Press release
Biological Safety Testing Market Expands with Innovations in Biopharmaceuticals - Persistence Market Research
The biological safety testing market is undergoing a period of transformative growth, driven by the expanding biopharmaceutical industry and the increasing complexity of biological products. As biologics, vaccines, and gene therapies continue to dominate the pharmaceutical pipeline, regulatory agencies worldwide are enforcing more rigorous safety assessments. Biological safety testing ensures the purity, identity, and safety of these products before they reach patients.The market has witnessed a steady rise in recent years, with strong momentum projected through 2032. A robust compound annual growth rate (CAGR) is anticipated, fueled by increasing demand for biologics, stem cell therapies, and personalized medicine. Governments and regulatory bodies, including the U.S. FDA and EMA, are mandating stringent quality checks to avoid contamination risks, boosting market growth across developed and developing regions.
Among the different product types, kits and reagents account for the largest share due to their widespread application in sterility, endotoxin, and bioburden testing. These consumables are essential in repeated testing across multiple production cycles, ensuring quality assurance throughout the drug development process.
✅Get a Sample Copy of Research Report (Use Corporate Mail id for Quick Response): https://www.persistencemarketresearch.com/samples/4767
North America currently dominates the global biological safety testing market, supported by a well-established biopharmaceutical manufacturing base, strong regulatory oversight, and the presence of major industry players. In contrast, the Asia-Pacific region is emerging rapidly, driven by increased investments in biotechnology research and growing clinical trial activity.
Key Highlights from the Report
➤ North America dominates the market due to advanced infrastructure.
➤ The kits and reagents segment accounts for the highest revenue share.
➤ Rising demand for biologics and cell therapies is fueling market expansion.
➤ Strategic partnerships are shaping competitive dynamics.
➤ Innovations in gene therapy safety testing are creating new growth avenues.
How is the Biological Safety Testing Market Segmented by Product, Application, and End-User?
The biological safety testing market is segmented into product types, applications, and end-users, reflecting its diverse usage across pharmaceutical and biotech industries.
By Product, the market includes:
Kits and Reagents: These dominate the market due to their repeated usage in sterility, endotoxin, and residual DNA testing. They're indispensable in both preclinical and post-market surveillance.
Instruments: Including automated systems for pathogen detection and cell line characterization, instruments are gaining popularity for streamlining complex assays.
Others: This includes services and accessories used in tandem with test kits and devices.
By Application, the primary segments are:
Vaccines and Therapeutics: The largest application segment, particularly in light of COVID-19 and emerging infectious diseases.
Blood and Blood Products, Tissue and Tissue Products, and Cellular & Gene Therapy Products follow closely, requiring rigorous testing to meet regulatory compliance.
Stem Cell Products are gaining traction as regenerative medicine progresses.
By End-User, the major categories include:
Pharmaceutical and Biotechnology Companies: These are the largest consumers, using testing services at various stages of drug development.
Academic and Research Institutions: They rely on safety testing for early-stage studies and preclinical evaluations.
Contract Research Organizations (CROs): Offering outsourced testing services to pharma firms, CROs represent a growing end-user category.
Which Regions Are Leading the Biological Safety Testing Market and Why?
North America continues to be the epicenter of the biological safety testing market. The region's leadership stems from its high concentration of biopharma companies, a mature healthcare infrastructure, and the presence of strict regulatory frameworks laid out by the FDA and USP. Moreover, the U.S. government's support for COVID-19-related R&D and initiatives like Operation Warp Speed spurred the rapid expansion of biological testing services.
Europe is another significant market, driven by the region's strong focus on biosimilars and advanced therapy medicinal products (ATMPs). Countries like Germany, the UK, and Switzerland are hubs for biologics research and manufacturing.
In Asia-Pacific, countries like China, India, South Korea, and Japan are emerging as powerful players. These countries are rapidly scaling up biopharmaceutical production, supported by government incentives, rising healthcare expenditures, and increasing demand for cost-effective medicines. India's push to be a global vaccine manufacturing hub, and China's emphasis on domestic biotech innovation, are particularly notable.
Latin America and Middle East & Africa represent untapped opportunities. While currently limited by infrastructure and regulatory gaps, increasing foreign direct investment and a rising burden of chronic diseases are expected to drive market adoption in these regions in the coming years.
What Factors Are Driving Growth in the Biological Safety Testing Market?
