Press release
Key Strategic Developments and Emerging Changes Shaping the Good Manufacturing Practice (GMP) Services Market for Cell and Gene Therapy
The Good Manufacturing Practice (GMP) services sector for cell and gene therapy is on track for significant expansion as innovative therapies continue to transform healthcare. With advancements in manufacturing technologies and growing demand for personalized medicine, this industry is poised for rapid growth in the coming years. Below, we explore the market's size, key players, driving forces, trends, and segment breakdowns shaping its future.Forecasted Market Value and Growth Trajectory of the GMP Services for Cell and Gene Therapy Market
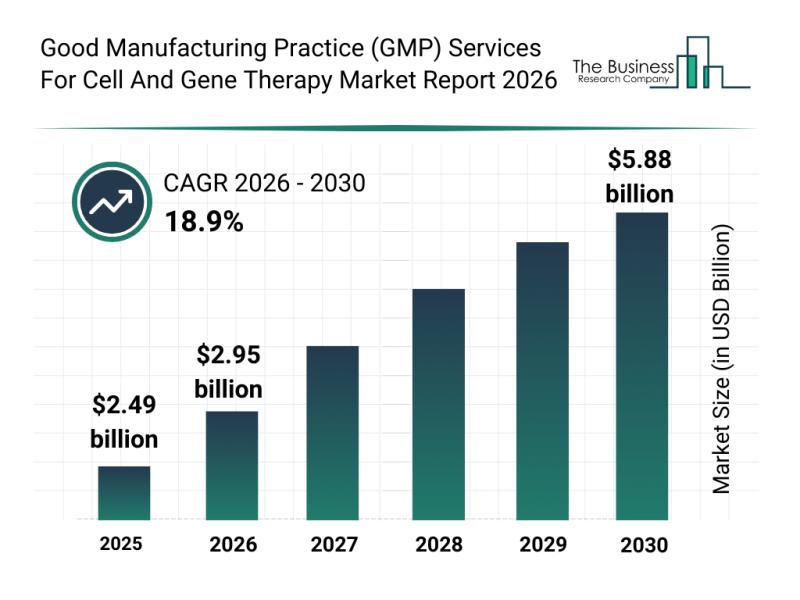
The GMP services market for cell and gene therapy is expected to experience robust growth, reaching a valuation of $5.88 billion by 2030. This expansion corresponds to a compound annual growth rate (CAGR) of 18.9%. The surge is largely fueled by the increasing commercialization of gene therapies, escalating need for scalable manufacturing capacity, expanding pipelines in personalized medicine, greater outsourcing to contract development and manufacturing organizations (CDMOs), and the widespread adoption of automated quality control technologies. Key trends shaping this growth include rising demand for commercial-scale manufacturing of these therapies, enhanced use of viral vector production services, growing emphasis on GMP-compliant automation, broader fill-finish and cold chain service offerings, and a stronger focus on regulatory compliance.
Download a free sample of the good manufacturing practice (gmp) services for cell and gene therapy market report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33124&type=smp&utm_source=OpenPR&utm_medium=Paid&utm_campaign=Feb_PR
Leading Players in the GMP Services Market for Cell and Gene Therapy
Several notable companies are driving innovation and market expansion in this sector. Major players include Novartis AG, Boehringer Ingelheim BioXcellence GmbH, Batavia Biosciences B.V., FUJIFILM Diosynth Biotechnologies, AGC Biologics Inc., Lonza Group AG, WuXi AppTec Co. Ltd., BioNTech Innovative Manufacturing Services, Charles River Laboratories International Inc., Catalent Inc., Samsung Biologics Co. Ltd., Miltenyi Biotec GmbH, SK pharmteco Co. Ltd., KBI Biopharma Inc., Oxford Biomedica plc, OmniaBio Inc., RoslinCT Ltd., Pluri CDMO, SCTbio Inc., Made Scientific Inc., and ENCell Co. Ltd.
In a notable development, Altaris LLC, a U.S.-based healthcare investment firm, acquired Minaris Regenerative Medicine GmbH in May 2025. This acquisition aims to enhance Altaris's global GMP capabilities within cell and gene therapy by leveraging Minaris's expertise in compliant manufacturing of advanced therapy medicinal products. Minaris Regenerative Medicine GmbH operates as a German CDMO specializing in GMP-compliant cell and gene therapy production.
