Press release
Relapsed/Refractory Myelodysplastic Syndrome Pipeline 2025: Pioneering Clinical Developments by More Than Five Global Companies - DelveInsight | Highlighting Cantex Pharmaceuticals, Novartis, Newave P
DelveInsight's, "Relapsed or Refractory Myelodysplastic Syndrome - Pipeline Insight, 2025," report provides comprehensive insights about 5+ companies and 5+ pipeline drugs in Relapsed or Refractory Myelodysplastic Syndrome pipeline landscape. It covers the pipeline drug profiles, including clinical and nonclinical stage products.As relapsed or refractory myelodysplastic syndrome (r/r MDS) continues to grow globally and contributes to comorbidities such as diabetes, cardiovascular disease, and certain cancers, there is an increasing need for safer and more effective treatments. According to DelveInsight, the r/r MDS pipeline includes more than five pharmaceutical and biotech companies developing over five therapeutic candidates, spanning various stages of clinical and preclinical development. This highlights the robust innovation and dedication to tackling this critical public health challenge.
DelveInsight's report, "Relapsed or Refractory Myelodysplastic Syndrome Pipeline Insight 2025," offers a comprehensive and strategic review of the R&D landscape. It examines clinical trial progress, emerging therapies, mechanisms of action, competitive positioning, and key company initiatives, serving as an essential resource for researchers, healthcare investors, and decision-makers seeking insights into the evolving r/r MDS therapeutics market and the breakthroughs shaping its future.
Explore the Cutting-Edge Landscape of Relapsed or Refractory Myelodysplastic Syndrome Drug Development [https://www.delveinsight.com/report-store/relapsed-or-refractory-myelodysplastic-syndrome-pipeline-insight?utm_source=abnewswire&utm_medium=market&utm_campaign=kpr]
Key Takeaways from the Relapsed or Refractory Myelodysplastic Syndrome Pipeline Report
*
DelveInsight's report on the relapsed or refractory myelodysplastic syndrome (r/r MDS) pipeline highlights a dynamic landscape with over five active companies developing more than five therapeutic candidates for r/r MDS treatment.
*
On October 24, 2023, the FDA approved ivosidenib (Tibsovo) for adult patients with r/r MDS carrying an IDH1 mutation. This approval was based on the AG120-C-001 trial, an open-label, single-arm, multicenter study with 18 adult participants, all receiving oral ivosidenib daily in 28-day cycles. The study reported complete responses in all patients.
*
Leading companies such as Cantex Pharmaceuticals, Novartis, Newave Pharmaceuticals, BioTheryX, Agios Pharmaceuticals, and others are actively developing new therapies to enhance the r/r MDS treatment landscape. Promising pipeline candidates include CX-01, LP-108, BTX-A51, among others, which are at various stages of development.
Relapsed or Refractory Myelodysplastic Syndrome Overview:
Myelodysplastic syndromes (MDS) are disorders marked by impaired blood cell production, resulting in cytopenias and genetic instability, which can eventually progress to acute myeloid leukemia (AML). Patients with MDS often experience significant clinical symptoms and face a higher risk of mortality due to both cytopenias and AML transformation. The main goals of MDS treatment are to relieve symptoms, slow disease progression, and improve survival and quality of life. Some individuals may remain symptom-free, with MDS discovered incidentally during routine blood tests. Treatment approaches for MDS include supportive care, pharmacologic therapies, and hematopoietic stem cell transplantation.
Download the Relapsed or Refractory Myelodysplastic Syndrome sample report to know in detail about the Relapsed or Refractory Myelodysplastic Syndrome treatment market [https://www.delveinsight.com/report-store/relapsed-or-refractory-myelodysplastic-syndrome-pipeline-insight?utm_source=abnewswire&utm_medium=market&utm_campaign=kpr]
Relapsed or Refractory Myelodysplastic Syndrome Pipeline Analysis
The Relapsed or Refractory Myelodysplastic Syndrome pipeline insights report 2025, provides insights into:
*
Provides comprehensive insights into key companies developing therapies in the Relapsed or Refractory Myelodysplastic Syndrome Market.
*
Categorizes Relapsed or Refractory Myelodysplastic Syndrome therapeutic companies by development stage: early, mid, and late-stage.
*
Highlights major companies involved in targeted therapy development, including both active and inactive (paused/discontinued) projects.