Market Drivers:
The biological safety testing market is being propelled by several structural and demand-side factors. First and foremost is the explosive growth of biologics - monoclonal antibodies, cell and gene therapies, and vaccines - which require stringent safety evaluation throughout their lifecycle. Regulatory bodies have reinforced safety testing mandates to address risks like viral contamination, residual host cell proteins, and mycoplasma presence.
Rising R&D investments by both public and private stakeholders are catalyzing growth. Global healthcare organizations are focusing on biosimilar development and regenerative therapies, boosting the need for frequent and advanced testing. In parallel, technological advancements such as real-time PCR, next-gen sequencing, and high-throughput assay platforms are improving test accuracy and reducing turnaround times, making testing more accessible.
Furthermore, the global response to the COVID-19 pandemic has ushered in a new era of public health vigilance, elevating biological safety testing as a strategic priority for governments and manufacturers alike.
Market Restraints:
Despite its promising trajectory, the market faces key restraints. High costs associated with biological safety testing infrastructure - including skilled labor, advanced instrumentation, and certification - often limit adoption, particularly among small-scale manufacturers and emerging economies.
Moreover, stringent regulatory standards, while crucial for safety, can delay product launches. Complying with guidelines from multiple international bodies (e.g., FDA, EMA, PMDA) adds complexity, especially for globally marketed products. Lack of standardization in some testing protocols across regions further complicates regulatory navigation.
Another challenge is the shortage of trained professionals capable of conducting intricate biosafety assays. This talent gap affects turnaround times and may compromise data accuracy in less-developed markets.
✅Request for Customization of the Research Report: https://www.persistencemarketresearch.com/request-customization/4767
Market Opportunities:
Opportunities abound in emerging markets, where biologics manufacturing is witnessing exponential growth. Nations like Brazil, Saudi Arabia, and Malaysia are ramping up local vaccine and therapeutic production, creating long-term demand for biological safety testing services.
There's also a growing interest in personalized medicine, which necessitates frequent, batch-specific testing for quality and sterility. As patient-specific treatments such as CAR-T therapy become more mainstream, so will demand for precise, rapid biosafety assays.
Strategic collaborations and outsourcing are reshaping the competitive landscape. Biopharma firms are increasingly partnering with CROs and CDMOs to streamline their product pipelines and focus on core R&D. This shift is driving demand for third-party biological safety testing providers who can offer scalable, regulatory-compliant solutions globally.
Frequently Asked Questions about the Biological Safety Testing Market
➤ How Big is the Biological Safety Testing Market in 2024?
➤ Who are the Key Players in the Global Biological Safety Testing Market?
➤ What is the Projected Growth Rate of the Biological Safety Testing Market?
➤ What is the Market Forecast for the Biological Safety Testing Market through 2032?
➤ Which Region is estimated to dominate the Biological Safety Testing Market during the Forecast Period?
Company Insights: Leading Players in Biological Safety Testing
✦ Charles River Laboratories International Inc.
✦ Lonza Group
✦ Merck KGaA (MilliporeSigma)
✦ SGS S.A.
✦ Wuxi AppTec
✦ Eurofins Scientific
✦ Thermo Fisher Scientific
✦ Avance Biosciences Inc.
Recent Developments in the Biological Safety Testing Market
■ Charles River Laboratories launched a new rapid biosafety platform targeting gene therapy products.
■ Lonza Group partnered with a South Korean CDMO to expand biosafety testing operations in the Asia-Pacific region.
Conclusion
Biological safety testing plays a critical role in safeguarding patient health and ensuring the integrity of biopharmaceutical products. As the global medical ecosystem pivots toward complex biologics, personalized therapies, and pandemic preparedness, the demand for reliable, precise, and compliant biosafety testing has never been more urgent.
The market's future is closely tied to innovations in testing technologies, evolving regulatory landscapes, and growing investments in life sciences. Companies that can adapt to changing global standards while maintaining operational agility will stand to gain the most in this dynamic, high-stakes industry.
Contact Us:
Persistence Market Research
G04 Golden Mile House, Clayponds Lane
Brentford, London, TW8 0GU UK
USA Phone: +1 646-878-6329
UK Phone: +44 203-837-5656
Email: sales@persistencemarketresearch.com
Web: https://www.persistencemarketresearch.com
About Persistence Market Research:
At Persistence Market Research, we specialize in creating research studies that serve as strategic tools for driving business growth. Established as a proprietary firm in 2012, we have evolved into a registered company in England and Wales in 2023 under the name Persistence Research & Consultancy Services Ltd. With a solid foundation, we have completed over 3600 custom and syndicate market research projects, and delivered more than 2700 projects for other leading market research companies' clients.
