Press release
Track-and-Trace and Serialization Market Grows Amid Regulatory Pressures, Says Persistence Market Research
Pharmaceutical companies, like many others, face a labyrinth of challenges related to supply chain management. Security lapses stemming from theft, product recalls, or diversions can trigger severe consequences for patient safety and public health-not merely on a national level but globally. The ripple effect extends to pharmaceutical companies themselves, eroding profitability, tarnishing goodwill, and jeopardizing planned research initiatives. As both the industry and regulators increasingly recognize the critical importance of safeguarding supply chains, the Track-and-Trace and Serialization Market has emerged as a vital solution.With regulatory bodies mandating compliance across various nations, stakeholders in the pharmaceutical supply chain have no choice but to implement serialization and traceability solutions. Rising incidences of product theft have turned the spotlight on advanced security technologies. Early solutions like tamper-proof packaging and holograms, once innovative, are no longer sufficient as counterfeiters become increasingly sophisticated. Today's approach focuses on assigning unique identifiers to even the smallest sale unit-such as a bottle-and tracking these serial numbers throughout the entire supply chain to enable instant authentication. While countries like China and Turkey have led the way in implementing such systems, nations including India and the United States are swiftly catching up, with global adoption expected to accelerate in the coming years.
✅Get a Sample Copy of Research Report (Use Corporate Mail id for Quick Response): https://www.persistencemarketresearch.com/samples/19702
Overview of the Market
The Track-and-Trace and Serialization Market has evolved into a crucial pillar of pharmaceutical supply chain security. With governments and industry stakeholders prioritizing patient safety, regulatory compliance, and brand protection, serialization technologies have transitioned from optional to mandatory. Persistence Market Research emphasizes how serialization allows for unique product identification and traceability throughout distribution channels, helping combat counterfeiting and theft. This market has witnessed significant momentum, fueled by stringent global regulations and a growing focus on digitalized supply chain operations.
Among the key segments, software solutions are leading the market, as they form the backbone of managing and analyzing serialization data. North America emerges as the leading geographical region, owing to robust regulatory mandates like the U.S. Drug Supply Chain Security Act (DSCSA). The region's well-developed pharmaceutical industry and sophisticated technological infrastructure further contribute to its dominance. Growing regulatory requirements across Europe and Asia-Pacific regions are propelling these markets as well, setting the stage for global expansion.
Key Highlights from the Report
➤ Rising pharmaceutical counterfeiting losses push demand for serialization solutions.
➤ North America dominates the market due to stringent regulatory frameworks.
➤ Software solutions lead in market share as the backbone for serialization data handling.
➤ High implementation costs pose challenges for market adoption among smaller players.
➤ Regulatory mandates in the EU and U.S. drive compliance-based market growth.
➤ Growing awareness of patient safety fosters investment in advanced tracking technologies.
Market Segmentation
The Track-and-Trace and Serialization Market is segmented based on product type, including hardware, software, and services. Hardware encompasses equipment like barcode scanners, labeling machines, and printers crucial for applying and reading unique identifiers. However, software solutions constitute the lion's share, enabling management, analysis, and compliance reporting of serialized data across complex supply chain networks. Services, such as consultancy and implementation support, are also critical, helping pharmaceutical companies navigate intricate regulatory landscapes and tailor solutions to their specific operational needs.
Segmentation also extends into end-user industries. Pharmaceutical manufacturers represent the largest segment due to the pressing need for compliance with evolving laws across major regions. Contract manufacturing organizations (CMOs) are increasingly investing in serialization infrastructure to maintain business with pharmaceutical brands. Distributors and wholesalers are vital participants as they need systems capable of verifying product authenticity and managing returns efficiently. Retail pharmacies and healthcare providers are gradually adopting serialization tools to ensure patient safety and protect brand integrity.
Regional Insights
North America stands at the forefront of the Track-and-Trace and Serialization Market, driven by stringent regulations such as the DSCSA in the U.S. The region's mature pharmaceutical ecosystem, paired with advanced technological infrastructure, has fostered rapid adoption of serialization solutions. Pharmaceutical companies are under mounting pressure to comply with regulations, creating a robust demand for hardware, software, and services supporting serialization initiatives.