*
Reviews emerging Relapsed or Refractory Myelodysplastic Syndrome drugs under development based on:
*
Stage of development
*
Relapsed or Refractory Myelodysplastic Syndrome Route of administration
*
Target receptor
*
Monotherapy vs. combination therapy
*
Relapsed or Refractory Myelodysplastic Syndrome Mechanism of action
*
Molecular type
*
Offers detailed analysis of:
*
Company-to-company and company-academia collaborations
*
Relapsed or Refractory Myelodysplastic Syndrome Licensing agreements
*
Funding and investment activities supporting future Relapsed or Refractory Myelodysplastic Syndrome market advancement.
Unlock key insights into emerging Relapsed or Refractory Myelodysplastic Syndrome therapies and market strategies here: https://www.delveinsight.com/report-store/relapsed-or-refractory-myelodysplastic-syndrome-pipeline-insight?utm_source=abnewswire&utm_medium=market&utm_campaign=kpr
Relapsed or Refractory Myelodysplastic Syndrome Emerging Drugs
*
CX-01: Cantex Pharmaceuticals
CX-01 is a novel agent derived from unfractionated heparin with minimal anticoagulant activity. It interferes with critical protein pathways that enable AML cells to migrate to and persist in the bone marrow, where they are protected from chemotherapy. CX-01 also targets proteins associated with chemotherapy resistance and delayed platelet recovery, increasing AML cells' sensitivity to treatment and potentially improving patient outcomes. These mechanisms make CX-01 a promising therapy for myelodysplastic syndrome, multiple myeloma, and lymphomas. It is currently being tested in Phase 2 clinical trials for Myelodysplastic Syndrome.
*
LP-108: Newave Pharmaceuticals
This drug acts as an inhibitor of the proto-oncogene protein c-bcl-2 and is currently being evaluated in Phase 1 clinical trials for the treatment of relapsed or refractory Myelodysplastic Syndrome.
*
BTX-A51: BioTheryX
BTX-A51 is thought to target CK1-alpha in leukemic stem cells, as well as super-enhancer-associated proteins CDK7 and CDK9, thereby inhibiting the transcription of key genes that drive cancer growth. Preclinical studies in animal models have demonstrated promising outcomes, indicating that BTX-A51 may effectively eradicate AML stem cells and could have potential applications in multiple cancer types. The drug is currently being evaluated in Phase 1 clinical trials for the treatment of Myelodysplastic Syndrome.
Relapsed or Refractory Myelodysplastic Syndrome Pipeline Therapeutic Assessment
Relapsed or Refractory Myelodysplastic Syndrome Assessment by Product Type
- Mono
- Combination
- Mono/Combination
Relapsed or Refractory Myelodysplastic Syndrome By Stage
- Late-stage products (Phase III)
- Mid-stage products (Phase II)
- Early-stage product (Phase I) along with the details of
- Pre-clinical and Discovery stage candidates
- Discontinued & Inactive candidates
Relapsed or Refractory Myelodysplastic Syndrome Assessment by Route of Administration
- Oral
- Parenteral
- Intravenous
- Subcutaneous
- Topical
Relapsed or Refractory Myelodysplastic Syndrome Assessment by Molecule Type
- Recombinant fusion proteins
- Small molecule
- Monoclonal antibody
- Peptide
- Polymer
- Gene therapy
Download sample pages to get an in-depth assessment of the emerging Relapsed or Refractory Myelodysplastic Syndrome therapies and key Relapsed or Refractory Myelodysplastic Syndrome companies [https://www.delveinsight.com/report-store/relapsed-or-refractory-myelodysplastic-syndrome-pipeline-insight?utm_source=abnewswire&utm_medium=market&utm_campaign=kpr]
Table of Contents
1. Report Introduction
2. Executive Summary
3. Relapsed or Refractory Myelodysplastic Syndrome Current Treatment Patterns