Our approach combines traditional market research methods with modern tools to offer comprehensive research solutions. With a decade of experience, we pride ourselves on deriving actionable insights from data to help businesses stay ahead of the competition. Our client base spans multinational corporations, leading consulting firms, investment funds, and government departments. A significant portion of our sales comes from repeat clients, a testament to the value and trust we've built over the years.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Biological Safety Testing Market Expands with Innovations in Biopharmaceuticals - Persistence Market Research here
News-ID: 4063087 • Views: …
More Releases from Persistence Market Research

Cryogenic Storage Tanks Market Predicted to Hit US$ 12.8 Billion by 2033 Driven …
According to the latest study by Persistence Market Research, the global cryogenic storage tanks market is likely to be valued at US$ 8.6 billion in 2026 and is projected to reach US$ 12.8 billion by 2033, expanding at a CAGR of 5.8% during the forecast period 2026-2033. Rising demand for liquefied gases across energy, healthcare, food processing, and industrial manufacturing sectors is emerging as a key driver shaping the market's…
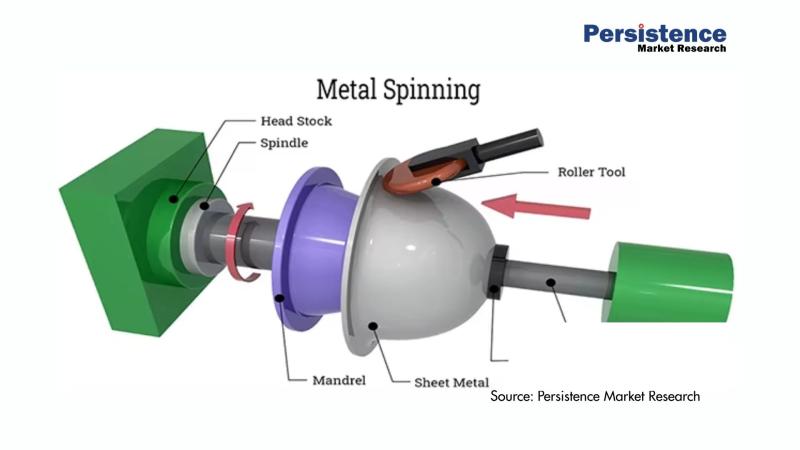
Metal Spinning Products Market Projected to Grow to US$ 4.0 billion by 2033 - Pe …
The global metal spinning products market is poised for substantial growth in the coming years. According to a recent study by Persistence Market Research, the market size is anticipated to reach US$ 4.0 billion by 2033, growing at a robust compound annual growth rate (CAGR) of 4.2% from its current valuation of US$ 3.0 billion in 2026. Metal spinning, a process of shaping metal into precise and symmetrical shapes, is…
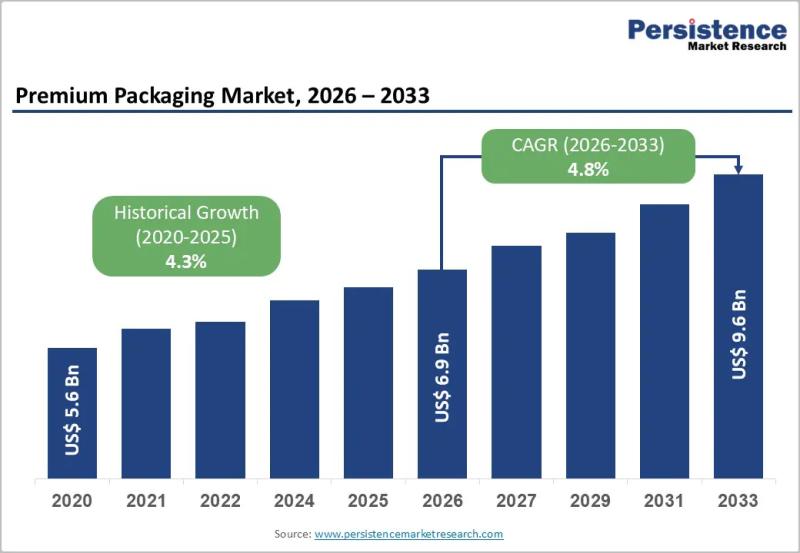
Premium Packaging Market Size Worth US$9.6 Billion by 2033 - Persistence Market …
The premium packaging market has evolved into a critical strategic element for brand differentiation across multiple high value consumer industries. Premium packaging goes beyond basic containment and protection to deliver enhanced aesthetics tactile appeal storytelling and emotional connection. Brands increasingly view packaging as an extension of their identity and a powerful marketing tool that influences purchasing decisions at the point of sale and during the unboxing experience. This shift is…
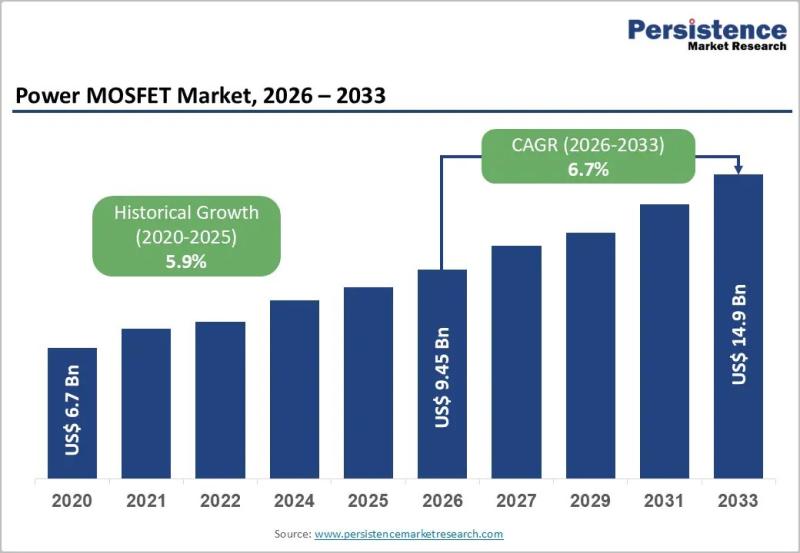
Power MOSFET Market Growth Driven by EVs Renewable Energy and Smart Automation
The global Power MOSFET market is entering a phase of sustained expansion, driven by the accelerating need for energy-efficient and high-performance power management components across industries. In 2026, the market is expected to be valued at US$ 9.45 billion and is forecast to reach US$ 14.9 billion by 2033, registering a healthy CAGR of 6.7% during the forecast period. Power MOSFETs are essential semiconductor devices that enable efficient switching and…
