Press release
GMP Cell Banking Service Market Estimated to Grow Strongly by 2024
GMP Cell Banking is an important component in the production of well-characterized biopharmaceutical products. A qualifies cell bank provides the opportunity to detect and identify possible contaminating agents such as viruses and mycoplasma before their use in a large scale. A cell bank ensures uniform population of cells preserved for better health to ensure better products. Cells preserved in banks are then used as a start material for each manufacturing. The importance of GMP cell banking ensure the integrity of cells and is maintained with sufficient supply of material to the cell for the better life of byproduct. Cells stored in GMP Cell banking is used for meeting manufacturing needs for the production of cell in recombinant biopharmaceuticals and vaccine production. In any GMP cell banking service, a proper cell-bank testing plan is necessary to certify purity and safety of a resulting biopharmaceutical product. All type of cell bank service requires expertise, constant monitoring, optimal equipment, appropriate quality control, proper environment, close communication and continuous troubleshoot. Cell banking service requires characterizes the cell line and require an array of tests to be performed in different scenarios. The various type of testing is performed to find the adventitious agents testing to identify and genetic stability testing. It is required to know what the best time to perform these tests is and how these tests impact the project outcome. The sudden increase in demand for ready-to-use cells is increasing the demand for GMP cell banking service. Services provided by GMP cell banking service provided includes: maintaining the master cell line for years, maintaining working cell line with the low passage, identity confirmation by fingerprinting, purity testing for mycoplasma, viruses and fungi detection, and stability testing for expression constructs and viral vectors.A Sample of this Report is Available Upon Request @ http://www.persistencemarketresearch.com/samples/13735
The cell banking service process includes both GMP production and Non-GMP production master cell bank. The type of GMP cell banking includes the type of services such as working cell bank (ECB), end-of-production (EOP) and research and development (R&D) cell banks. GMP cell banking service market is an emerging field. There are various tests and procedures to be followed by the service providers to a high-quality service. Many biopharmaceutical and pharmaceutical companies are very active in the field of GMP cell banking service. The ongoing worldwide research is expected to fuel the GMP cell banking service market. Within the diagnostic segment of the GMP cell banking service, most of the focus has been on the detection of contaminations and maintaining the health of cells. Most of the activity and interest of the companies is focused on the therapeutic research of the GMP cell banking service field.
The global market for GMP cell banking service market is expected to be driven by the advancement in technology of molecular and genetic studies. The key drivers of the market are the increasing cases prevalence of cancer, infectious disease and other diseases creating focus towards the requirement of more research for drug discovery. Moreover, the increasing research activities for drug development, growing research on cancer and increasing collaboration between research organizations, pharmaceutical, and biopharmaceuticals companies is also acting as a fuel to the GMP cell banking service market and is expected to drive the market within the forecast period of 2016-2026. However, the high cost of the procedures and the lack of fund can be the restraint for the growth of this market.
The number of companies developing methods to detect and modifying DNA for diagnosis and therapeutic purposes has significantly changed in recent years. The increasing investment by the pharmaceutical and biotechnology companies towards research and development is expected to drive the market. Based on the product working cell bank (ECB), master cell bank (MCB), end-of-production (EOP), virus bank and research and development (R&D) cell banks.
By end user, the global GMP Cell Banking Service market has been segmented into academic research institutes, biotechnology companies, biopharmaceutical companies, pharmaceutical companies, contract research organizations and contract manufacturing organizations. GMP cell banking service procedures are useful in many research oriented areas such as developmental biology, oncology, drug discovery and others.
By regional presence, GMP cell banking service market is segmented into five key regions viz. North America, Latin America, Europe, Asia-Pacific, and the Middle East & Africa. North America will continue to dominate the GMP cell banking service market for due to high availability of funding for research and better economic conditions. Europe is expected to hold second largest market share in global GMP cell banking service market. The growing government initiatives to support research activities and increasing number of biotechnology and biopharmaceutical companies in APAC are also driving the market for GMP cell banking service in APAC.
To View TOC of this Report is Available Upon Request @ http://www.persistencemarketresearch.com/toc/13735
Some of the major players in global GMP cell banking service market include Virusure, Syngene, Sigma-Aldrich Co. LLC., WuXi AppTec, Zelle Biotechnology Private Limited, Microbiology Quality & Associates, Charles River Laboratories International, Inc., IHC World, LLC., Paragon Bioservices, Inc., Austrianova, VertMarkets, Inc. and others.
