Press release
Generic Injectables Manufacturing Plant DPR & Unit Setup - 2026: Machinery Cost, CapEx/OpEx, & ROI
Setting up a Generic Injectables Manufacturing Plant requires meticulous planning, strict regulatory compliance, and advanced sterile manufacturing infrastructure. Generic injectable drugs are widely used in hospitals and healthcare facilities for treating critical conditions, chronic diseases, and emergency care. Due to their direct administration into the bloodstream or tissues, manufacturing quality, sterility, and safety standards are significantly higher than for oral formulations. As healthcare systems worldwide transition toward cost-effective treatment solutions and governments prioritize universal healthcare access, establishing a generic injectables manufacturing plant presents a strategically compelling business opportunity for entrepreneurs and pharmaceutical investors seeking to capitalize on this growing and essential market.Market Overview and Growth Potential:
The global generic injectables market demonstrates a strong growth trajectory, valued at USD 54.51 Billion in 2025. According to IMARC Group's comprehensive market analysis, the market is projected to reach USD 99.21 Billion by 2034, exhibiting a robust CAGR of 6.9% from 2026 to 2034. This sustained expansion is driven by rising demand for cost-effective injectable therapies, expanding chronic disease prevalence, patent expirations of key biologics, and supportive government policies promoting local pharmaceutical manufacturing.
Generic injectables are pharmaceutical products that contain the same active pharmaceutical ingredient (API), strength, dosage form, and route of administration as branded injectable drugs, but are marketed after patent expiration. They are available in forms such as vials, ampoules, prefilled syringes, and IV bags, and are commonly used for antibiotics, oncology drugs, pain management, anesthetics, and critical care medicines.
The global generic injectables industry is primarily driven by the rapidly expanding healthcare sector and increasing demand for affordable, high-quality injectable pharmaceutical products. Patent expirations of key biologics and technological advancements in biopharmaceutical manufacturing are further strengthening market prospects. The growth of hospital networks, increasing geriatric population, rising burden of chronic disease, and favorable regulatory frameworks supporting generic drug approvals continue to reinforce market expansion across both developed and emerging economies.
Plant Capacity and Production Scale:
The proposed generic injectables manufacturing facility is designed with an annual production capacity ranging between 50-100 Million Vials/Ampoules per Year, enabling economies of scale while maintaining operational flexibility. This capacity range allows manufacturers to cater to diverse market segments - from chronic disease management and oncology to anesthesia, antibiotics, and emergency care - ensuring steady demand and consistent revenue streams across multiple therapeutic verticals. The facility is designed to serve both domestic healthcare supply chains and regulated export market requirements, positioning the plant at the intersection of pharmaceutical manufacturing excellence and global healthcare access.
Request for a Sample Report: https://www.imarcgroup.com/generic-injectables-manufacturing-plant-project-report/requestsample
Financial Viability and Profitability Analysis:
The generic injectables manufacturing business demonstrates healthy profitability potential under normal operating conditions. The financial projections reveal:
Gross Profit Margins: 55-65%
Net Profit Margins: 25-30%
These margins are supported by stable demand across hospitals, clinics, oncology centers, and critical care units, value-added specialty injectable positioning including sterile lyophilized and biosimilar formulations, and the critical role of injectable medicines in enabling effective therapeutic outcomes worldwide. The project demonstrates strong return on investment (ROI) potential, making it an attractive proposition for both new entrants and established pharmaceutical or contract manufacturing organizations (CMOs) looking to diversify their portfolio.
Cost of Setting Up a Generic Injectables Manufacturing Plant:
Operating Cost Structure
Understanding the operating expenditure (OpEx) is crucial for effective financial planning and cost management. The cost structure for a generic injectables manufacturing plant is primarily driven by:
Raw Materials: 40-50% of OpEx
Utilities: 15-20% of OpEx
Other Expenses: Including labor, packaging, transportation, maintenance, depreciation, and taxes
Raw materials constitute the largest portion of operating costs, with Active Pharmaceutical Ingredients (APIs) being the primary input material. Excipients, solvents, buffers, stabilizers, and primary packaging components such as vials, ampoules, rubber stoppers, and caps form secondary raw material requirements. Establishing long-term contracts with reliable API and excipient suppliers helps mitigate price volatility and ensures consistent raw material supply, which is critical given that API price fluctuations and regulatory sourcing requirements represent the most significant cost variables in generic injectables manufacturing.
