprehensive monitoring and treatment adjustments as needed.
Market Drivers:-
• Rising Adoption of Advanced Thrombopoietin Receptor Agonists (TPO-RAs): In the U.S., strong uptake of TPO-RAs such as Promacta (Novartis) and Nplate (Amgen) is driving treatment revenues due to their proven long-term platelet response. In Japan, expanded indications and reimbursement approvals for TPO-RAs have improved access, particularly among elderly ITP patients, accelerating prescription volumes.
• Regulatory Approvals of Novel Therapies: The U.S. FDA's approval of Tavalisse for chronic ITP has expanded second-line treatment options, supporting market growth. In Japan, the Ministry of Health, Labour and Welfare (MHLW) has approved newer immunomodulatory agents and biosimilars, enhancing competition and broadening therapeutic availability.
• Growing Prevalence of Autoimmune Disorders and Aging Population: The U.S. reports increasing autoimmune disease incidence, contributing to higher diagnosed ITP cases annually. In Japan, a rapidly aging demographic-where ITP prevalence is higher among individuals aged 60+-is significantly increasing demand for long-term platelet management therapies.
• Strategic Collaborations and Clinical Development Initiatives: U.S.-based biotechnology firms are advancing FcRn inhibitors and BTK inhibitors for ITP through clinical partnerships and pipeline expansion. Japanese pharmaceutical companies are collaborating with global biotech firms to conduct local Phase III trials, strengthening domestic commercialization strategies and accelerating innovative drug entry.
• Favorable Reimbursement and Rare Disease Support Programs: In the U.S., chronic ITP benefits from orphan drug incentives and premium pricing structures, improving manufacturer margins. Japan's national health insurance system ensures broad reimbursement coverage for high-cost biologics, supporting patient access and sustaining consistent treatment adoption rates.
Regional insights:-
North America: 48% - Largest share, led by the United States due to advanced healthcare infrastructure, strong R&D, supportive reimbursement policies, and early adoption of novel therapies (chronic ITP dominates overall ITP care in the region). The US accounts for the majority of this share with robust clinical pipelines and high treatment uptake.
Asia Pacific: 20% - Fastest-growing region, driven by expanding healthcare access, increasing disease awareness, rising healthcare expenditure, and growing demand in key countries including Japan. Japan itself represents a significant portion of the APAC regional share given its well-developed hematology treatment landscape, though exact Japan-only share isn't separately published in the major regional breakdown reports.
Europe: 25% - Steady share, supported by high treatment awareness, established clinical guidelines, and structured national healthcare systems that facilitate ITP therapy uptake (including chronic ITP management).
Get Customization in the report as per your requirements:https://www.datamintelligence.com/customize/chronic-immune-thrombocytopenia-market?prtk
Request for 2 Days FREE Trial Access:
https://www.datamintelligence.com/reports-subscription?pratik
✅ Competitive Landscape
✅ Technology Roadmap Analysis
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Consumer Behavior & Demand Analysis
✅ Import-Export Data Monitoring
✅ Live Market & Pricing Trends
Have a look at our Subscription Dashboard:
https://www.youtube.com/watch?v=x5oEiqEqTWg
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About DataM Intelligence
DataM Intelligence is a renowned provider of market research, delivering deep insightsthrough pricing analysis, market share breakdowns, and competitive intelligence. Thecompany specializes in strategic reports that guide businesses in high-growth sectors suchas nutraceuticals and AI-driven health innovations.
To find out more, visit https://www.datamintelligence.com/ or follow us on Twitter,LinkedIn and Facebook.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Chronic Immune Thrombocytopenia Treatment Market Set for Steady Growth to USD 4.0 Billion by 2035, Led by North America's 48% Market Share | Key Players - Amgen, Novartis, Bristol Myers Squibb here
News-ID: 4406010 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Autonomous/Self-Driving Cars Market to Reach US$ 790.82 Billion by 2032 at 28.60 …
The Autonomous/Self-Driving Cars Market reached US$ 126.27 billion in 2024 and is projected to reach US$ 790.82 billion by 2032, growing at a CAGR of 28.60 percent during the forecast period 2025 to 2032.
Market growth is driven by rapid advancements in artificial intelligence, LiDAR, radar, camera based vision systems, and high performance onboard computing platforms that enable vehicles to operate with minimal or no human intervention. Increasing demand for safer…
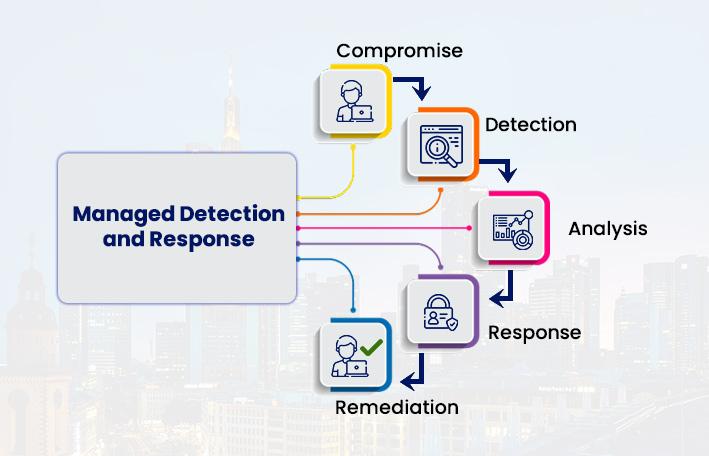
Managed Detection and Response Market Set for Explosive Growth to US$ 10.41 Bill …
The Managed Detection and Response (MDR) Market reached US$ 2.27 billion in 2025 and is expected to reach US$ 10.41 billion by 2034, growing at a robust CAGR of 17.7% during the forecast period 2026-2034.
