Press release
Biosimilars Market to Reach US$ 100.5 Bn by 2032 Driven by Cost Savings and Patent Expiry
✅ Biosimilars Market: Unlocking Access and Affordability in Modern HealthcareThe biosimilars market has emerged as a vital component of the global biopharmaceutical landscape, providing cost-effective alternatives to biologic therapies without compromising on efficacy or safety. Biosimilars are biologic medical products highly similar to already approved reference biologics. Their adoption is accelerating due to increasing demand for affordable treatments for chronic diseases such as cancer, diabetes, and autoimmune disorders. With mounting healthcare costs and the expiration of patents for blockbuster biologics, biosimilars are becoming a cornerstone in delivering accessible and sustainable healthcare.
The global biosimilars market is projected to grow significantly, from US$ 39.1 billion in 2024 to US$ 100.5 billion by 2032, at a robust CAGR of 14.1%. This surge is largely driven by rising incidences of chronic diseases, cost containment strategies by healthcare systems, and supportive regulatory pathways across developed and emerging economies. Among the therapeutic segments, oncology biosimilars currently dominate due to high cancer prevalence and the need for affordable biologic alternatives. Regionally, Europe leads the global biosimilars market, attributed to its mature regulatory framework, early biosimilar adoption, and emphasis on cost-effective healthcare solutions.
Get a Sample PDF Brochure of the Report (Use Corporate Email ID for a Quick Response): https://www.persistencemarketresearch.com/samples/2782
✅ Key Highlights from the Report
➤The global biosimilars market is projected to reach US$ 100.5 Bn by 2032.
➤Europe is currently the leading region in terms of biosimilar adoption and sales.
➤Oncology holds the largest market share among therapeutic segments.
➤Biosimilars adoption is driven by increasing chronic disease burden and patent expirations.
➤Supportive regulatory environments are encouraging biosimilar development and approvals.
➤Leading players are actively engaged in strategic collaborations and product launches.
✅ 📌 What is the difference between biosimilars and generics?
Biosimilars and generics are both types of follow-on drugs, but they differ fundamentally in structure, complexity, and manufacturing. Generics are chemical-based and identical copies of small-molecule drugs, whereas biosimilars are highly similar but not identical to original biologic products. This is due to the inherent complexity of biologics, which are large, protein-based molecules derived from living organisms. Because of this complexity, biosimilars undergo a more rigorous approval process involving analytical, preclinical, and clinical testing to demonstrate similarity in efficacy, safety, and immunogenicity. Moreover, biosimilars are not considered interchangeable by default, unlike generics, which can be automatically substituted. The production of biosimilars is also more complex and cost-intensive than generics, necessitating advanced biotechnological expertise. As healthcare systems strive for cost-effective solutions, understanding the distinction between biosimilars and generics is crucial for informed decision-making and efficient policy implementation.
✅ Market Segmentation Analysis
Biosimilars can be segmented based on product type into monoclonal antibodies, recombinant hormones (e.g., insulin, erythropoietin), and immunomodulators. Among these, monoclonal antibodies dominate due to their extensive use in treating cancer and autoimmune diseases. Their higher cost has made them a prime target for biosimilar development, offering potential savings for both healthcare systems and patients. Insulin biosimilars are also seeing increased demand due to the global rise in diabetes prevalence.
By end-user, the market is segmented into hospitals, clinics, and homecare settings. Hospitals account for the largest share owing to their extensive use of biologics in cancer, renal, and autoimmune care. However, the rise in outpatient treatments and patient preference for convenience are driving the uptake of biosimilars in homecare and retail pharmacy settings, indicating evolving treatment models.
✅ Regional Insights: Emerging and Leading Geographies
Europe continues to lead the global biosimilars market, driven by robust regulatory support from the European Medicines Agency (EMA), early biosimilar approvals, and public healthcare cost containment measures. Nations such as Germany, the UK, and France have adopted favorable reimbursement policies, encouraging physician and patient acceptance.
Asia Pacific is witnessing rapid biosimilars market expansion, particularly in India, China, and South Korea. These countries benefit from strong local manufacturing capabilities, a large patient pool, and increasing government investment in biologics and biosimilars. Additionally, regulatory reforms in these regions are enabling faster product approvals and international partnerships.
✅ Market Dynamics: Drivers, Restraints, and Opportunities
Market Drivers
The global rise in chronic and lifestyle-related diseases such as cancer, rheumatoid arthritis, and diabetes is propelling demand for affordable biologic treatments. Additionally, the expiration of patents for high-cost reference biologics opens opportunities for biosimilar competition. Governments and payers are promoting biosimilar uptake to reduce healthcare costs, further driving market momentum.