Key Drivers Supporting Growth in the GMP Services Market
Leading companies in the GMP services sector are increasingly focused on broadening their advanced manufacturing capabilities to support regional clinical trials and accelerate the development of novel therapies. Cell therapy manufacturing is a critical area, involving GMP-compliant processes such as cell collection, modification, expansion, and formulation to ensure product quality, safety, and clinical efficacy.
For example, in July 2025, AGC Biologics, a U.S.-based CDMO, introduced cell therapy process development and clinical manufacturing services at its Yokohama Technical Center in Japan. This facility supports GMP-compliant production for preclinical and clinical stages, including CAR-T therapies, induced pluripotent stem cells, and other stem cell platforms. This expansion strengthens the company's global GMP footprint across Europe, North America, and Asia, enabling quicker and more compliant advancement of cell and gene therapies toward market approval.
View the full good manufacturing practice (gmp) services for cell and gene therapy market report:
https://www.thebusinessresearchcompany.com/report/good-manufacturing-practice-gmp-services-for-cell-and-gene-therapy-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Feb_PR
Emerging Trends Boosting the GMP Services Market Growth
The GMP services market is witnessing a rising adoption of viral vector production services, which are vital for gene therapy development and manufacturing. Companies are also focusing on integrating automation compliant with GMP standards to improve manufacturing efficiency and consistency.
Additionally, the market is seeing expansion in fill-finish and cold chain logistics services, which are crucial for maintaining product integrity during handling and distribution. There is also an enhanced emphasis on regulatory compliance, ensuring therapies meet stringent quality standards required by health authorities worldwide.
Breakdown of Key Segments in the GMP Services for Cell and Gene Therapy Market
This market is segmented across several dimensions to provide a detailed understanding of service offerings and application areas:
1) By Service Type: Process Development; Manufacturing; Analytical and Quality Control; Fill-Finish; Other Services
2) By Therapy Type: Cell Therapy; Gene Therapy; Combination Therapy
3) By Development Phase: Preclinical; Clinical; Commercial
4) By End-User: Pharmaceutical and Biotechnology Companies; Academic and Research Institutes; Other End-Users
Further subcategories include:
- Process Development: Vector Design Optimization; Cell Line Development; Upstream Process Optimization; Downstream Purification Development; Process Scale-Up Studies
- Manufacturing: Clinical Trial Material Production; Commercial Scale Manufacturing; Viral Vector Manufacturing; Plasmid Production Services; Cell Therapy Manufacturing
- Analytical and Quality Control: Method Development and Validation; Potency and Efficacy Testing; Safety and Sterility Testing; Stability and Shelf Life Studies; Release Testing Services
- Fill-Finish: Aseptic Filling Services; Vial and Syringe Filling; Lyophilization Services; Packaging and Labeling; Cold Chain Handling
- Other Services: Regulatory Support Services; Technology Transfer Services; Supply Chain Management; Storage and Distribution; Post Manufacturing Support
Global Market Outlook and Regional Focus
While North America currently holds the largest share of the GMP services market for cell and gene therapy, the Asia-Pacific region is anticipated to exhibit the fastest growth during the forecast period. The comprehensive market analysis spans key regions including Asia-Pacific, Western Europe, Eastern Europe, North America, South America, and the Middle East and Africa, providing insights into global market dynamics.
In summary, the GMP services market for cell and gene therapy is set for dynamic growth, driven by technological advancements, increasing therapy commercialization, and evolving regulatory requirements. The continued focus on scalable manufacturing, automation, and expanded service offerings will likely define the future trajectory of this promising sector.
Reach out to us:
The Business Research Company: https://www.thebusinessresearchcompany.com/,
Americas +1 310-496-7795,
Europe +44 7882 955267,
Asia & Others +44 7882 955267 & +91 8897263534,
Email us at info@tbrc.info.
