Press release
Diffuse Large B-Cell Lymphoma Clinical Trial Pipeline Accelerates as 70+ Pharma Companies Rigorously Develop Drugs for Market Entry | DelveInsight
DelveInsight's, "Diffuse Large B-Cell Lymphoma Pipeline Insights 2026" report provides comprehensive insights about 70+ companies and 75+ pipeline drugs in Diffuse Large B-Cell Lymphoma pipeline landscape. It covers the Diffuse Large B-Cell Lymphoma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Diffuse Large B-Cell Lymphoma therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive Diffuse Large B-Cell Lymphoma pipeline products in this space.Download DelveInsight's comprehensive Diffuse Large B-Cell Lymphoma Pipeline Report to explore emerging therapies @ [https://www.delveinsight.com/sample-request/diffuse-large-b-cell-lymphoma-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Key Takeaways from the Diffuse Large B-Cell Lymphoma Pipeline Report
* On February 20, 2026, Merck Sharp & Dohme LLC conducted a phase 3 study is to evaluate if zilovertamab vedotin with standard treatment can help people live longer without the cancer growing or spreading than people who receive standard treatment alone.
* On February 13, 2026, Tanabe Pharma Corporation initiated a phase I/II Open-Label Study of MT-2111 in Patients With Relapsed/Refractory DLBCL. To investigate the safety, tolerability, and pharmacokinetics of MT-2111 monotherapy in patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL). In addition, the dose to be used in the Phase II part will be confirmed.
* On February 06, 2026- Hoffmann-La Roche announced a Phase III, randomized, double-blind, placebo-controlled study will compare the efficacy, safety, and pharmacokinetics of polatuzumab vedotin plus R-CHP versus R-CHOP in participants with previously untreated diffuse large B-cell lymphoma (DLBCL).
* DelveInsight's Diffuse Large B-Cell Lymphoma pipeline report depicts a robust space with 70+ active players working to develop 75+ pipeline therapies for Diffuse Large B-Cell Lymphoma treatment.
* The leading Diffuse Large B-Cell Lymphoma Companies such as Miltenyi Biomedicine, Adicet Bio, VelosBio, Novartis Pharmaceuticals, Sanofi, Eisai Co, Schrodinger, Sana Biotechnology, Ranok Therapeutics, Monte Rosa Therapeutic, Otsuka Pharmaceutical, OncoNano Medicine, Regeneron Pharmaceuticals, Hoffmann-La Roche, Celgene, Nurix Therapeutics, NovalGen, Nektar Therapeutics, Genentech, CSPC ZhongQi Pharmaceutical Technology and others.
* Promising Diffuse Large B-Cell Lymphoma Therapies such as Ibrutinib, Bendamustine, Rituximab, R-miniCHOP, Selinexor, Tafasitamab, Lenalidomide, and others.