Capital Investment Requirements:
Setting up a generic injectables manufacturing plant requires substantial capital investment across several critical categories:
Land and Site Development:
Selection of an optimal location with strategic proximity to API and excipient suppliers, regulatory infrastructure, and skilled pharmaceutical workforce. Proximity to target healthcare institutions, hospitals, and distribution networks will help minimize supply chain costs. The site must have robust infrastructure, including reliable power supply, Water for Injection (WFI) systems, utilities, cleanroom-grade construction, and waste management systems. Compliance with local zoning laws, pharmaceutical manufacturing regulations, GMP standards, and environmental requirements must also be ensured.
Machinery and Equipment:
The largest portion of capital expenditure (CapEx) covers specialized sterile manufacturing equipment essential for production. Key machinery includes:
• Mixing and blending tanks: for preparation of API solutions and suspension formulations under controlled conditions
• Sterilization units (autoclaves): for terminal sterilization of product, equipment, and cleanroom components
• Filtration systems: for sterile filtration of injectable solutions using 0.22 micron membrane filters to remove particulates and microorganisms
• Aseptic filling machines: for automated filling of vials, ampoules, or prefilled syringes under ISO 5 (Grade A) cleanroom conditions
• Sealing and capping machines: for high-speed stoppering, crimping, and sealing of filled containers
• Cleanroom and HVAC systems: for maintenance of ISO-classified environments ensuring sterility assurance throughout production
• Lyophilization (freeze-drying) units: for production of lyophilized injectable formulations requiring moisture removal for stability
• Inspection and labeling machines: for automated visual inspection of filled containers and application of regulatory-compliant labels
• Packaging and cartoning machines: for secondary packaging of finished injectable products including cartons, blisters, and outer packaging
• Quality control and analytical instruments: for sterility testing, endotoxin testing, potency assay, particulate analysis, and container integrity verification
• Water for Injection (WFI) generation systems: for production of pharmaceutical-grade water meeting pharmacopeial specifications
Civil Works:
Building construction, factory layout optimization, and infrastructure development designed to enhance workflow efficiency, ensure GMP compliance, maintain cleanroom integrity, and minimize cross-contamination risks throughout the production process. The layout should be optimized with separate areas for raw material storage, cleanroom production zone, aseptic filling unit, sterilization section, lyophilization area, packaging and labelling line, quality control laboratory, finished goods warehouse, WFI utility block, effluent treatment area, and administrative block.
Other Capital Costs:
Pre-operative expenses, machinery installation costs, regulatory compliance certifications including GMP approvals, initial working capital requirements, and contingency provisions for unforeseen circumstances during plant establishment.
Speak to Analyst for Customized Report: https://www.imarcgroup.com/request?type=report&id=7630&flag=C
Major Applications and Market Segments:
Generic injectable products find extensive applications across diverse market segments, demonstrating their versatility and critical importance in modern healthcare delivery:
Chronic Disease Management:
Generic injectables, particularly insulin and biologics, play a key role in managing diabetes, autoimmune diseases, and other long-term conditions requiring injectable therapies. The rising prevalence of chronic non-communicable diseases globally drives consistent, non-cyclical demand for these products across primary and specialist care settings.
Oncology:
Chemotherapy agents, targeted therapies, and supportive care injectables represent a large and high-value segment. Generic oncology injectables provide life-saving treatments at significantly reduced costs compared to branded counterparts, expanding access across both developed and emerging healthcare markets and driving strong volume demand.
Anesthesia and Critical Care:
Anesthetic agents, sedatives, analgesics, and emergency care injectables are essential in hospitals, surgical centers, and intensive care units. The indispensable nature of these products in clinical settings ensures reliable, demand-driven procurement from hospital formularies and government healthcare systems worldwide.
Anti-infectives and Antibiotics:
Parenteral antibiotics, antifungals, and antivirals represent a high-volume segment driven by hospital-acquired infection management, surgical prophylaxis, and treatment of serious systemic infections requiring intravenous administration for rapid therapeutic response and high bioavailability.
Hospitals and Healthcare Facilities:
The primary distribution channel for generic injectables encompasses hospital pharmacies, government medical stores, and institutional procurement agencies that source large volumes of sterile injectable medications for inpatient and outpatient therapeutic use, representing the backbone of generic injectable market demand globally.
Why Invest in Generic Injectables Manufacturing?
Several compelling factors make generic injectables manufacturing an attractive investment opportunity:
Essential Healthcare Products with Non-Cyclical Demand:
Generic injectables are essential medicines required by patients across all economic cycles and healthcare systems. This structural indispensability ensures consistent, non-cyclical demand that spans hospitals, clinics, emergency care, and chronic disease management, providing investors with a stable and predictable revenue base.