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Learn More About The Business Research Company
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead.Our flagship product, the Global Market Model (GMM), is a premier market intelligence platform delivering comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Key Strategic Developments and Emerging Changes Shaping the Good Manufacturing Practice (GMP) Services Market for Cell and Gene Therapy here
News-ID: 4402435 • Views: …
More Releases from The Business Research Company
In-Depth Examination of Segments, Industry Developments, and Key Players in the …
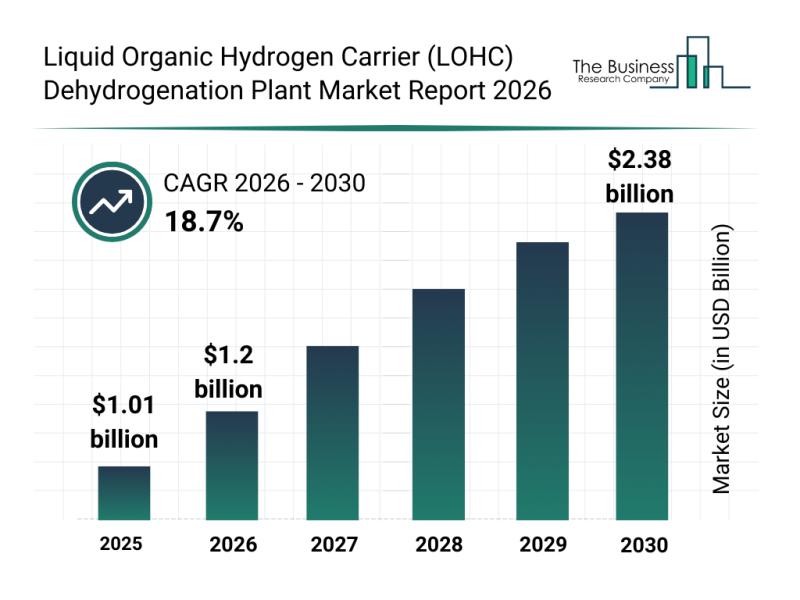
The liquid organic hydrogen carrier (LOHC) dehydrogenation plant market is on track for significant expansion as the global hydrogen economy evolves. With increasing attention on sustainable energy solutions, this sector is set to play a crucial role in enabling efficient hydrogen storage and transport. Let's delve into the market's size, key players, driving trends, major segments, and regional outlook to understand its growth trajectory.
Projected Market Size and Growth Outlook for…
Key Players and Competitive Dynamics in the Large Language Model Grounding with …
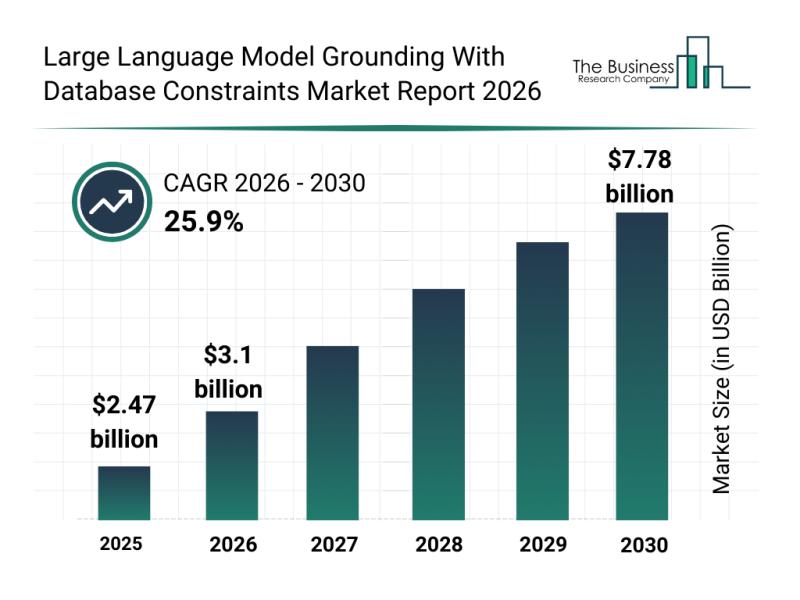
The large language model grounding with database constraints market is positioned for remarkable expansion as AI technologies become increasingly integrated with enterprise data systems. This sector's growth is driven by the rising need for reliable and compliant AI solutions that can interact seamlessly with structured databases. Let's explore the market's size, key players, emerging trends, and segmentation to understand the trajectory of this transformative technology area.