Access DelveInsight's in-depth pipeline Analysis for a closer look at promising breakthroughs @ Diffuse Large B-Cell Lymphoma Clinical Trials and Studies [https://www.delveinsight.com/sample-request/diffuse-large-b-cell-lymphoma-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Diffuse Large B-Cell Lymphoma Emerging Drugs Profile
* Brentuximab vedotin: Pfizer
Brentuximab vedotin (Adcetris) is an anti-neoplastic agent. It is indicated for the treatment of patients with Classical Hodgkin lymphoma (HL) after failure of autologous hematopoietic stem cell transplantation (auto-HSCT) or after failure of at least two prior multi-agent chemotherapy regimens in patients who are not auto-HSCT candidates. Adcetris is also indicated for the treatment of adult patients with CD30+ Hodgkin lymphoma at increased risk of relapse or progression following autologous stem cell transplant (ASCT), also indicated for the treatment of adult patients with relapsed or refractory systemic anaplastic large cell lymphoma, for the treatment of adult patients with primary cutaneous anaplastic large cell lymphoma (pcALCL) and CD30-expressing mycosis fungoides (MF) who have received prior systemic therapy, for the treatment of adult patients with relapsed or refractory CD30+ cutaneous T- cell lymphoma (CTCL) after at least 1 prior systemic therapy. Adcetris is indicated for the first-line pediatric treatment for CD30-positive Hodgkin lymphoma. Currently, the drug is in Phase III stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
* THOR-707: Sanofi
THOR-707 is a precisely PEGylated version of IL-2, where the PEG chain is attached to a novel amino acid inserted at a location on IL-2 that prevents it from engaging the alpha-receptor and binding to immune receptors that cause drug toxicities (IL-2R-alpha, CD25). The engineered IL-2 retains near-native binding to the beta-gamma receptors that selectively expand tumor-killing T effector cells and Natural Killer (NK) cells without the alpha-mediated immunosuppressive effects of regulatory T cells or eosinophil-mediated vascular leak syndrome. Currently, the drug is in Phase II stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
* Abexinostat: Xynomic Pharmaceuticals
Abexinostat (Xynomic Pharmaceuticals) is a broad histone deacetylase (HDAC) inhibitor. HDAC enzymes (also known as lysine deacetylase) cleave acetyl groups from N-acetyl lysine amino acids on a histone. HDAC inhibition leads to highly acetylated histones and chromatin reshaping. In addition to altering histone acetylation, HDAC inhibitors can also influence the degree of acetylation on non-histone proteins, increasing or repressing their activity. Currently, the drug is in Phase II stage of its clinical trial for the treatment of Diffuse Large B-cell lymphoma.
* RNK05047: Ranok Therapeutics
RNK05047 is a first-in-class, small-molecule, tumor- and BRD4-selective protein degrader that was discovered and developed using Ranok's proprietary approach to targeted protein degradation, CHAMPTM. The bromodomain transcription factor BRD4 is a key regulator of oncogenes such as MYC and BCL2 and is involved in diverse cancer types. CHAMP-1 is a Phase I/II trial of RNK05047 currently underway in the US that will assess its safety, tolerability, and pharmacokinetics, and also includes measures of anti-tumor activity and pharmacodynamics readouts as secondary endpoints.
* BMF-219: Biomea Fusion
BMF-219 is an oral investigational covalent menin inhibitor. Data suggests that optimized covalent inhibitors can provide deeper inhibition while being better tolerated than some conventional reversible inhibitors. BMF-219 is being developed for genetically defined AML, ALL, DLBCL, MM and CLL patients. BMF-219 blocks the interaction of menin and MLL (AML, ALL), and limits the activity and/or expression of NPM1, MYC, HOX, and MEIS1, all known drivers of oncogenic proliferation and survival. Currently, the drug is in Phase I stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
* ADI-001: Adicet Bio
ADI-001 is an investigational allogeneic gamma delta CAR T cell therapy being developed as a potential treatment for relapsed or refractory B-cell NHL. ADI-001 targets malignant B-cells via an anti-CD20 CAR and via the gamma delta innate and T cell endogenous cytotoxicity receptors. Gamma delta T cells engineered with an anti-CD20 CAR have demonstrated potent anti-tumor activity in preclinical models, leading to long-term control of tumor growth. In April 2022, ADI-001 was granted Fast Track Designation by the FDA for the potential treatment of relapsed or refractory B-cell NHL. Currently, the drug is in Phase I stage of its clinical trial for the treatment of Diffuse Large B Cell Lymphoma.
The Diffuse Large B-Cell Lymphoma pipeline report provides insights into
* The report provides detailed insights about companies that are developing therapies for the treatment of Diffuse Large B-Cell Lymphoma with aggregate therapies developed by each company for the same.
* It accesses the Different therapeutic candidates segmented into early-stage, mid-stage, and late-stage of development for Diffuse Large B-Cell Lymphoma Treatment.
