Press release
Japan Nipah Virus Market (2026-2033) | Market Enters Clinical Development, Strengthening Global Preparedness Market, Market Growth Insights
The Nipah Virus (NiV) is a highly dangerous zoonotic infection transmitted primarily by fruit bats, leading to severe respiratory illness and encephalitis in humans. With reported fatality rates ranging between 40% and 75%, Nipah continues to pose a serious public health threat. Since its discovery in 1998, recurring outbreaks have been reported mainly across South and Southeast Asia, including India, Bangladesh, and Malaysia.Request a Custom Research Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/custom-research?kb
Market Dynamics and Growth Trends
Market Presence and Regional Share
The Asia-Pacific region leads the Nipah virus therapeutics landscape, supported by recurring outbreaks, heightened surveillance, and sustained government and international health funding. Meanwhile, North America and Europe play a pivotal role through advanced research ecosystems, clinical trial infrastructure, and pandemic preparedness initiatives.
Diagnostics and Testing Landscape
⇥ The diagnostics segment is expanding steadily as health authorities emphasize early detection and outbreak containment. Among available technologies, RT-PCR-based diagnostic kits dominate the market due to their high sensitivity, specificity, and reliability in identifying viral infections.
Economic and Cost Perspective
⇥ Market expansion is being driven by increased R&D investments, emergency preparedness programs, and global funding initiatives, with the sector expected to witness multi-million-dollar growth over the coming decade.
Therapeutic Categories and Development Approaches
Key Treatment Strategies Under Development
✅ Antiviral Therapies: Broad-spectrum antivirals such as Remdesivir and Favipiravir are being evaluated for their potential efficacy against Nipah virus infections.
✅ Monoclonal Antibodies: Several neutralizing antibody candidates targeting viral surface proteins are progressing through preclinical and early clinical evaluation.
✅ Vaccine Platforms: Multiple vaccine technologies-including viral vector-based, live attenuated, and mRNA platforms-are advancing through clinical and preclinical pipelines.
✅ Innovative Drug Discovery Methods: The integration of computational modeling, AI-based drug repurposing, and nanotechnology-enabled delivery systems is accelerating candidate identification and optimization.
Emerging Drug and Vaccine Pipelines
Experimental Antiviral Therapies
• Remdesivir: Utilized on an emergency and compassionate-use basis during severe outbreaks, particularly in India.
• Favipiravir: Demonstrated promising survival outcomes in animal models of Nipah infection.
• Repurposed Antivirals: In-silico research has identified compounds such as Saquinavir and Nelfinavir for further investigation.
Vaccine Candidates in Clinical Development
1• ChAdOx1 NipahB (Oxford Vaccine Group): Currently in Phase II clinical trials in Bangladesh, representing the most advanced Nipah vaccine program worldwide.
2• University of Tokyo Vaccine Program: A Japanese-developed Nipah vaccine is scheduled to enter first-in-human trials in Europe starting April 2026.
3• HeV-sG-V Candidate: Early-stage human trials have demonstrated favorable safety and immunogenicity results.
Biologic Innovations
Monoclonal antibody m102.4 remains one of the most promising biologic candidates under evaluation for clinical use.
• Research, Development, and Clinical Trials
• Clinical Development Landscape
• Ongoing Phase II trials for ChAdOx1 NipahB in endemic regions.
• Japan-led measles-vectored Nipah vaccine trials initiating in 2026.
• Multiple early-stage candidates progressing through Phase I and animal studies across academic and biotech organizations.
R&D Process for Nipah Virus
✅ Global organizations such as CEPI continue to fund diversified vaccine platforms while maintaining emergency investigational stockpiles. Advanced computational biology, molecular modeling, and virology research are shortening development timelines. Regional centers, including India's Institute of Advanced Virology, play a crucial role in pathogen research and therapeutic validation.
✅ Research begins with virus surveillance and genomic sequencing to understand transmission, mutations, and therapeutic targets.
✅ Preclinical studies follow, using cell cultures and animal models to evaluate vaccine candidates, antivirals, and monoclonal antibodies for safety and efficacy.
✅ Promising candidates advance into Phase I-II clinical trials, often supported by global agencies like WHO and CEPI to accelerate development for this high-priority pathogen.
Japan's Strategic Role in the Nipah Virus Market
✦ Although Japan has not reported domestic Nipah outbreaks, it remains actively involved in vaccine innovation and global preparedness efforts. Leveraging strong biotech capabilities, Japanese institutions are contributing significantly to early-stage vaccine research and international clinical collaborations.
Japan's Market Contribution
✦ Development of a Nipah vaccine candidate entering human trials overseas.
✦ Ongoing public-health monitoring despite the virus being classified as low domestic risk.
✦ Participation in the broader Asia-Pacific region, which accounts for the largest share of therapeutic and diagnostic demand globally.
Japan-Developed Nipah Vaccine - Clinical Trial Overview
Vaccine Candidate
➠ Developer: Researchers at The University of Tokyo's Center for Advanced Science and Technology, Japan.