4. Relapsed or Refractory Myelodysplastic Syndrome - DelveInsight's Analytical Perspective
5. Therapeutic Assessment
6. Relapsed or Refractory Myelodysplastic Syndrome Late-Stage Products (Phase-III)
7. Relapsed or Refractory Myelodysplastic Syndrome Mid-Stage Products (Phase-II)
8. Early Stage Products (Phase-I)
9. Pre-clinical Products and Discovery Stage Products
10. Inactive Products
11. Dormant Products
12. Relapsed or Refractory Myelodysplastic Syndrome Discontinued Products
13. Relapsed or Refractory Myelodysplastic Syndrome Product Profiles
14. Relapsed or Refractory Myelodysplastic Syndrome Key Companies
15. Relapsed or Refractory Myelodysplastic Syndrome Key Products
16. Dormant and Discontinued Products
17. Relapsed or Refractory Myelodysplastic Syndrome Unmet Needs
18. Relapsed or Refractory Myelodysplastic Syndrome Future Perspectives
19. Relapsed or Refractory Myelodysplastic Syndrome Analyst Review
20. Appendix
21. Report Methodology
Request the sample PDF to get detailed insights about the Relapsed or Refractory Myelodysplastic Syndrome pipeline reports offerings [https://www.delveinsight.com/report-store/relapsed-or-refractory-myelodysplastic-syndrome-pipeline-insight?utm_source=abnewswire&utm_medium=market&utm_campaign=kpr]
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
Media Contact
Company Name: DelveInsight Business Research LLP
Contact Person: Kritika Rehani
Email:Send Email [https://www.abnewswire.com/email_contact_us.php?pr=relapsedrefractory-myelodysplastic-syndrome-pipeline-2025-pioneering-clinical-developments-by-more-than-five-global-companies-delveinsight-highlighting-cantex-pharmaceuticals-novartis-newave-p]
Phone: +14699457679
Address:304 S. Jones Blvd #2432
City: Las Vegas
State: Nevada
Country: United States
Website: https://www.delveinsight.com/
Legal Disclaimer: Information contained on this page is provided by an independent third-party content provider. ABNewswire makes no warranties or responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you are affiliated with this article or have any complaints or copyright issues related to this article and would like it to be removed, please contact retract@swscontact.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Relapsed/Refractory Myelodysplastic Syndrome Pipeline 2025: Pioneering Clinical Developments by More Than Five Global Companies - DelveInsight | Highlighting Cantex Pharmaceuticals, Novartis, Newave P here
News-ID: 4192718 • Views: …
More Releases from ABNewswire
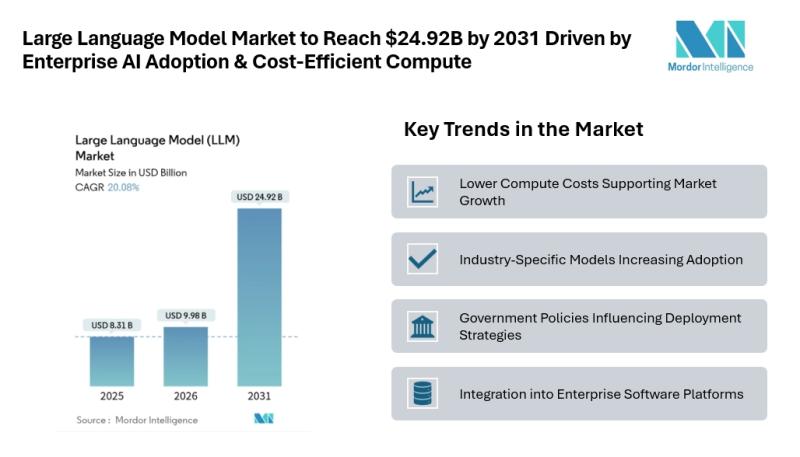
Large Language Model Market to Reach $24.92B by 2031 Driven by Enterprise AI Ado …
Mordor Intelligence has published a new report on the large language model market, offering a comprehensive analysis of trends, growth drivers, and future projections.
Large Language Model Market Outlook
According to Mordor Intelligence, the LLM market size [https://www.mordorintelligence.com/industry-reports/large-language-model-llm-market?utm_source=abnewswire] was valued at USD 8.31 billion in 2025 and is estimated to grow to USD 9.98 billion in 2026, reaching USD 24.92 billion by 2031 at a CAGR of 20.08% during the forecast period.…

Self Employed Tax Software UK: Why Freelancers and Sole Traders Are Switching to …
With Many individuals are seeking software that simplifies tax filing while ensuring full compliance with HMRC requirements. Manual spreadsheets and paper-based calculations are being replaced by real-time, automated systems that give users visibility over their tax position throughout the year. Among the platforms gaining traction is Pie, a UK-based digital tax app built specifically to support self-employed individuals with modern income needs.