Market Restraints
Despite strong growth prospects, the market faces challenges including complex manufacturing processes, high initial investment, and lengthy regulatory approvals. Physician and patient skepticism in some regions also slows down adoption. Intellectual property litigation and limited interchangeability policies can delay biosimilar launches and hinder market expansion.
Market Opportunities
With the increasing aging population and rising healthcare access in emerging economies, biosimilars offer a vast untapped opportunity. Expansion of distribution networks, improved physician awareness, and strategic collaborations between big pharma and biosimilar developers are creating fertile ground for growth. Technological advancements in analytical and bioprocessing tools are also streamlining development and reducing costs.
✅ Reasons to Buy the Report
☑Gain a comprehensive understanding of the current and projected market dynamics.
☑Identify key growth drivers, restraints, and opportunities shaping the industry.
☑Evaluate regional performance and emerging opportunities in developing economies.
☑Track strategic moves of major market players including collaborations and launches.
☑Make informed business decisions using accurate forecasts and segmentation insights.
✅ Company Insights
✦Amgen Inc.
✦Pfizer Inc.
✦Samsung Bioepis Co., Ltd.
✦Sandoz (A Novartis Division)
✦Biocon Limited
✦Celltrion Inc.
✦Coherus BioSciences, Inc.
✦Mylan N.V.
✦Fresenius Kabi
✦Dr. Reddy's Laboratories Ltd.
■In 2024, Biocon Biologics completed the acquisition of Viatris' biosimilars business, significantly expanding its global footprint.
■Amgen received FDA approval for its biosimilar Amjevita (adalimumab-atto), making it a competitor to Humira in the U.S. market.
✅ Conclusion
The biosimilars market is witnessing dynamic evolution, fueled by the need for cost-effective biologic alternatives, favorable regulatory frameworks, and increasing chronic disease burden. As patent cliffs continue and healthcare systems prioritize affordability, biosimilars offer a promising solution to democratize access to life-saving therapies. With Europe maintaining its lead and Asia Pacific gaining momentum, the global market is set to undergo transformative growth. Investing in biosimilars not only makes economic sense but also enhances therapeutic access on a global scale, setting a strong foundation for the future of healthcare innovation.
✅About Persistence Market Research:
At Persistence Market Research, we specialize in creating research studies that serve as strategic tools for driving business growth. Established as a proprietary firm in 2012, we have evolved into a registered company in England and Wales in 2023 under the name Persistence Research & Consultancy Services Ltd. With a solid foundation, we have completed over 3600 custom and syndicate market research projects, and delivered more than 2700 projects for other leading market research companies' clients.
Our approach combines traditional market research methods with modern tools to offer comprehensive research solutions. With a decade of experience, we pride ourselves on deriving actionable insights from data to help businesses stay ahead of the competition. Our client base spans multinational corporations, leading consulting firms, investment funds, and government departments. A significant portion of our sales comes from repeat clients, a testament to the value and trust we've built over the years.
Contact Us:
Persistence Market Research
G04 Golden Mile House, Clayponds Lane
Brentford, London, TW8 0GU UK
USA Phone: +1 646-878-6329
UK Phone: +44 203-837-5656
Email: sales@persistencemarketresearch.com
Web: https://www.persistencemarketresearch.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Biosimilars Market to Reach US$ 100.5 Bn by 2032 Driven by Cost Savings and Patent Expiry here
News-ID: 4082562 • Views: …
More Releases from Persistence Market Research

Bicycle Spokes Market Set for Strong Growth at 5.4% CAGR Through 2032 - Persiste …
The global bicycle spokes market is rapidly gaining traction as bicycles continue to be adopted as preferred choices for commuting, fitness, recreation, and eco‐friendly mobility. The global bicycle spokes market size is likely to be valued at US$2.9 billion in 2025 and is expected to reach US$4.2 billion by 2032, registering a steady CAGR of 5.4 % between 2025 and 2032.