➠ Platform: The vaccine is built on a modified measles virus vector designed to stimulate immune responses that protect against Nipah virus infection.
Clinical Trial Plans
✅ Phase I Human Trial - Belgium (Starting April 2026)
✅ The first stage of human clinical testing is scheduled to begin in April 2026 in Belgium.
✅ This phase will primarily assess safety and immune response in healthy adult volunteers.
✅ It follows successful animal studies showing promising safety and efficacy in hamsters and other models.
Future Phases - Bangladesh (Second Half of 2027)
➠ If Phase I shows favorable results, Phase II trials will be conducted in Bangladesh, a region at higher risk for Nipah outbreaks.
➠ These later trials will expand evaluation to both adult and pediatric populations, focusing on efficacy and broader safety.
Funding and Support
• The Nipah vaccine project receives backing from SCARDA (Japan's Strategic Center for Advanced Research and Development), which funds advanced vaccine R&D.
• It involves international collaboration, leveraging European clinical trial infrastructure for early human testing.
Current Clinical Practice and Dosage Use
In the absence of approved therapies, supportive care remains the standard treatment approach. Antivirals like Remdesivir and Favipiravir are administered on an off-label or compassionate-use basis, with dosing protocols largely adapted from previous antiviral applications and preclinical data.
Vaccine Supply Chain & Manufacturing Updates
1. Strategic Vaccine Reserve & Manufacturing Scale-Up
CEPI (Coalition for Epidemic Preparedness Innovations), the University of Oxford, and the Serum Institute of India (SII) are collaborating to establish an investigational vaccine reserve for ChAdOx1 NipahB.
✔ SII is manufacturing up to 100,000 doses of the candidate vaccine that will support:
✔ Ongoing Phase II clinical trials
✔ Potential emergency deployment during future outbreaks
✔ This reserve will be held at SII's facilities (Pune, India) and serves as a pre-deployed emergency supply if outbreaks occur in high-risk regions.
2. Manufacturing Partnerships and Expertise
SII is leveraging its proven capacity (from producing billions of doses of other vaccines) to support:
✅ Process development
✅ Clinical supply production
✅ Long-term stockpiling for outbreak preparedness
✅ Funding for this production effort includes up to $7.3 million from CEPI, indicating targeted investment in manufacturing readiness.
3. Expanded R&D & Candidate Development Support
Additional funding flows (e.g., CEPI's $13 million support to Gennova Biopharmaceuticals for its saRNA Nipah vaccine candidate) are helping expand the portfolio of vaccine technologies in the pipeline. This broadens potential supply options beyond a single vaccine platform.
4. Integration of International Trial Material Supply
Clinical trial material (vaccine doses) is being supplied internationally to support:
Trials in Bangladesh
• Planned trials linked to other regions (Europe, Japan-linked trials)
• These international supplies ensure clinical programs can progress while local regulatory reviews continue.
Key Industry Highlights
✔ Nipah virus continues to be recognized as a high-risk emerging infectious disease with no approved treatments to date.
✔ Vaccine candidates from Oxford and Japanese research groups represent the most advanced solutions in development.
✔ Asia-Pacific remains the primary market and innovation hub, driven by outbreak risk and public-health investment.
✔ Global regulatory support and international funding initiatives are accelerating the path toward effective countermeasures.
Related Reports
Vaccines Market: https://www.datamintelligence.com/download-sample/vaccines-market?kb
Influenza Vaccine Market: https://www.datamintelligence.com/download-sample/influenza-vaccines-market?kb
Vaccine Delivery Devices Market: https://www.datamintelligence.com/download-sample/vaccine-delivery-devices-market?kb
Asia-Pacific Vaccine Market: https://www.datamintelligence.com/download-sample/asia-pacific-vaccine-market?kb
Hepatitis B Vaccine Market: https://www.datamintelligence.com/download-sample/hepatitis-b-vaccine-market?kb
Preventive Vaccine Market: https://www.datamintelligence.com/download-sample/preventive-vaccine-market?kb
Peptide Cancer Vaccine Market: https://www.datamintelligence.com/download-sample/peptide-cancer-vaccine-market?kb
Polio Vaccine Market: https://www.datamintelligence.com/download-sample/polio-vaccine-market?kb
BCG Vaccine Market: https://www.datamintelligence.com/download-sample/bcg-vaccine-market?kb
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Japan Nipah Virus Market (2026-2033) | Market Enters Clinical Development, Strengthening Global Preparedness Market, Market Growth Insights here