* Diffuse Large B-Cell Lymphoma Companies are involved in targeted therapeutics development with respective active and inactive (dormant or discontinued) projects.
* Diffuse Large B-Cell Lymphoma Drugs under development based on the stage of development, route of administration, target receptor, monotherapy or combination therapy, a different mechanism of action, and molecular type.
* Detailed analysis of collaborations (company-company collaborations and company-academia collaborations), licensing agreement and financing details for future advancement of the Diffuse Large B-Cell Lymphoma market.
Explore DelveInsight's expert-driven report today! @ Diffuse Large B-Cell Lymphoma Unmet Needs [https://www.delveinsight.com/sample-request/diffuse-large-b-cell-lymphoma-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Diffuse Large B-Cell Lymphoma Companies
Miltenyi Biomedicine, Adicet Bio, VelosBio, Novartis Pharmaceuticals, Sanofi, Eisai Co, Schrodinger, Sana Biotechnology, Ranok Therapeutics, Monte Rosa Therapeutic, Otsuka Pharmaceutical, OncoNano Medicine, Regeneron Pharmaceuticals, Hoffmann-La Roche, Celgene, Nurix Therapeutics, NovalGen, Nektar Therapeutics, Genentech, CSPC ZhongQi Pharmaceutical Technology and others.
Diffuse Large B-Cell Lymphoma pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as
* Oral
* Intravenous
* Subcutaneous
* Parenteral
* Topical
Diffuse Large B-Cell Lymphoma Products have been categorized under various Molecule types such as
* Recombinant fusion proteins
* Small molecule
* Monoclonal antibody
* Peptide
* Polymer
* Gene therapy
Download DelveInsight's latest report to gain strategic insights into upcoming Drugs Developments @ Diffuse Large B-Cell Lymphoma Market Drivers and Barriers, and Future Perspectives [https://www.delveinsight.com/sample-request/diffuse-large-b-cell-lymphoma-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Scope of the Diffuse Large B-Cell Lymphoma Pipeline Report
* Coverage- Global
* Diffuse Large B-Cell Lymphoma Companies- Miltenyi Biomedicine, Adicet Bio, VelosBio, Novartis Pharmaceuticals, Sanofi, Eisai Co, Schrodinger, Sana Biotechnology, Ranok Therapeutics, Monte Rosa Therapeutic, Otsuka Pharmaceutical, OncoNano Medicine, Regeneron Pharmaceuticals, Hoffmann-La Roche, Celgene, Nurix Therapeutics, NovalGen, Nektar Therapeutics, Genentech, CSPC ZhongQi Pharmaceutical Technology and others.
* Diffuse Large B-Cell Lymphoma Therapies - Ibrutinib, Bendamustine, Rituximab, R-miniCHOP, Selinexor, Tafasitamab, Lenalidomide, and others.
* Diffuse Large B-Cell Lymphoma Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
* Diffuse Large B-Cell Lymphoma Therapeutic Assessment by Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
Find out in DelveInsight's exclusive pipeline Report-access it now! @ Diffuse Large B-Cell Lymphoma Emerging Drugs and Major Companies [https://www.delveinsight.com/sample-request/diffuse-large-b-cell-lymphoma-pipeline-insight?utm_source=abnewswire&utm_medium=pressrelease&utm_campaign=ypr]
Table of Content
* Introduction
* Executive Summary
* Diffuse Large B-Cell Lymphoma: Overview
* Pipeline Therapeutics
* Therapeutic Assessment
* Diffuse Large B-Cell Lymphoma- DelveInsight's Analytical Perspective
* Late Stage Products (Phase III)
* Brentuximab vedotin: Pfizer
* Drug profiles in the detailed report.....
* Mid Stage Products (Phase II)
* THOR-707: Sanofi
* Drug profiles in the detailed report.....
* Early Stage Products (Phase I)
* ADI-001: Adicet Bio
* Drug profiles in the detailed report.....
