Press release
Biological Safety Testing Market to Reach US$ 14.21 Billion by 2032 at 11.8% CAGR; North America Leads with 42% Share | Key Players Thermo Fisher Scientific, Charles River, Eurofins Scientific
The Biological Safety Testing market reached US$ 5.82 billion in 2024 and is expected to reach US$ 14.21 billion by 2032, growing at a CAGR of 11.8% during the forecast period 2025-2032. Market growth is driven by the increasing production of biologics, vaccines, cell and gene therapies, and biosimilars, along with stringent regulatory requirements to ensure product safety, purity, and quality before commercialization.Biological safety testing plays a critical role in detecting contaminants such as viruses, mycoplasma, endotoxins, and residual DNA in biologic products. The market is high in North America, supported by a strong biopharmaceutical manufacturing base, advanced regulatory frameworks, and high R&D spending. Europe follows with steady demand due to strict quality standards, while Asia-Pacific is emerging as a high-growth region driven by expanding biopharma manufacturing, increasing outsourcing of testing services, and rising investments in life sciences infrastructure.
Get a Free Sample PDF Of This Report (Get Higher Priority for Corporate Email ID):- https://www.datamintelligence.com/download-sample/biological-safety-testing-market?sai-v
The biological safety testing market refers to the global industry focused on testing services and technologies used to ensure the safety, purity, and quality of biologics, pharmaceuticals, and medical products.
Key Developments
✅ January 2026: In North America, adoption of automated biological safety testing platforms increased across pharmaceutical, biotech, and medical device sectors to accelerate safety profiling and regulatory compliance, with leading technologies from Thermo Fisher Scientific, PerkinElmer, and Agilent Technologies enabling high-throughput cytotoxicity and genotoxicity assays.
✅ January 2026: In Europe, harmonization of regulatory requirements by authorities (including updated ISO and EMA guidelines) drove demand for integrated bioassay and molecular diagnostic safety testing workflows, supported by solutions from bioMérieux, Roche Diagnostics, and QIAGEN that enhance pathogen detection and endotoxin assessment.
✅ December 2025: In Asia-Pacific, expansion of contract testing and CRO networks accelerated uptake of cell-based and immunotoxicity testing services, with service expansion by SGS, Intertek, and Eurofins Scientific improving access to GLP-compliant biological safety testing for local and regional developers.
✅ December 2025: Globally, advancements in next-generation sequencing (NGS)-enabled safety assays and high-content screening enhanced detection of adventitious agents and biological contaminants, with innovation from Illumina, PerkinElmer, and Luminex Corporation advancing assay sensitivity and throughput.
✅ November 2025: In Latin America, increased regulatory emphasis on biologics, vaccines, and advanced therapeutic product safety supported expanded use of microbial and sterility testing services, delivered by providers such as Eurofins Scientific, Charles River Laboratories, and Toxikon Corporation.
✅ October 2025: Worldwide, integration of AI and machine learning analytics into biological safety testing platforms improved predictive toxicology and data interpretation, with contributions from ExxonMobil Data Analytics (life sciences division), Certara, and Dotmatics enhancing data quality and risk assessment.
Mergers & Acquisitions
✅ January 2026: In the United States, Thermo Fisher Scientific acquired a biological safety testing automation technology company to expand its integrated safety testing solutions and strengthen laboratory workflow offerings.
✅ December 2025: In Europe, bioMérieux completed the acquisition of a molecular safety assay developer to broaden its pathogen detection and biological safety testing portfolio.
✅ November 2025: In Asia-Pacific, Eurofins Scientific acquired a regional contract biological testing and safety services provider to enhance its GLP testing network and service capabilities.
Key Players
Avance Biosciences | Cytovance Biologics | Eurofins Scientific | Lonza | Merck KGaA | Promega Corporation | Thermo Fisher Scientific | Toxikon | WuXi AppTec | Charles River Laboratories | Others
Key Highlights
Thermo Fisher Scientific holds 22% share, driven by its broad portfolio of laboratory services, strong global infrastructure, and deep integration across biopharma research, development, and manufacturing workflows.
Charles River Laboratories holds 18% share, supported by its leadership in preclinical research services, toxicology testing, and long-term partnerships with pharmaceutical and biotechnology companies.
Eurofins Scientific holds 15% share, leveraging its extensive global laboratory network, strong expertise in bioanalytical and safety testing, and diversified life sciences service offerings.
Lonza holds 14% share, driven by its strong position in biologics development and manufacturing, advanced cell and gene therapy capabilities, and end-to-end CDMO services.
WuXi AppTec holds 12% share, supported by its integrated discovery, development, and manufacturing platforms and strong client base across global biopharma companies.
Merck KGaA holds 8% share, leveraging its life science tools, reagents, and process solutions widely used in biopharmaceutical research and quality testing.