➤ Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/30615
Bicycle…

Herbal Toothpaste Market Growth Poised at 6.5% CAGR Through 2033 Amid Rising Hea …
The global oral care industry is undergoing a transformational shift as consumers increasingly prioritize natural, chemical free alternatives. Central to this transformation is the herbal toothpaste market, which is rapidly emerging as a mainstream segment driven by rising health consciousness, sustainability trends, and demand for botanical formulations. The global herbal toothpaste market size is likely to be valued at US$ 2.6 billion in 2026 and is projected to reach US$…

Dead Sea Mud Cosmetics Market Set for Steady Expansion Amid Rising Demand for Na …
The global beauty and personal care industry continues to evolve as consumers shift toward natural, mineral-based, and wellness-oriented skincare solutions. Among these, Dead Sea mud cosmetics have gained strong traction for their mineral content and perceived therapeutic benefits. According to industry estimates, the global dead sea mud cosmetics market is likely to be valued at US$1.5 billion in 2026 and is projected to reach US$2.3 billion by 2033, expanding at…
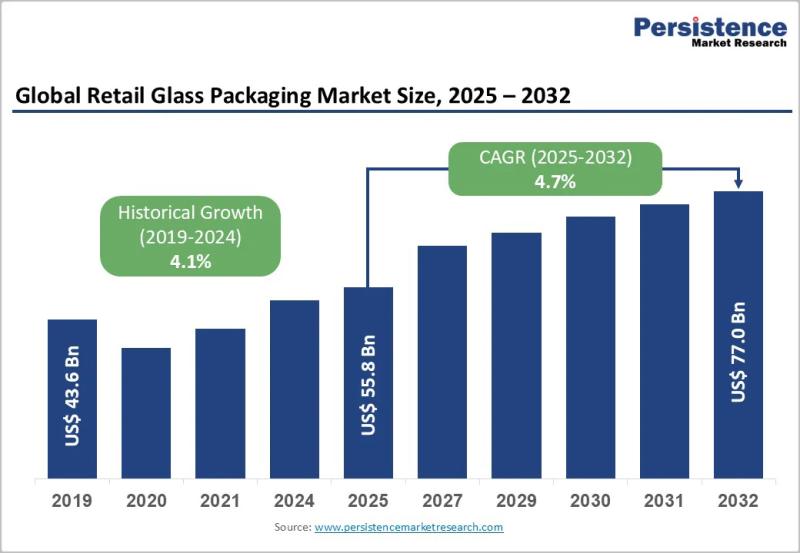
Retail Glass Packaging Market Projected to Reach US$77.0 Billion by 2032 at 5.3% …
The retail glass packaging market continues to play a crucial role in the global packaging ecosystem, particularly across food, beverage, cosmetics, and pharmaceutical retail channels. Glass packaging remains a preferred solution due to its premium appearance, chemical inertness, recyclability, and ability to preserve product integrity. As consumers increasingly prioritize sustainability, safety, and high quality packaging, retail glass packaging has regained strategic importance across both developed and emerging economies. Brands are…
More Releases for Biosimilars
Transformative Trends Impacting the Biosimilars Market Landscape: Innovative Pro …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
Biosimilars Market Size Valuation Forecast: What Will the Market Be Worth by 2025?
The dimensions of the biosimilars market have been rapidly expanding over the last couple of years. The escalation, from a worth of $18.65 billion in 2024 to an estimated value of $21.95 billion in 2025, denotes…
Evolving Market Trends In The Biosimilars Industry: Innovative Product Launched …
The Biosimilars Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Expected Biosimilars Market Size During the Forecast Period?
The dimension of the biosimilars market has experienced substantial expansion in the last few years. The market value, which was at $18.65 billion in 2024,…
Prominent Rituximab Biosimilars Market Trend for 2025: Collaborative Innovations …
Which drivers are expected to have the greatest impact on the over the rituximab biosimilars market's growth?
The rituximab biosimilars market is anticipated to grow due to the projected increase in non-Hodgkin's lymphoma (NHL) cases. NHL is a cancer that originates in the white blood cells and lymphocytes, which are integral parts of the body's immune system. For example, the American Cancer Society, a cancer advocacy group based in the US,…
Global Oncology Biosimilars Market | Global Oncology Biosimilars Industry | Onco …
The oncology biosimilars market involves of sales of medicine and drug interrelated products for cancer treatment. Biosimilars are pharmaceuticals which are produced using cell lines and are fashionable to the manufacturer. The manufacturing of such cell line processes is a multipart and time-consuming procedure.
According to the report analysis, ‘Oncology Biosimilars Market Global Report 2020-30’ states that the worldwide oncology biosimilars market was worth USD 2990.34 million in 2019. It is…
Global Biosimilars Market | Global Biosimilars Industry | Global Biosimilars Mar …
The biosimilars market involves of sales of biosimilars and associated services that are cast-off to treat chronic sicknesses such as diabetes, arthritis, and cancer. The Biosimilars are pharmaceuticals that are produced using cell lines and offers no clinical difference as linked to biologics. The Biosimilars are made once the patent of biologics is deceased.
According to the report analysis, ‘Biosimilars Market Global Report 2020-30’ states that the worldwide biosimilars market was…
Insulin Biosimilars Market, by Biosimilar Type Rapid-acting Biosimilars, Long-ac …
Diabetes is a group of metabolic disease characterized by high blood sugar level due to inadequate secretion of insulin. Common symptoms of diabetes include increased hunger, tiredness, weight loss, and excessive thirst and urination. The prevalence of diabetes is increasing, in turn, boosting demand for insulin biosimilar. For instance, according the World Health Organization (WHO) report in 2014, globally around 422 million adults were living with diabetes and 1.5 million…