Patent Expirations Creating Market Entry Opportunities:
The expiration of patents for numerous high-value branded injectable biologics, biosimilars, and small molecule injectables creates significant market entry opportunities for generic manufacturers. Regulatory-approved generic versions can capture substantial market share at premium-to-commodity price points, driving attractive revenue and margin profiles.
Government Support and Healthcare Access Mandates:
Governments worldwide actively promote generic drug manufacturing through abbreviated approval pathways, pricing policies, and public procurement preferences. Supportive regulatory frameworks and essential medicine list inclusions create favorable market conditions and stable institutional demand for generic injectable manufacturers.
Import Substitution and Export Market Potential:
Emerging economies with significant injectable import dependency represent strong import substitution opportunities, while regulated export markets including the US FDA, EU EMA, and WHO-prequalified markets offer premium pricing and large-volume export potential for quality-compliant generic injectable manufacturers.
Biosimilar and Complex Injectable Growth Frontier:
The growing biosimilar segment - covering monoclonal antibodies, insulin analogs, erythropoietins, and other biological injectables - represents a high-growth and high-value frontier for manufacturers capable of investing in biopharmaceutical manufacturing capabilities, offering significantly enhanced margin profiles versus small molecule generics.
Manufacturing Process Excellence:
The generic injectables manufacturing process involves several precision-controlled stages to deliver sterile, safe, and regulatory-compliant products:
• Raw Material Dispensing: APIs and excipients are accurately weighed and dispensed in controlled environments under qualified personnel supervision to prevent contamination and cross-contamination
• Solution Preparation: APIs and excipients are dissolved or suspended using Water for Injection (WFI) in stainless steel blending tanks under controlled temperature, pH, and mixing conditions
• Filtration: The prepared solution is subjected to sterile filtration through 0.22 micron membrane filters to remove particulates, bioburden, and microorganisms prior to aseptic filling
• Aseptic Filling: Sterile-filtered solution is aseptically filled into pre-sterilized vials, ampoules, or prefilled syringes under ISO 5 (Grade A) laminar airflow conditions within a classified cleanroom environment
• Stoppering and Sealing: Filled vials are stoppered with rubber closures and sealed with aluminum crimp caps using automated sealing and capping equipment to ensure container closure integrity
• Lyophilization (where applicable): Products requiring freeze-drying are loaded into lyophilization chambers for controlled freeze-drying cycles to remove moisture while preserving product stability and potency
• Visual Inspection: Filled and sealed containers undergo automated and manual visual inspection for particulates, container defects, and fill volume compliance prior to labeling
• Secondary Packaging: Products undergo secondary packaging, labelling, serialization, and batch documentation for traceability and regulatory compliance
• Quality Control: Comprehensive testing for sterility, potency, purity, endotoxin levels, and packaging integrity is conducted before product release to the market
Buy Now: https://www.imarcgroup.com/checkout?id=7630&method=2175
Industry Leadership:
The global generic injectables industry is led by established pharmaceutical manufacturers with extensive production capabilities and diverse therapeutic portfolios. Key industry players include:
• AstraZeneca
• Baxter International, Inc.
• Biocon
• Fresenius SE & Co. KGaA
• GlaxoSmithKline plc
• Hikma Pharmaceuticals plc
• Johnson & Johnson Services, Inc.
• Lupin Ltd.
• Merck KGaA
These companies serve diverse end-use sectors including hospitals and healthcare facilities, clinics and outpatient services, pharmacies, and pharmaceutical manufacturing companies, demonstrating the broad market applicability of generic injectable products across global healthcare supply chains.
Recent Industry Developments:
October 2025: Lupin Limited introduced its Liraglutide Injection, 18 mg/3 mL (6 mg/mL), in a single-patient-use prefilled pen in the United States. The formulation is therapeutically equivalent to Victoza, originally developed by Novo Nordisk Inc.. It is prescribed alongside diet and exercise to enhance glycemic control in adults and children aged 10 years and above diagnosed with type 2 diabetes mellitus.
August 2025: Teva Pharmaceuticals, Inc. secured approval from the U.S. Food and Drug Administration (FDA) for its generic version of Saxenda and subsequently launched the product in the U.S. market. This approval represents the first generic GLP-1 therapy in the country specifically indicated for weight management.