News-ID: 4384628 • Views: …
More Releases from DataM Intelligence 4 Market Research LLP

Oncology Biosimilars Market (2025-2033) | Monoclonal Antibodies, Oncology Drugs, …
DataM Intelligence has released a new research report titled "Oncology Biosimilars Market Size 2025" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…
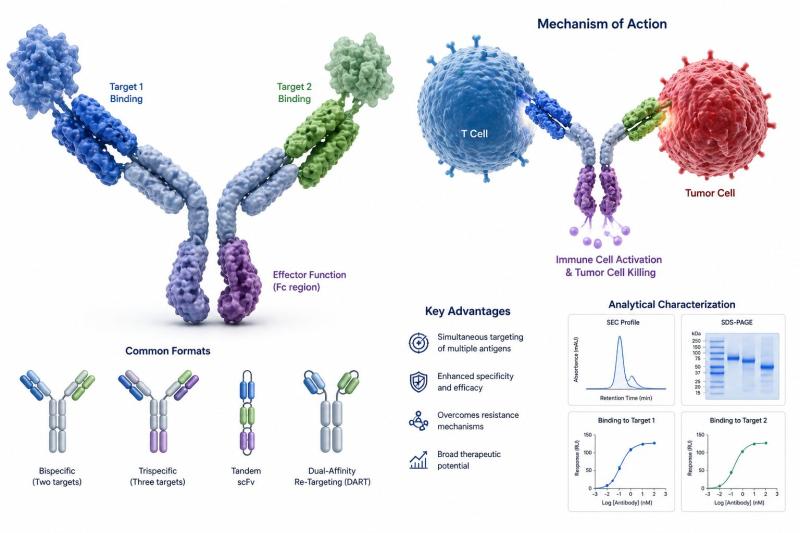
Multispecific Antibodies Market to Reach USD 98.45 Billion by 2033 at 25.9% CAGR …
The global multispecific antibodies market reached USD 15.87 billion in 2025 and is expected to reach USD 98.45 billion by 2033, growing at a CAGR of 25.9% during the forecast period from 2026 to 2033. The market is experiencing rapid expansion driven by the growing adoption of precision medicine, increasing integration of AI-enabled R&D, and rising demand for advanced therapies, alongside accelerating diagnostics demand and ongoing reimbursement pressure across healthcare…

Automotive Silicone Market (2026) | Automotive Elastomers, Automotive Fluids, Sp …
DataM Intelligence has released a new research report titled "Automotive Silicone Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Tungsten-Based Materials Market (2026-2033) | Tungsten Metal, Tungsten Alloys, S …
DataM Intelligence has released a new research report titled "Tungsten-Based Materials Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…
More Releases for Nipah
Nipah virus testing Market - Global Share, Size & Changing Dynamics 2020-2033
The latest study released on the Global Nipah virus testing Market by HTF MI Research evaluates market size, trend, and forecast to 2033. The Nipah virus testing study covers significant research data and proofs to be a handy resource document for managers, analysts, industry experts and other key people to have ready-to-access and self-analyzed study to help understand market trends, growth drivers, opportunities and upcoming challenges and about the competitors.
Consider…
Nipah Virus Infection Testing Market: A Guide to Understanding the Process
The Nipah Virus (NiV) Infection Testing Market was about USD 290 million in 2024 and is projected to reach ~USD 510 million by 2033, implying a ~6.4% CAGR (2025-2033).
Nipah Virus Infection Testing Market Overview
The Nipah Virus Infection Testing Market is gaining increasing attention due to the virus's high fatality rate and the recurring outbreaks reported in parts of Asia. Rising global health concerns, coupled with the need for early…
Nipah Virus Infection Testing Market Share, Trends, Future Outlook, Analysis and …
Global Nipah virus infection testing market is anticipated to grow at a CAGR of 4.8% during the forecast period (2023-2030). Nipah virus testing is primarily used for diagnostic purposes to detect genetic material or antibodies in patient samples. Different methods such as PCR, ELISA, and rapid diagnostic tests can be used for this. Regular testing is conducted to monitor the presence of the Nipah virus in specific areas and among…
Nipah Virus Infection Testing Market 2023 Driving Factors Forecast Research 2029
Nipah virus infection testing market is anticipated to grow at a CAGR of 4.8% during the forecast period (2023-2030). The market's growth is attributed to the growing incidence of Nipah virus infection across the globe. NiV can be passed from infected animals (such as pigs or bats) or their bodily fluids to individuals through contact. It involves fluids including blood, urine, and saliva. According to the National Institute of Health,…
Nipah Virus Infection Testing Market Outlook 2029: Presents Market Insights & De …
Global Nipah virus infection testing market is anticipated to grow at a CAGR of 4.8% during the forecast period (2023-2030). Nipah virus testing is primarily used for diagnostic purposes to detect genetic material or antibodies in patient samples. Different methods such as PCR, ELISA, and rapid diagnostic tests can be used for this. Regular testing is conducted to monitor the presence of the Nipah virus in specific areas and among…
Creative Diagnostics Introduces New Nipah Virus Antigens and Antibodies
As a global leading supplier of raw materials, antibodies, and reagents for bio-technology industry, Creative Diagnostics recently launches a series of NiV recombinant antigen and antibody pairs. All of these new products are produced using standardized production processes to ensure the highest quality. Tests developed using these reagents can assist researchers in routine diagnostics and NiV vaccine development.
Nipah virus (NiV) was initially isolated and identified in 1999 during an outbreak…