Promega Corporation holds 5% share, driven by its specialized biochemical assays, reagents, and tools supporting drug discovery and toxicology research.
Toxikon holds 3% share, focusing on GLP-compliant toxicology testing and safety assessment services for medical devices and pharmaceuticals.
Avance Biosciences holds 2% share, contributing through niche bioanalytical and in vivo testing services for early-stage drug development.
Cytovance Biologics holds 1% share, focusing on biologics development, microbial fermentation, and protein expression services.
Others hold 0-5% share, comprising regional CROs and emerging life science service providers expanding specialized testing and biopharmaceutical support capabilities.
Buy Now & Unlock 360° Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=biological-safety-testing-market?sai-v
(Single User Report: USD 4350 & One Year Database Subscription: USD 12K)
Market Drivers
- Increasing regulatory requirements and stringent safety standards for biologics, vaccines, and therapeutic proteins driving demand for biological safety testing.
- Rising global biopharmaceutical R&D and manufacturing activities necessitating comprehensive safety evaluation throughout development and commercialization.
- Growing emphasis on patient safety and risk mitigation leading to mandatory testing for contaminants, adventitious agents, and impurities.
- Advancements in molecular and analytical technologies enhancing sensitivity, - speed, and accuracy of biological safety assays.
- Expansion of contract testing and CDMO services enabling scalable safety testing support for biotech and pharmaceutical developers.
Industry Developments
- Adoption of next-generation sequencing (NGS), PCR, and high-throughput screening platforms for rapid detection of pathogens and contaminants.
- Integration of automated and digital testing workflows to improve laboratory efficiency, reproducibility, and data integrity.
- Strategic partnerships between biopharma companies, CROs, and safety testing service providers to expand global testing capacity.
- Growing implementation of in-silico safety assessment tools and AI-driven analytics to complement traditional biological assays.
- Development of specialized assays and kits for emerging biologics, cell and gene therapies, and viral vector products.
Regional Insights
North America - 42% share: "Driven by strong biopharmaceutical R&D ecosystem, stringent regulatory oversight, high adoption of advanced testing technologies, and extensive safety compliance requirements."
Europe - 28% share: "Supported by robust pharma and biotech sectors, harmonized regulatory frameworks, and widespread use of cutting-edge safety testing platforms."
Asia Pacific - 22% share: "Fueled by expanding biologics manufacturing, increasing clinical trial activity, rising healthcare investments, and growing regulatory alignment."
Latin America - 5% share: "Driven by improving biopharma infrastructure, increasing contract testing demand, and expanding biotech activities."
Middle East & Africa - 3% share: "Supported by emerging biotechnology initiatives, growing healthcare investments, and increasing focus on biologics safety compliance."
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/customize/biological-safety-testing-market?sai-v
Key Segments
By Product
Reagents and kits dominate the market due to their recurring use in routine testing and quality control processes. Instruments account for a significant share, supported by increasing automation and demand for high-throughput and reliable testing systems. Other products include consumables and accessories that support laboratory operations and compliance requirements.
By Application
Vaccines and therapeutics represent a major application segment, driven by stringent regulatory requirements for safety and quality assurance. Cellular and gene therapy is a fast-growing segment, supported by rising development of advanced therapies and the need for rigorous biological safety testing. Blood and blood-based therapy hold a substantial share due to continuous demand for sterility and endotoxin testing. Other applications include medical devices, tissue engineering, and biopharmaceutical research.
By Test
Sterility tests account for the largest share, driven by mandatory regulatory standards across biopharmaceutical manufacturing. Endotoxin tests are widely adopted to ensure product safety and prevent pyrogenic reactions. Bioburden tests play a critical role in monitoring microbial load during manufacturing processes. Other tests include mycoplasma testing and environmental monitoring, supporting comprehensive biological safety assurance.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription
Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTWg
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Biological Safety Testing Market to Reach US$ 14.21 Billion by 2032 at 11.8% CAGR; North America Leads with 42% Share | Key Players Thermo Fisher Scientific, Charles River, Eurofins Scientific here