About Us:
IMARC Group is a global management consulting firm that helps the world's most ambitious changemakers create a lasting impact. The company excels in understanding its clients' business priorities and delivering tailored solutions that drive meaningful outcomes. IMARC Group provides a comprehensive suite of market entry and expansion services, including market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: (+1-201-971-6302)
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Generic Injectables Manufacturing Plant DPR & Unit Setup - 2026: Machinery Cost, CapEx/OpEx, & ROI here
News-ID: 4400680 • Views: …
More Releases from IMARC Group

Fantasy Sports Market in India 2026: Size, Share, Growth Rate & Industry Forecas …
India Fantasy Sports Market Overview
According to IMARC Group's report titled "India Fantasy Sports Market Size, Share, Trends and Forecast by Sports Type, Platform, Demographics, and Region, 2026-2034" the report offers a comprehensive analysis of the industry, including market share, growth, trends, and regional insights.
The India fantasy sports market size was valued at USD 27.0 Billion in 2025. IMARC Group estimates that the market will expand to reach USD 519.8 Billion…
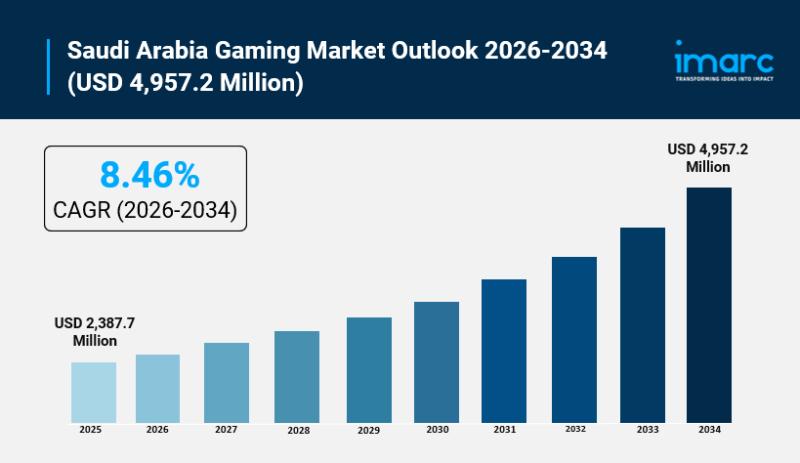
Saudi Arabia Gaming Market is Set For Strong Growth, Projected to Reach USD 4,95 …
Saudi Arabia Gaming Market Overview
Market Size in 2025: USD 2,387.7 Million
Market Forecast in 2034: USD 4,957.2 Million
Market Growth Rate 2026-2034: 8.46%
According to IMARC Group's latest research publication, "Saudi Arabia Gaming Market Report by Device Type (Consoles, Mobiles and Tablets, Computers), Platform (Online, Offline), Revenue (In-Game Purchase, Game Purchase, Advertising), Type (Adventure/Role Playing Games, Puzzles, Social Games, Strategy, Simulation, and Others), Age Group (Adult, Children), and Region 2026-2034", the Saudi Arabia…
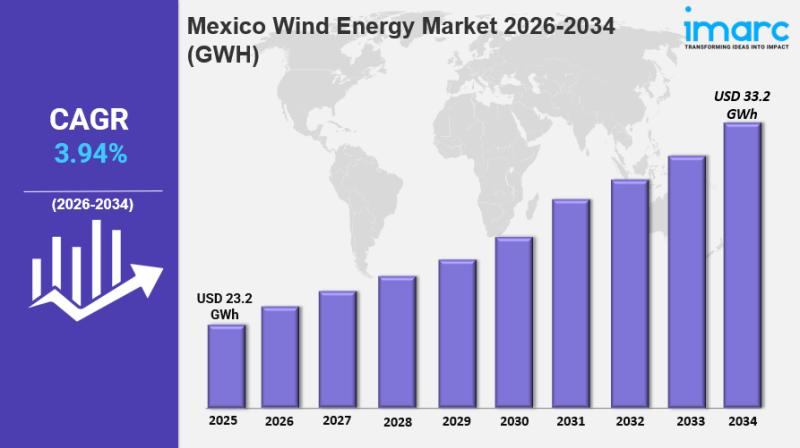
Mexico Wind Energy Market Size, Share, Industry Overview, Trends and Forecast 20 …
IMARC Group has recently released a new research study titled "Mexico Wind Energy Market Report by Component (Turbine, Support Structure, Electrical Infrastructure, and Others), Rating (≤ 2 MW, >2 ≤ 5 MW, >5 ≤ 8 MW, >8 ≤ 10 MW, >10 ≤ 12 MW, >12 MW), Installation (Offshore, Onshore), Turbine Type (Horizontal Axis, Vertical Axis), Application (Utility, Industrial, Commercial, Residential), and Region 2026-2034", offers a detailed analysis of the market…
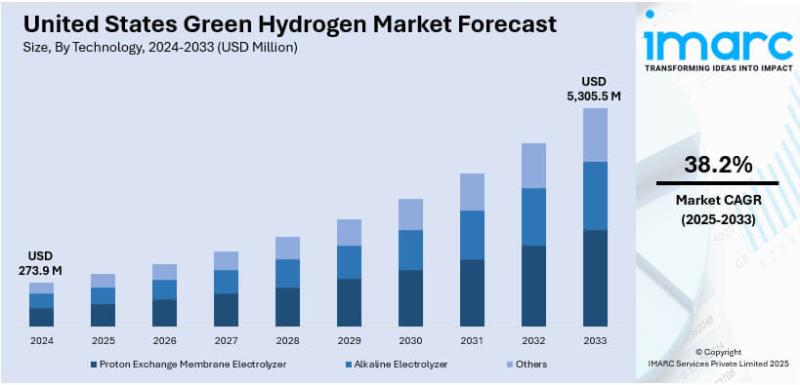
U.S. Green Hydrogen Market to Reach USD 5,305.5 Million by 2033 | CAGR 38.2%
Market Overview
The United States green hydrogen market size was valued at USD 273.9 Million in 2024 and is expected to reach USD 5,305.5 Million by 2033, exhibiting a CAGR of 38.2% during the forecast period 2025-2033. Growth is primarily driven by supportive government policies including the Bipartisan Infrastructure Law and Inflation Reduction Act, rapid technological advancements in electrolyzer efficiency, expanding applications in transportation and heavy industry, growing private-sector investments, and…
More Releases for Generic
Prominent Generic Oncology Drugs Market Trend for 2025: Novel Formulations Trans …