* Preclinical and Discovery Stage Products
* Drug name: Company name
* Drug profiles in the detailed report.....
* Inactive Products
* Diffuse Large B-Cell Lymphoma Key Companies
* Diffuse Large B-Cell Lymphoma Key Products
* Diffuse Large B-Cell Lymphoma- Unmet Needs
* Diffuse Large B-Cell Lymphoma- Market Drivers and Barriers
* Diffuse Large B-Cell Lymphoma- Future Perspectives and Conclusion
* Diffuse Large B-Cell Lymphoma Analyst Views
* Diffuse Large B-Cell Lymphoma Key Companies
* Appendix
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Yash Bhardwaj
Email:Send Email [https://www.abnewswire.com/email_contact_us.php?pr=diffuse-large-bcell-lymphoma-clinical-trial-pipeline-accelerates-as-70-pharma-companies-rigorously-develop-drugs-for-market-entry-delveinsight]
Phone: 09650213330
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/report-store/diffuse-large-b-cell-lymphoma-pipeline-insight
Legal Disclaimer: Information contained on this page is provided by an independent third-party content provider. ABNewswire makes no warranties or responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you are affiliated with this article or have any complaints or copyright issues related to this article and would like it to be removed, please contact retract@swscontact.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Diffuse Large B-Cell Lymphoma Clinical Trial Pipeline Accelerates as 70+ Pharma Companies Rigorously Develop Drugs for Market Entry | DelveInsight here
News-ID: 4403782 • Views: …
More Releases from ABNewswire

Understanding the Etiwanda Heights Development: Scope, Background, and Community …
Rancho Cucamonga residents have heard about the Etiwanda Heights but don't fully understand the scope of the project.
Residents across Rancho Cucamonga are increasingly seeking clear, factual information about the proposed Etiwanda Heights development, a project that has evolved significantly since its original approval.
The Etiwanda Heights project, located in the northern portion of Rancho Cucamonga, was initially approved for approximately 3,000 residential units. Updated plans now reflect a proposed buildout of…

South Tampa Medical Practice Prioritizes Blood Filtration Protocols to Address P …
AgeRejuvenation utilizes EBOO therapy for chronic pain in South Tampa to address the biological triggers of persistent discomfort. This advanced protocol employs extracorporeal blood filtration and oxygenation to help patients manage systemic inflammation and toxic load. High-performing professionals can access this non-invasive, drug-free solution to restore functional vitality. The medical team focuses on purifying the internal environment to support long-term wellness.
SOUTH TAMPA, FL - April 15, 2026 - AgeRejuvenation utilizes…
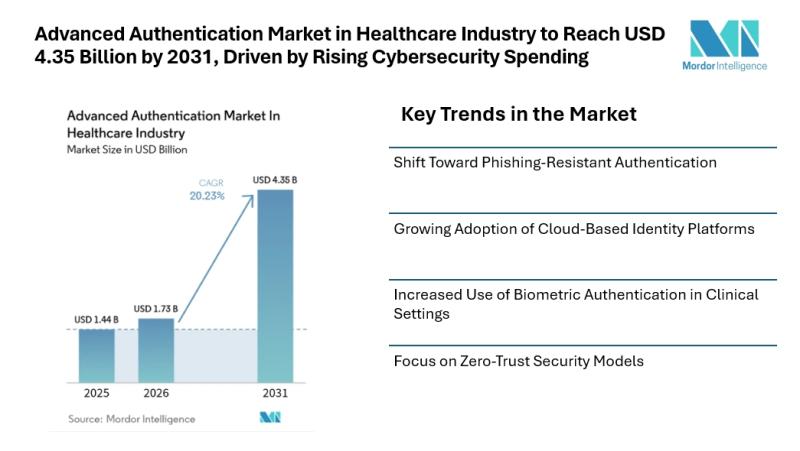
Advanced Authentication Market in Healthcare Industry to Reach USD 4.35 Billion …
Mordor Intelligence has published a new report on the advanced authentication market in healthcare industry, offering a comprehensive analysis of trends, growth drivers, and future projections
Advanced Authentication Market in Healthcare Industry Overview
According to Mordor Intelligence, the advanced authentication market in healthcare industry size [https://www.mordorintelligence.com/industry-reports/advanced-authentication-market-in-healthcare-industry?utm_source=abnewswire] was valued at USD 1.44 billion in 2025 and is estimated to grow from USD 1.73 billion in 2026 to reach USD 4.35 billion by 2031,…

Western Rooter Saves Azusa Manufacturer from Sewer Crisis with Trenchless Repair
Western Rooter resolved a critical sewer failure at an Azusa manufacturing facility where a backward-flowing line threatened operations. Using trenchless pipe bursting and repairs, the team restored proper flow in just three days, preventing shutdown and stabilizing the system.