In Europe, regulatory requirements under the Falsified Medicines Directive (FMD) have significantly boosted market momentum. Countries in Western Europe, including Germany, the U.K., and France, are leading the charge, adopting sophisticated serialization practices to counter counterfeit drugs. Meanwhile, Asia-Pacific markets like China and India are emerging swiftly, as governments enforce laws mandating serialization for both domestic consumption and exports.
Market Drivers
Maintaining the integrity of increasingly complex pharmaceutical supply chains has emerged as a central driver of the Track-and-Trace and Serialization Market. Persistence Market Research highlights that the pharmaceutical industry loses tens of billions annually due to counterfeiting and cargo theft. Such losses extend beyond revenue, jeopardizing patient safety and potentially leading to product recalls that damage brand reputation. Serialization provides a mechanism for identifying and segregating products bound for different markets, enhancing both product safety and regulatory compliance.
Another powerful driver is regulatory pressure. The U.S. and EU, collectively the world's largest pharmaceutical markets, have introduced stringent serialization requirements. The EU's FMD and the U.S. DSCSA have set tight compliance deadlines, compelling pharmaceutical companies to act swiftly. Compliance is non-negotiable for firms looking to maintain access to these lucrative markets. This regulatory momentum is cascading globally, as developing economies increasingly adopt similar measures to protect patients and secure supply chains.
Beyond security and compliance, technological advancements serve as a catalyst for market growth. Cloud-based platforms, advanced analytics, and IoT integration are transforming the track-and-trace landscape. These innovations simplify complex data management tasks, helping companies gain insights into supply chain dynamics and detect potential threats in real time. As pharmaceutical manufacturers look to modernize their operations, serialization becomes an integral part of digital transformation strategies.
Market Restraints
Despite the numerous advantages, the Track-and-Trace and Serialization Market faces significant challenges, chief among them being high implementation costs. Upgrading packaging lines to accommodate serialization demands a substantial financial outlay, ranging from hundreds of thousands to millions of dollars per line. Smaller pharmaceutical firms and CMOs often struggle with such investments, risking operational disruptions during equipment upgrades and production downtime.
Data management emerges as another formidable hurdle. Serialization generates vast quantities of data that must be stored, managed, and retrieved efficiently to comply with regulations. Ensuring data security, traceability, and integration with existing business systems can overwhelm companies lacking advanced IT capabilities. Pharmaceutical firms face the dual challenge of maintaining compliance while avoiding disruptions to daily business operations, leading many to seek specialized partners for managing serialization infrastructure.
Moreover, the lack of global harmonization in serialization standards poses further constraints. Different countries enforce diverse regulations, formats, and reporting requirements, complicating global supply chain operations for multinational pharmaceutical companies. Navigating this patchwork of compliance obligations increases costs and operational complexity, creating another barrier to seamless implementation.
✅Request for Customization of the Research Report: https://www.persistencemarketresearch.com/request-customization/19702
Market Opportunities
Amidst challenges, the Track-and-Trace and Serialization Market is ripe with opportunities. Growing demand for supply chain visibility and transparency opens new avenues for solution providers. Pharmaceutical companies increasingly seek systems capable of not just regulatory compliance but also delivering business intelligence and process optimization. Advanced track-and-trace systems can provide valuable insights into demand forecasting, inventory management, and market trends, creating competitive advantages beyond compliance.
Emerging markets offer fertile ground for market expansion. Countries across Asia-Pacific, Latin America, and the Middle East are tightening their regulatory environments, presenting significant opportunities for vendors specializing in affordable and scalable serialization solutions. As governments invest in combating counterfeit drugs, pharmaceutical companies are more inclined to allocate budgets toward serialization initiatives, unlocking new customer bases for global solution providers.
Additionally, the integration of serialization with emerging technologies, such as blockchain, AI, and machine learning, presents transformative potential. Blockchain, in particular, offers immutable data records, enhancing the trust and security of supply chain transactions. Solution providers capable of blending serialization systems with these technologies stand to capture significant market share as pharmaceutical companies pursue innovation and digital transformation.
Reasons to Buy the Report
✔ Gain strategic insights into regulatory trends shaping global serialization mandates.