News-ID: 4368197 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Oncology Biosimilars Market (2025-2033) | Monoclonal Antibodies, Oncology Drugs, …
DataM Intelligence has released a new research report titled "Oncology Biosimilars Market Size 2025" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…
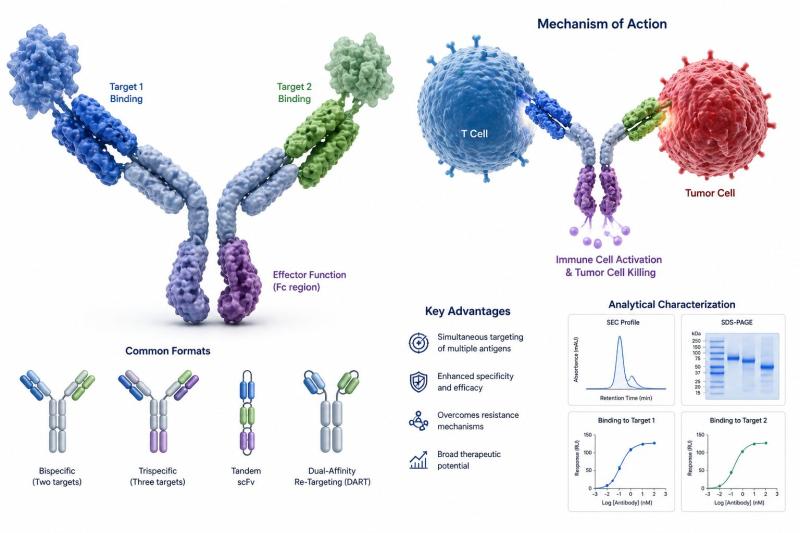
Multispecific Antibodies Market to Reach USD 98.45 Billion by 2033 at 25.9% CAGR …
The global multispecific antibodies market reached USD 15.87 billion in 2025 and is expected to reach USD 98.45 billion by 2033, growing at a CAGR of 25.9% during the forecast period from 2026 to 2033. The market is experiencing rapid expansion driven by the growing adoption of precision medicine, increasing integration of AI-enabled R&D, and rising demand for advanced therapies, alongside accelerating diagnostics demand and ongoing reimbursement pressure across healthcare…

Automotive Silicone Market (2026) | Automotive Elastomers, Automotive Fluids, Sp …
DataM Intelligence has released a new research report titled "Automotive Silicone Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Tungsten-Based Materials Market (2026-2033) | Tungsten Metal, Tungsten Alloys, S …
DataM Intelligence has released a new research report titled "Tungsten-Based Materials Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…
More Releases for Scientific
Introducing The Future of Multipurpose Digital Shaker Market: Thermoline Scienti …
The Multipurpose Digital Shaker report compiles the market information depending upon market development and growth factors, optimizing the growth path. In addition, it highlights the strategies and market share of the leading vendors in the particular market. The report follows a robust research methodology model that helps to make informed decisions. It obtains both qualitative and quantitative market information supported by primary research.
Download FREE Sample Report @ https://www.reportsnreports.com/contacts/requestsample.aspx?name=7760790
The Multipurpose…
Four Blocks Dry Bath Incubator Market | Benchmark Scientific, Biolab Scientific, …
The global four blocks dry bath incubator market report is a comprehensive report that provides a detailed analysis of the current status and future trends of the four blocks dry bath incubator market worldwide. This report provides valuable information to industry stakeholders by offering an in-depth perspective on market dynamics, competitive landscape, growth opportunities, and key challenges faced by industry participants.
From the perspective of market dynamics, this report explores the…
Incubator Shakers market: Leading Market Trends to Look for in 2022 | Biolab Sci …
"
The Incubator Shakers global market is thoroughly researched in this report, noting important aspects like market competition, global and regional growth, market segmentation and market structure. The report author analysts have estimated the size of the global market in terms of value and volume using the latest research tools and techniques. The report also includes estimates for market share, revenue, production, consumption, gross profit margin, CAGR, and other key factors.…
U.S. Biomedical Refrigerators and Freezers Market 2020 Trends with Top Players - …
U.S. biomedical refrigerators and freezers market is poised to gain substantial traction in the near future. This can be mainly attributed to strong demand for the product across hospitals, diagnostic centers, pharmacies, research labs and blood banks. Biomedical refrigerators and freezers offer optimal conditions for efficient storage of biological samples.
These refrigerators and freezers are prominently used in healthcare facilities for the storage of biological samples and chemicals such as blood,…
5-Amino-3-Methylisoxazole Market Research Report 2025 | Global Key Player -Pure …
This research report titled Global (United States, European Union and China) 5-Amino-3-Methylisoxazole Market Research Report 2019-2025 has been added to the wide online database managed by Market Research Hub (MRH). The study discusses the prime market growth factors along with future projections expected to impact the 5-Amino-3-Methylisoxazole Market during the period between 2019 and 2025. The concerned sector is analyzed based on different market factors including drivers, restraints and opportunities…
2019 Blood Bank Market By Key Player's Follett , Helmer Scientific , Norlake sci …
The Blood Bank Market report offers a detailed Outlook and future prospects of the Industry. The Blood Bank market report includes various topics like market size & share, Product types, applications, key market drivers & restraints, challenges, growth opportunities, key players, competitive landscape, Regions etc.
Request for the sample copy@ https://www.supplydemandmarketresearch.com/home/contact/44471?ref=Sample-and-Brochure&toccode=SDMRPH44471&utm_source=Ss
The key manufacturers in the Blood Bank include
• Panasonic Biomedical
• Dometic
• Thermo Fisher Scientific
• Vestfrost Solutions
• Follett
• Helmer Scientific
• Norlake scientific…