Which drivers are expected to have the greatest impact on the over the generic oncology drugs market's growth?
The upward trend in cancer incidence is predicted to fuel the expansion of the generic oncology drugs market. Cancer, characterized by unregulated cell proliferation affecting numerous organs, results in considerable morbidity and mortality globally. The provision of affordable generic oncology drugs is an instrumental factor in cancer care, expanding access to vital treatments,…
Generic Theater Presents Flyin' West
NORFOLK, VA (August 2023) - Generic Theater inaugurates its 43rd season with Flyin' West, written by award-winning playwright and New York Times bestselling author Pearl Cleage. Terrance Afer-Anderson directs this production, running weekends September 8th - October 1st at Generic Theater, Norfolk's underground theater located in the basement of Chrysler Hall.
Following the end of the Civil War, many former slaves took advantage of The Homestead Act and went West to…
Generic Injectables Market growth is attributed to the increasing demand for Onc …
According to Precision Business Insights (PBI), the latest report, the generic injectables market will be worth USD 22.0 billion in 2022, growing at an 11.0% CAGR from 2022 to 2028. The global generic injectables market is segmented into the following types: Product Type (Monoclonal Antibodies, Cytokines, Insulin, Peptide Hormones, Blood Factors, Immunoglobulins, Peptide Antibiotics, Vaccines, and Others), Indication (Diabetes, Cancer, Cardiovascular Diseases, Musculoskeletal, CNS, Infections, and Others), Distribution Channel (Hospital…
A Demand On Generic Drugs Market And The Need To Push The Market Of Generic Drug …
Global Generic Drugs Market
A generic drug is pharmaceutical drug, which is bio-equivalent to a branded drug in all forms such as route of administration, strength, dosage, quality, intended use and performance. Generic drugs are usually approved after patent expiration of patent drugs. Generic drugs are safe, effective and FDA approved. The global market is filled with 44% of generic drugs.
The global generic drugs market is driven by the rise of…
Generic Drug Market: Global Generic Drug Share to Reach USD 380.60 Billion by 20 …
Zion Market Research has published a new report titled “Generic Drug Market by Brand (Pure Generic and Branded Generic) for Central Nervous System (CNS), Cardiovascular, Dermatology, Oncology, Respiratory and Others Therapeutic Applications - Global Industry Perspective, Comprehensive Analysis and Forecast, 2015 – 2021”. According to the report, the global generic drug market accounted for around USD 200.20 billion in 2015 and is expected to reach approximately USD 380.60 billion by…
Generic Drugs: Global Collaboration Opportunities
Collaboration is the key to growth
With the recent economic turmoil affecting many markets around the world, the generic and branded sectors face similar problems.
At one time the generic sector was seen as a valuable way to bring effective products to a wider audience. This, allied to the rising cost of new advanced branded products, led to rapid expansion of the generics sector. Even markets traditionally based in the provision of…