LONDON, United Kingdom - February 19, 2026 - Demand…
CivicMail.org Reinvents Postcard Campaigns for Grassroots Advocacy
CivicMail.org aims to bring civic engagement back to basics through the power of pen, paper, and postage.
Image: https://www.abnewswire.com/upload/2026/02/2addd1e9e0381d7e2262e1edbb064123.jpg
CivicMail.org [https://civicmail.org/] has announced its launch to help Americans send real, physical postcards to their elected officials with just a few clicks, delivering personalized messages directly to the desks of decision-makers at the local, state, and federal levels.
Research shows [https://www.concordia.ca/news/stories/2021/09/24/personalized-messages-are-more-likely-to-get-a-response-from-politicians-new-research-finds.html] that physical mail carries more weight with elected officials than petitions, emails, or…

New Children's Story: The Story of Sharin' Bear
A Heartfelt Message Of Courage, Kindness, And The True Meaning Of Giving
A pleasant new story for children, The Story of Sharin' Bear by Sharon Woods , introduces families to a lovable little cub whose journey of bravery and compassion changes him into a representation of sharing for children globally.
Entrenched in adventure, innocence, and emotional growth, this uplifting tale offers an unforgettable reminder that even the smallest acts of kindness can…
More Releases for Myelodysplastic
Myelodysplastic Syndrome Drugs Market Research and Treatment Advancements
On April 11, 2025, Exactitude Consultancy., Ltd. announces the release of the report "Global Myelodysplastic Syndrome Drugs Market 2025 by Manufacturers, Regions, Type and Application, Forecast to 2034". The report is a detailed and comprehensive analysis presented by region and country, type and application. As the market is constantly changing, the report explores the competition, supply and demand trends, as well as key factors that contribute to its changing demands…
Prominent Myelodysplastic Syndrome (MDS) Drugs Market Trend for 2025: Innovative …
What Are the Projected Growth and Market Size Trends for the Myelodysplastic Syndrome (MDS) Drugs Market?
The myelodysplastic syndrome (MDS) drugs market has expanded steadily in recent years. It is projected to increase from $3.47 billion in 2024 to $3.73 billion in 2025, reflecting a CAGR of 7.5%. The past growth can be attributed to the aging population, advancements in targeted therapies, ongoing clinical trials, and increased awareness.
The myelodysplastic syndrome (MDS)…
Myelodysplastic Syndrome (MDS) Treatment Market Insights, Forecast to 2031
"Myelodysplastic Syndrome (MDS) Treatment Market 2024,"
The objectives outlined in the report are multifaceted and aimed at offering a comprehensive understanding of the Myelodysplastic Syndrome (MDS) Treatment market dynamics. These objectives encompass a meticulous analysis and forecast of the market's dimensions, encompassing both its value and volume aspects. Additionally, the report seeks to discern and delineate the market shares held by major segments within the Myelodysplastic Syndrome (MDS) Treatment industry, providing stakeholders…
Myelodysplastic Syndrome Pipeline Outlook Report 2024 (Updated)
DelveInsight's, "Myelodysplastic Syndrome Pipeline Insight 2024" report provides comprehensive insights about 120+ companies and 150+ pipeline drugs in Myelodysplastic Syndrome pipeline landscape. It covers the Myelodysplastic Syndrome pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Myelodysplastic Syndrome pipeline therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Key Takeaways from the Myelodysplastic…
Myelodysplastic Syndrome Market Size, Epidemiology, Analysis & Trends 2023-2033
Market Overview:
The myelodysplastic syndrome market is expected to exhibit a CAGR of 6.51% during 2023-2033. The myelodysplastic syndrome market report offers a comprehensive analysis of the market in the United States, EU5 (including Germany, Spain, Italy, France, and the United Kingdom), and Japan. It covers aspects such as treatment methods, drugs available in the market, drugs in development, the market share of various therapies, and the market's performance in the…
Myelodysplastic Syndrome Market to see Huge Growth by 2029
Myelodysplastic Syndrome Market was valued at USD 2.8 billion in 2022 and is expected to grow at a CAGR of 10% between 2023 and 2029. Important changes in the business allow key players to attain larger profits. This Myelodysplastic Syndrome Market study report is the best way to make changes with the help of entire market condition and metrics provided here. These metrics also allow getting ahead in the industry.…