✔ Understand market dynamics, drivers, and challenges affecting investment decisions.
✔ Access comprehensive segmentation data to identify high-growth opportunities.
✔ Evaluate leading regional markets and forecast trends to support market entry strategies.
✔ Benchmark competitors and discover recent innovations transforming the market landscape.
Company Insights
The Track-and-Trace and Serialization Market is home to several prominent players dedicated to innovating solutions for supply chain security and regulatory compliance. Leading names include:
✦ TraceLink
✦ Infosys
✦ Bosch Packaging
✦ SAP
✦ Axway
✦ Xyntec Inc.
Two notable recent developments highlight the market's ongoing evolution. TraceLink announced an AI-powered platform designed to deliver advanced supply chain analytics, enabling pharmaceutical companies to proactively detect anomalies and optimize operations. Meanwhile, Bosch Packaging expanded its serialization equipment offerings, introducing high-speed modular solutions tailored for CMOs and mid-sized pharmaceutical firms, helping them meet compliance requirements without significant infrastructure overhauls.
The Road Ahead
Looking forward, the Track-and-Trace and Serialization Market appears poised for continued expansion. With patient safety at the forefront of global healthcare priorities and the relentless evolution of regulatory environments, serialization is no longer a luxury-it's an operational imperative. Persistence Market Research emphasizes that companies that invest early in robust serialization infrastructures stand to benefit from not only compliance but also greater supply chain transparency, efficiency, and brand protection.
As technology continues to advance and global markets grow more interconnected, the demand for sophisticated track-and-trace solutions will only intensify. Players who innovate rapidly, maintain agility in responding to regulatory shifts, and integrate emerging technologies into their offerings will likely emerge as market leaders in this essential domain.
The market's journey is far from over. As counterfeiters become more sophisticated, the technologies used to combat them must evolve at an equal-if not faster-pace. The Track-and-Trace and Serialization Market is not merely an industry trend; it is a cornerstone of the pharmaceutical industry's future, ensuring that medicines remain safe, effective, and trustworthy from factory floor to patient hands.
Contact Us:
Persistence Market Research
G04 Golden Mile House, Clayponds Lane
Brentford, London, TW8 0GU UK
USA Phone: +1 646-878-6329
UK Phone: +44 203-837-5656
Email: sales@persistencemarketresearch.com
Web: https://www.persistencemarketresearch.com
About Persistence Market Research:
At Persistence Market Research, we specialize in creating research studies that serve as strategic tools for driving business growth. Established as a proprietary firm in 2012, we have evolved into a registered company in England and Wales in 2023 under the name Persistence Research & Consultancy Services Ltd. With a solid foundation, we have completed over 3600 custom and syndicate market research projects, and delivered more than 2700 projects for other leading market research companies' clients.