About Us
Persistence Market Research (PMR) is a U.S.-based full-service market intelligence firm specializing in syndicated re-search, custom research, and consulting services. PMR boasts market research expertise across the Healthcare, Chemicals and Materials, Technology and Media, Energy and Mining, Food and Beverages, Semiconductor and Electronics, Con-sumer Goods, and Shipping and Transportation industries. The company draws from its multi-disciplinary capabilities and high-pedigree team of analysts to share data that precisely corresponds to clients’ business needs.
PMR stands committed to bringing more accuracy and speed to clients’ business decisions. From ready-to-purchase market research reports to customized research solutions, PMR’s en-gagement models are highly flexible without compromising on its deep-seated research values.
Contact
Persistence Market Research Pvt. Ltd
305 Broadway
7th Floor, New York City,
NY 10007, United States,
USA – Canada Toll Free: 800-961-0353
Email: sales@persistencemarketresearch.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release GMP Cell Banking Service Market Estimated to Grow Strongly by 2024 here
News-ID: 436712 • Views: …
More Releases from Persistence Market Research Pvt. Ltd

Innovative Rehabilitation Technologies Drive Growth in Stroke Rehabilitation Mar …
Stroke Rehabilitation Market Size
The global stroke rehabilitation market size was valued at US$ 266.4 million in 2021 and is expected to grow at a CAGR of 9% from 2022 to 2032. Revenue generated from the sales of stroke rehabilitation solutions will surge to US$ 679.6 Million by the end of 2032.
The growth of the market is being driven by the increasing incidence of strokes, the growing demand for innovative rehabilitation…
Launch of New Laser-Based Devices by Key Players Fueling Growth of Global Aesthe …
Market Study on Aesthetic Lasers
The aesthetic lasers market reached US$2.8 billion in global revenue in 2021, and it will continue to expand quickly, with a value CAGR of 6.4%, to reach US$5.5 billion by the end of 2032. The growth of the market is being driven by the increasing demand for minimally invasive cosmetic procedures, rising disposable income, technological advancements, and growing medical tourism.
Stay Ahead with Market Intelligence Get Free…

Predictive Insights for Precision Medicine: The Global Biosimulation Market is S …
Biosimulation Market Research
The Global Biosimulation Market Size was valued at USD 2.9 billion in 2022 and is expected to reach USD 10.0 billion by 2030, registering a CAGR of 16.9% during the forecast period. The growth of the biosimulation market is being driven by the increasing use of biosimulation in a variety of applications, such as drug discovery, clinical trials, and toxicity prediction.
Market Overview
The biosimulation market has experienced rapid growth…
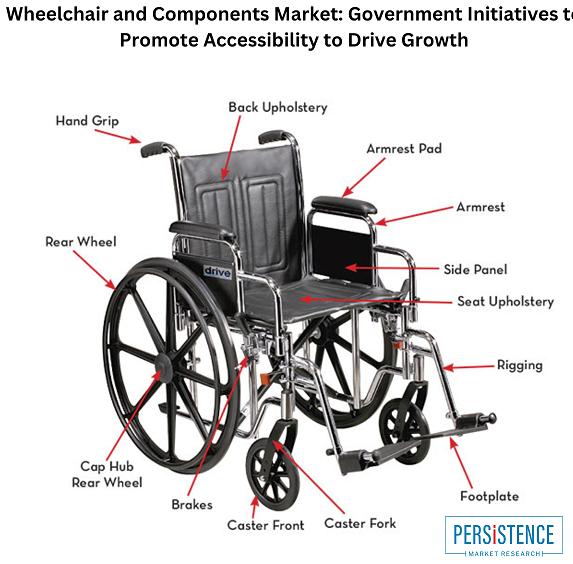
Wheelchair and Components Market: Growing Demand for Lightweight and Smart Wheel …
Wheelchair and Components Market
The global wheelchair and components market is projected to reach USD 17.9 billion by 2033, at a CAGR of 3.7% from 2022-2033. The growth of the market is driven by the increasing prevalence of disabilities, the rising geriatric population, and technological advancements in wheelchair design and manufacturing.
Click Here to Get Free Sample Copy of this Report@ https://www.persistencemarketresearch.com/samples/12961
The global wheelchair and components market has witnessed significant growth in…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…