Western Rooter [https://westernrooter.com/] has completed a major sewer repair project for a manufacturing facility in Azusa, helping prevent a full shutdown caused by a failing sewer system. The project highlights growing demand…
More Releases for Diffuse
Diffuse Large B-cell Lymphoma Therapeutics: A Leading Driver Behind Rising Preva …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
What Will the Diffuse Large B-cell Lymphoma Therapeutics Industry Market Size Be by 2025?
The market for therapeutics used in the treatment of diffuse large B-cell lymphoma has seen significant growth in recent times. The market size is projected to expand from $4.74 billion in 2024 to $5.15 billion…
Diffuse Large B-cell Lymphoma Pipeline Therapeutics Assessment Report 2024 (Upda …
DelveInsight's, "Diffuse Large B-Cell Lymphoma Pipeline Insight 2024" report provides comprehensive insights about 70+ companies and 75+ pipeline drugs in Diffuse Large B-Cell Lymphoma pipeline landscape. It covers the Diffuse Large B-Cell Lymphoma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Diffuse Large B-Cell Lymphoma therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in…
Diffuse Large B-cell Lymphoma Pipeline Drugs Report 2024 (Updated) | DelveInsigh …
DelveInsight's, "Diffuse Large B-Cell Lymphoma Pipeline Insight 2024" report provides comprehensive insights about 70+ companies and 75+ pipeline drugs in Diffuse Large B-Cell Lymphoma pipeline landscape. It covers the Diffuse Large B-Cell Lymphoma pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Diffuse Large B-Cell Lymphoma therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in…
Global Diffuse Sensors Market Analysis By Manufacturers and Competitive landscap …
Diffuse photoelectric sensor means that when the transmitter emits a beam, the target will produce diffuse reflection, and the transmitter and receiver form a standard component that will change the state of the switch when enough combined light is returned to the receiver.
Diffuse Sensors report published by QYResearch reveals that COVID-19 and Russia-Ukraine War impacted the market dually in 2022. Global Diffuse Sensors market is projected to reach US$ 777.7…
Diffuse-reflective Photoelectric Sensors Market Size, Share, Development by 2024
LP INFORMATION offers a latest published report on Diffuse-reflective Photoelectric Sensors Market Analysis and Forecast 2019-2025 delivering key insights and providing a competitive advantage to clients through a detailed report.
According to this study, over the next five years the Diffuse-reflective Photoelectric Sensors market will register a xx% CAGR in terms of revenue, the global market size will reach US$ xx million by 2024, from US$ xx million in 2019.…
Diffuse Large B-Cell Lymphoma Market Report 2017 Analysis & Research
Diffuse Large B-Cell Lymphoma Market: Overview
With a noticeable rise in the prevalence of diffuse large B-cell lymphoma (DLBCL), the market for DLBCL market is witnessing strong growth across the world. Analysts expect the trend to remain so over the next few years, owing to the increasing number of elderly people, the most prone to DLBCL segment of the overall population.
Diffuse large B-cell lymphoma (DLBCL) is the most common…