Market growth is driven by escalating cyber threats, rising demand for 24/7 proactive security monitoring, and the complexity of modern attack surfaces including ransomware and cloud environments. Advancements in AI-powered threat detection, expanding adoption among SMEs…

Orthopedic Bone Cement Market Future Scope (2026-2033): Hospitals & Surgical Cen …
DataM Intelligence has published a new research report on "Orthopedic Bone Cement Market Size 2025". The report explores comprehensive and insightful Information about various key factors like Regional Growth, Segmentation, CAGR, Business Revenue Status of Top Key Players and Drivers. The purpose of this report is to provide a telescopic view of the current market size by value and volume, opportunities, and development status.
Get a Sample PDF Of This Report…
Cerebral Somatic Oximeters Market Future Scope Report (2026-2033): Growth Across …
DataM Intelligence has published a new research report on "Cerebral Somatic Oximeters Market Size 2025". The report explores comprehensive and insightful Information about various key factors like Regional Growth, Segmentation, CAGR, Business Revenue Status of Top Key Players and Drivers. The purpose of this report is to provide a telescopic view of the current market size by value and volume, opportunities, and development status.
Get a Sample PDF Of This Report…
More Releases for ITP
ITP Therapeutics Market to Reach USD 1.28 Billion by 2035
Is the ITP Therapeutics Market Ready to Address the Rising Burden of Immune Thrombocytopenia Worldwide?
ITP Therapeutics Market: Comprehensive Global Insights and Forecast
The ITP Therapeutics Market is gaining increasing attention within the global hematology and rare disease landscape as awareness of immune thrombocytopenia (ITP) improves and treatment options continue to evolve. ITP is an autoimmune disorder characterized by low platelet counts, leading to an increased risk of bruising, bleeding, and, in…
Immune Thrombocytopenia ITP Market Booming with New Therapeutic Approaches | Coh …
📊 Immune Thrombocytopenia ITP Market Snapshot 2025-2032
Immune Thrombocytopenia (ITP) Market is expected to reach USD 3.1 Billion by 2032, growing at a CAGR of 4.9% during 2025 to 2032.
Coherent Market Insights unveils its latest data-driven Immune Thrombocytopenia ITP Market Research Report featuring transformative insights into the U.S. Immune Thrombocytopenia ITP Market. Spanning from 2025 to 2032, this report offers granular forecasts, payer-provider models, and supply chain intelligence. As the U.S.…
ITP Therapeutics Market to Reach US$ 1.0 Billion by 2032: Fact.MR
The global Idiopathic Thrombocytopenic Purpura (ITP) therapeutics market has experienced notable growth in recent years, driven by factors such as increased awareness of autoimmune diseases, advancements in targeted therapies, and a rising prevalence of ITP among the geriatric population. According to Fact.MR, the market was valued at approximately US$ 587.0 million in 2021 and is projected to reach US$ 1.0 billion by 2032, reflecting a compound annual growth rate (CAGR)…
Exploring the Idiopathic Thrombocytopenic Purpura (ITP) Treatment Drugs Market
Idiopathic Thrombocytopenic Purpura (ITP), also known as Immune Thrombocytopenic Purpura, is an autoimmune disorder characterized by abnormally low platelet levels, leading to an increased risk of bruising and bleeding. The condition can affect both children and adults, with treatment options tailored to individual cases based on severity and response to therapy. The growing prevalence of ITP and advancements in treatment approaches have fueled the demand for effective therapeutic drugs, making…
Idiopathic Thrombocytopenic Purpura (ITP) Therapeutics Market Analysis and Futur …
The world of the idiopathic thrombocytopenic purpura (itp) therapeutics market is a complex and ever-evolving landscape, shaped by consumer demands and technological advancements. In this report, we delve into the depths of this market to provide a profound and comprehensive analysis, catering to a diverse audience that includes manufacturers, suppliers, distributors, and investors. Our primary goal is to empower industry stakeholders with invaluable insights to make informed decisions in a…
Idiopathic Thrombocytopenic Purpura Market - Defy the Odds: Transforming Lives t …
Newark, New Castle, USA: The "Idiopathic Thrombocytopenic Purpura Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors.
Idiopathic Thrombocytopenic Purpura Market: https://www.growthplusreports.com/report/idiopathic-thrombocytopenic-purpura-market/8875
This latest report researches the industry structure,…
