Press release
Ensuring Analytical Excellence: Explore Aquigen Bio's Halcinonide Impurity Standards
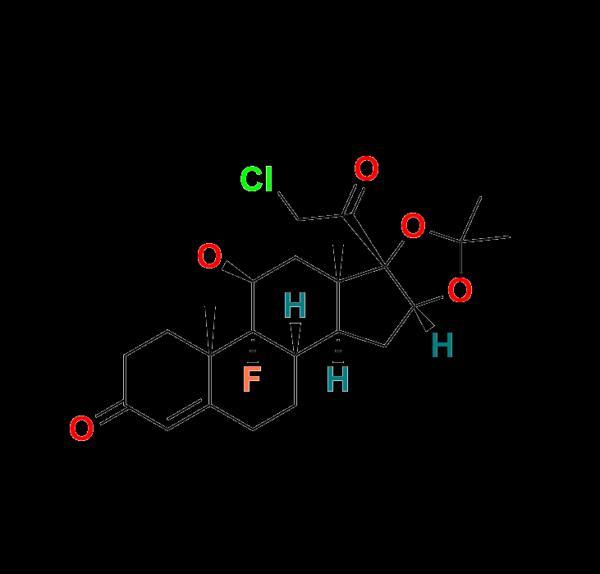
In the pursuit of pharmaceutical quality and regulatory compliance, accurate impurity profiling is critical. At Aquigen Bio, we're committed to supporting your analytical needs with precision-manufactured Halcinonide Impurity Standards, enabling researchers, formulators, and QC professionals to maintain the highest benchmarks of drug purity and safety.Why Halcinonide Impurity Standards Matter
Halcinonide, a potent corticosteroid used to treat various skin conditions, must be rigorously tested for impurities to meet global regulatory guidelines like ICH, USP, and FDA requirements. Identifying and quantifying these impurities is essential in ensuring product efficacy and patient safety.
Aquigen Bio offers a comprehensive set of Halcinonide impurity standards designed for:
Method validation
System suitability testing
Stability studies
Routine quality control
Each standard is developed with high purity, detailed characterization, and comes with a Certificate of Analysis (CoA) to support traceability and compliance.
Featured Products in the Halcinonide Category
Explore our key offerings to support your impurity profiling studies:
Halcinonide (API Standard)
For analytical testing, calibration, and reference.
https://aquigenbio.com/product/halcinonide/
Halcinonide Impurity 10
Ideal for use in HPLC, GC, and mass spectrometry analysis.
https://aquigenbio.com/product/halcinonide-impurity-10/
Halcinonide Impurity 15
Well-suited for impurity profiling and stability studies.
https://aquigenbio.com/product/halcinonide-impurity-15/
Each product is carefully synthesized and validated under strict quality controls to ensure accuracy in pharmaceutical R&D and manufacturing environments.
Why Choose Aquigen Bio?
GMP-like production standards
Analytical-grade purity
Global shipping capabilities
Rapid customer support
Customized impurity synthesis available
With our broad and ever-expanding catalog of impurity standards, Aquigen Bio remains a trusted partner for pharmaceutical companies, CROs, and academic institutions worldwide.
Boost your quality control confidence.
Trust Aquigen Bio's Halcinonide Impurity Standards for unmatched precision and consistency in your analytical processes.