Projected Market Growth and Size…
Market Segmentation, Dynamics, and Competitive Landscape in the In-Vehicle Artif …
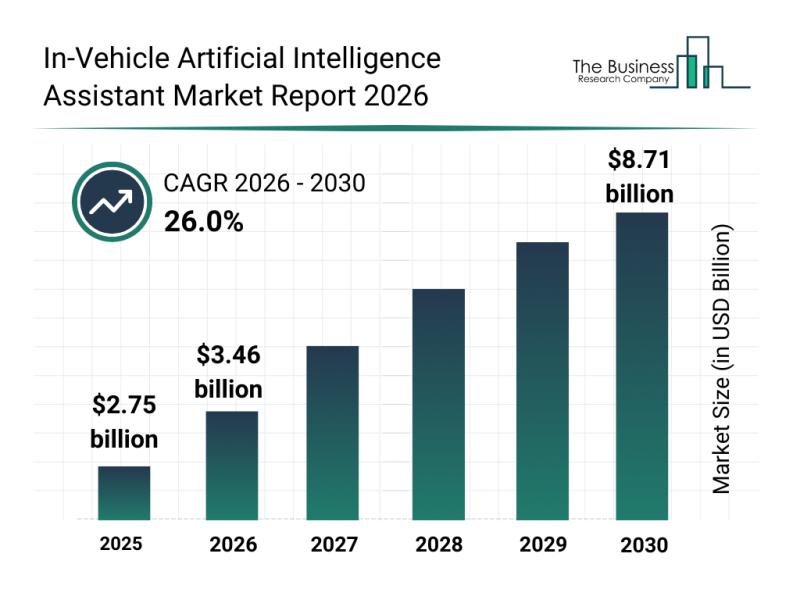
The in-vehicle artificial intelligence assistant market is on the verge of remarkable expansion as advances in automotive technology continue to evolve. With the growing integration of AI systems in vehicles, the market is set to transform how drivers and passengers interact with their cars, enhancing safety, convenience, and overall user experience. Let's explore the current market outlook, key players, emerging trends, and important segments shaping this exciting industry.
Projected Market Size…
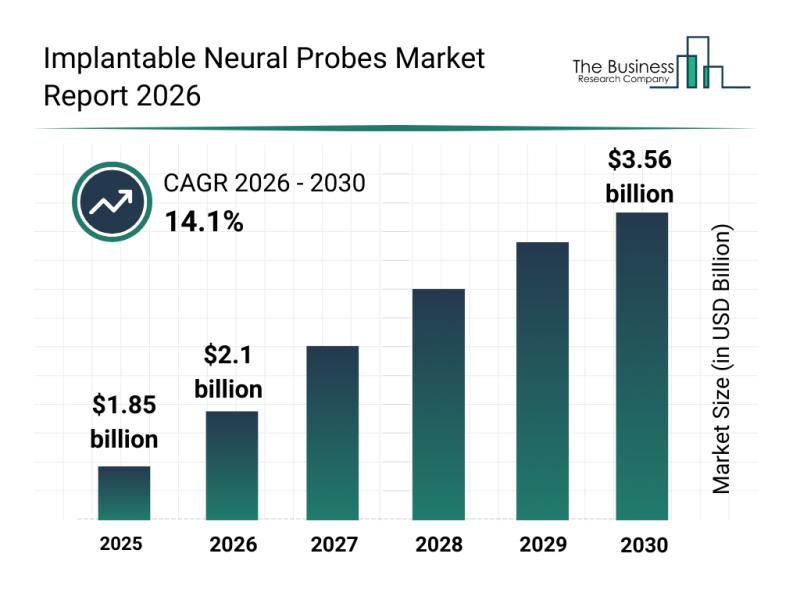
Leading Companies Advancing Innovation and Growth in the Implantable Neural Prob …
The implantable neural probes market is on a remarkable growth path, driven by advancements in neurotechnology and increasing investments in brain-machine interfacing. This sector is gaining significant attention due to its potential to revolutionize treatments for neurological disorders and enhance human-machine communication. Let's explore the market size, key players, emerging trends, and segmentation details shaping this dynamic industry.
Projected Growth and Market Size of the Implantable Neural Probes Market
The…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…