Our approach combines traditional market research methods with modern tools to offer comprehensive research solutions. With a decade of experience, we pride ourselves on deriving actionable insights from data to help businesses stay ahead of the competition. Our client base spans multinational corporations, leading consulting firms, investment funds, and government departments. A significant portion of our sales comes from repeat clients, a testament to the value and trust we've built over the years.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Track-and-Trace and Serialization Market Grows Amid Regulatory Pressures, Says Persistence Market Research here
News-ID: 4096835 • Views: …
More Releases from Persistence Market Research
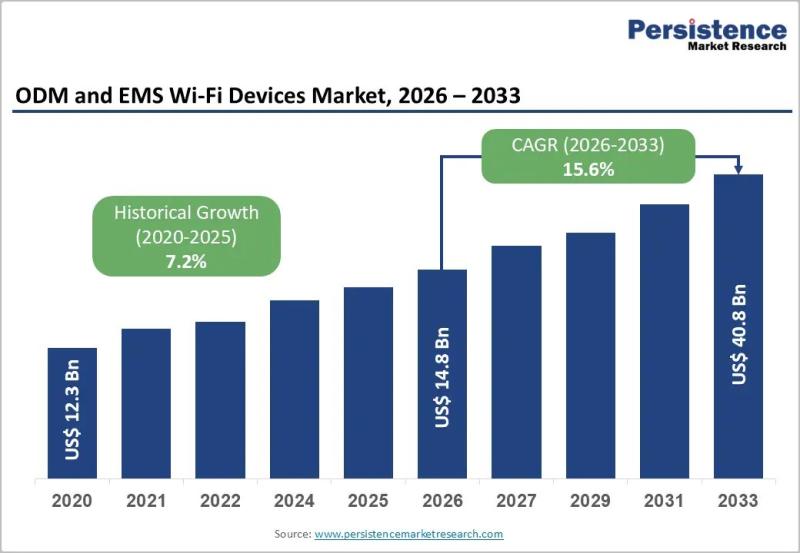
ODM and EMS Wi-Fi Devices Market to Reach US$ 40.8 Billion by 2033 at 15.6% CAGR
The global ODM and EMS Wi-Fi devices market is projected to be valued at US$ 14.8 billion in 2026 and is forecast to surge to US$ 40.8 billion by 2033, registering a robust CAGR of 15.6% between 2026 and 2033. This rapid growth reflects the accelerating demand for advanced wireless connectivity solutions across residential, enterprise, and industrial environments. The expansion of 5G infrastructure, enterprise digital transformation strategies, and large-scale IoT…
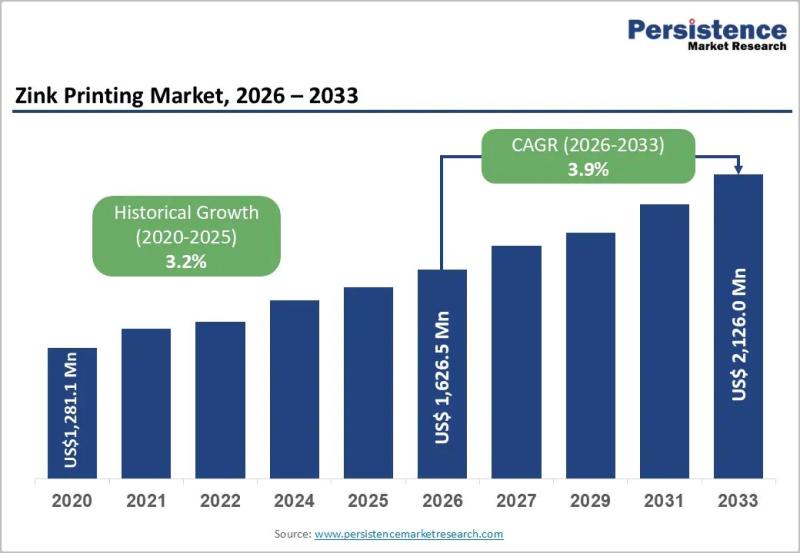
Zink Printing Market to Reach US$ 2,126.0 Million by 2033 at 3.9% CAGR
Zink Printing Market Size and Trends Analysis
The global Zink printing market is projected to be valued at US$ 1,626.5 million in 2026 and is expected to reach US$ 2,126.0 million by 2033, expanding at a CAGR of 3.9% between 2026 and 2033. Zink (Zero Ink) printing technology eliminates the need for ink cartridges by using heat-activated color crystals embedded within specialized paper. This innovation has positioned Zink printers as a…
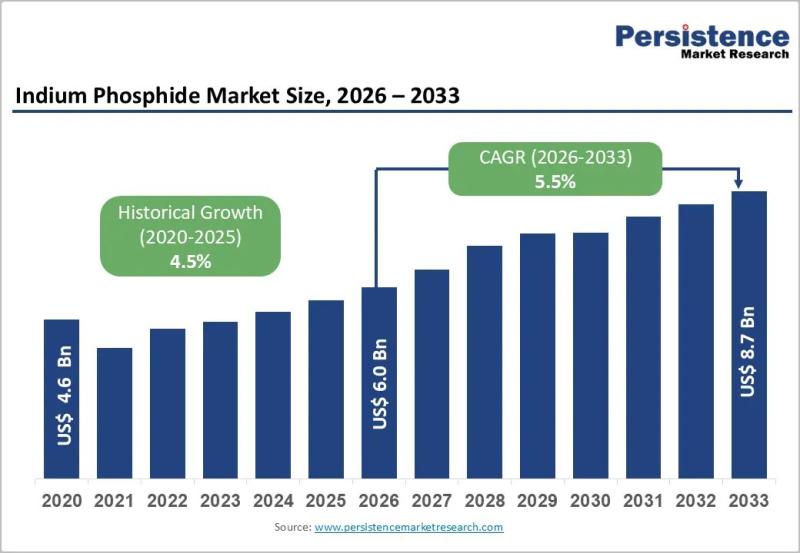
Indium Phosphide Market to Reach US$ 8.7 Billion by 2033 at 5.5% CAGR
The global Indium Phosphide (InP) market is poised for steady expansion, with its valuation expected to reach US$ 6.0 billion by 2026 and further grow to US$ 8.7 billion by 2033, registering a CAGR of 5.5% between 2026 and 2033. Indium phosphide, a high-performance compound semiconductor material, is widely used in optoelectronics, high-frequency electronics, and photonic integrated circuits (PICs). The market growth is largely fueled by the accelerating deployment of…

Roasted Corn Market to Reach $5.5B by 2033 Driven by Rising Snack Demand
The global roasted corn market is poised for steady expansion, driven by rising consumer preference for convenient, ready-to-eat snacks and increasing awareness of healthier alternatives to traditional fried snack options. Current market estimates indicate that the roasted corn market is valued at approximately US$ 3.8 billion in 2026 and is projected to reach US$ 5.5 billion by 2033, reflecting a compound annual growth rate (CAGR) of 5.4% between 2026 and…
More Releases for Serialization
Carton Serialization Machine Market: Competitive Dynamics & Global Outlook 2025