For bulk inquiries or technical documentation, connect with us today at info@aquigenbio.com or visit our full product range at
https://aquigenbio.com/products/impurity-standards/halcinonide/
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Visit: www.aquigenbio.com
Aquigen Bio Sciences stands as India's premier resource for high-quality N-Nitroso impurity standards, impelling advancements in pharmaceutical research and manufacturing. Specializing in providing precise impurity standards - including degradation impurities, process impurities, and deuterated isotopes - Aquigen Bio Sciences empowers the industry to comply with the most rigorous global regulations. The contract research organization's commitment to precision and reliability makes it the trusted partner for addressing complex issues like N-Nitroso impurities in pharmaceuticals.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Ensuring Analytical Excellence: Explore Aquigen Bio's Halcinonide Impurity Standards here
News-ID: 4090412 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Halcinonide
Aquigen Bio Expands its Impurity Standards Portfolio with Malathion & Dithiodisu …
Aquigen Bio Sciences, a leading pharmaceutical impurity‐standards manufacturer based in India, continues to broaden its catalog with new high‐quality analytical reference standards. These include Malathion EP Impurity A, O,O‐Dimethyl Malathion, and Tetraethyl Dithiodisuccinate, designed to support rigorous research, validation, and regulatory compliance in agrochemical and pharmaceutical analysis.
Introducing New Additions to the Impurity Standards Line-up
https://aquigenbio.com/product/tetraethyl-dithiodisuccinate/
Malathion EP Impurity A
This compound-diethyl (2R)-2‐((methoxy(methylthio)phosphoryl)thio)succinate-offers traceability to pharmacopeial standards, such as USP or EP. It is…
Aquigen Bio Expands Its Impurity Standards Portfolio with High-Purity Compounds …
Aquigen Bio, a leading global supplier of pharmaceutical impurity standards, is proud to announce the expansion of its Impurity Standards portfolio with newly developed, high-purity reference materials for Halcinonide, Ibandronate, Kaempferol, and Labetalol. These additions reflect the company's commitment to supporting pharmaceutical quality control, regulatory compliance, and R&D efforts worldwide.
Newly Introduced Impurity Standards
1. Halcinonide Impurity 14
https://aquigenbio.com/product/halcinonide-impurity-14/
Derived from the potent corticosteroid Halcinonide, this impurity is offered with detailed characterization data…
Halcinonide Market 2022- Global Key Players, Trends, Share, Industry Size, Growt …
LOS ANGELES, United States: QY Research has recently published a new report, titled Global Halcinonide Market Outlook 2022 This report offers an in-depth analysis of the global Halcinonide market taking into account market dynamics, segmentation, geographical expansion, and competitive landscape, and various other key aspects. The market analysts who have prepared the report have thoroughly studied the global Halcinonide market and have offered reliable and accurate data. They understand the needs of…
Topical Corticosteroids Market is Trending with CAGR of 5.8 % by 2023, Key Playe …
Market Research Future has announced the addition of the "Global Topical Corticosteroids Market Research Report which gives Market Size, Shares and Forecasts till 2023. The Topical Corticosteroids Industry Report Provide analysis and evaluation of the current and prospective profitability.
Topical Corticosteroids Market -Highlights
Topical corticosteroids (steroids) are medication and available in various forms such as creams, lotions, and solutions. They are directly applied to the skin to reduce inflammation and irritation.
Increasing prevalence…
Global Topical Corticosteroids Market 2018 by Top Vendors - AstraZeneca plc, Gla …
Market Research Future has announced the addition of the "Global Topical Corticosteroids Market Research Report which gives Market Size, Shares and Forecasts till 2023. The Topical Corticosteroids Industry Report Provide analysis and evaluation of the current and prospective profitability.
Topical Corticosteroids Market -Highlights
Topical corticosteroids (steroids) are medication and available in various forms such as creams, lotions, and solutions. They are directly applied to the skin to reduce inflammation and irritation.
Increasing prevalence…
Global Topical Corticosteroids Market 2018 by Top Vendors - AstraZeneca plc, Gla …
Market Research Future Present’s “Global Topical Corticosteroids Market Size, Status and Forecast 2023” magnify the decision making potentiality and helps to create an effective counter strategies to gain competitive advantage. The report provides an extensive research and detailed analysis of the present market along with future outlook.
Highlights of the Topical Corticosteroids Market
Topical corticosteroids (steroids) are medication and available in various forms such as creams, lotions, and solutions. They are directly applied…