More Releases for Biological
Biological Control Market Projected to Witness Massive Growth - Koppert Biologic …
Biological Control Market Snapshot
The Biological Control Market is projected to reach USD 9.7 billion by 2032, growing at a CAGR of 9.4% from 2025 to 2032.
Coherent Market Insights proudly presents its latest Biological Control Market Research Report, delivering a detailed examination of the U.S. Biological Control Market outlook from 2025 to 2032. This comprehensive analysis provides strategic forecasts at both national and regional levels, addressing evolving supply networks, payer-provider collaboration,…
Biological and Chemical Indicators Market - Beyond Measure: Biological and Chemi …
Newark, New Castle, USA: The "Biological and Chemical Indicators Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors.
Biological and Chemical Indicators Market: https://www.growthplusreports.com/report/biological-and-chemical-indicators-market/8887
This latest report researches the…
Biological APIs Market - Biological APIs: The Driving Force Behind Cutting-Edge …
Newark, New Castle, USA: The "Biological APIs Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors.
Biological APIs Market: https://www.growthplusreports.com/report/biological-apis-market/8769
This latest report researches the industry structure, sales, revenue,…
Human Immunoglobulin for Intravenous Injection Market demand and future scope wi …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global Human Immunoglobulin for Intravenous Injection. On the basis of historic growth analysis and current scenario of Human Immunoglobulin for Intravenous Injection place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from…
Global Biological safety testing Market | Global Biological safety testing Indus …
Biologics are termed as those product that attained from living organism and feature a differentiate set of properties. The biologics want extreme safety from contamination thus biological safety testing is incorporate timely to make sure its safety quality and non-contamination of vaccines & biopharmaceuticals, along with comply with the bound rules applicable on it. A number of the foremost biological tests are sterility testing, bioburden testing, residual host contaminant detection…
Meningococcal Infections Vaccine Market Insights, Forecast to 2025 : Sanofi-Past …
Researchmoz added Most up-to-date research on "Global Meningococcal Infections Vaccine Market Insights, Forecast to 2025" to its huge collection of research reports.
This report studies the global market size of Meningococcal Infections Vaccine in key regions like North America, Europe, Asia Pacific, Central & South America and Middle East & Africa, focuses on the consumption of Meningococcal Infections Vaccine in these regions.
This research report categorizes the global Meningococcal Infections Vaccine market…