The latest research report"Global Carton Serialization Machine Market Growth 2025-2031" studied by LP Information offers a comprehensive overview of theCarton Serialization Machine market, providing insights into its drivers, restraints, and future trends. This study utilizes a comprehensive and thorough research methodology to provide an objective perspective on the global Carton Serialization Machine industry, helping decision-makers make informed business decisions.
According to the report "Global Carton Serialization Machine Market Growth Trends 2025-2031"…
Pharmaceutical Serialization Market Share, Trends & Forecasts By 2031
The global Pharmaceutical Serialization Market has been sailing through robust waters, witnessing a valuation of US$ 15.1 billion 2022. As we set our sights on the horizon, the market is poised to embark on a journey of steady growth, with a projected Compound Annual Growth Rate (CAGR) of 5.1% from 2023 to 2031. By the end of 2031, the industry is expected to anchor at an impressive US$ 24.8 billion.
The…
Pharmaceutical Serialization Software Market Size, Players, Revenue Analysis 202 …
Pharmaceutical Serialization Software Market Overview 2023:
The Pharmaceutical Serialization Software Market 2023 Report makes available the current and forthcoming technical and financial details of the industry. It is one of the most comprehensive and important additions to the Prudent Markets archive of market research studies. It offers detailed research and analysis of key aspects of the Pharmaceutical Serialization Software market. This report explores all the key factors affecting the growth of…
Product Serialization Software Market Size, Players, Revenue Analysis 2023-2030
Product Serialization Software Market Overview 2023:
The Product Serialization Software Market 2023 Report makes available the current and forthcoming technical and financial details of the industry. It is one of the most comprehensive and important additions to the Prudent Markets archive of market research studies. It offers detailed research and analysis of key aspects of the Product Serialization Software market. This report explores all the key factors affecting the growth of…
Pharmaceutical Serialization Solution Market Growing Trends and Technology Forec …
The report offers detailed coverage of Pharmaceutical Serialization Solution and main market trends. The market research includes historical data from 2016 - 2021 and forecast market data from 2022 - 2030, demand, application details, price trends, and company shares of the leading market by geography. The report splits the market size, by volume and value, on the basis of application type and geography. In addition to this data, the report…
Read Dotted Barcodes from TIFF Images & Enhanced XML Serialization using Java
What's New in this Release?
Aspose team is pleased to announce the new release of Aspose.BarCode for Java 18.12. The new release of Aspose.BarCode adds the ability to read dotted barcodes from Tiff images. The code sample is provided on blog announcement page that show how to read such barcodes. Other than the ability to read dotted barcodes from Tiff images, XML serialization was enhanced and made compatible between .NET…
