Press release
Aquigen Bio Launches High-Purity Gefitinib EP Impurity A: Elevating Standards in Pharmaceutical Research
Pune, India - May 2025 - Aquigen Bio, a global leader in pharmaceutical reference materials and impurity profiling, proudly announces the release of its latest product: Gefitinib EP Impurity A. This compound is an essential analytical standard used in the development, validation, and quality assurance of Gefitinib, a widely prescribed EGFR inhibitor for treating non-small cell lung cancer (NSCLC).As the pharmaceutical industry continues to raise the bar on quality and compliance, reliable access to highly pure and well-characterized impurities like Gefitinib EP Impurity A is critical. Aquigen Bio's latest offering is designed to support pharmaceutical manufacturers, CROs, and research institutions involved in drug development and regulatory submissions.
Understanding Gefitinib EP Impurity A
https://aquigenbio.com/product/gefitinib-ep-impurity-a/
Gefitinib EP Impurity A is one of the specified impurities listed in the European Pharmacopoeia (EP) monograph for Gefitinib. It plays a key role in impurity profiling and stability studies during the production of the active pharmaceutical ingredient (API). By monitoring and quantifying this impurity, researchers and manufacturers can ensure the safety, efficacy, and regulatory compliance of the final pharmaceutical product.
Product Highlights:
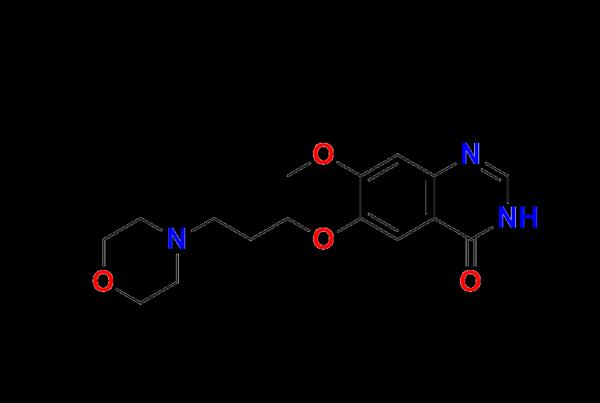
Chemical Name: 7-methoxy-6-[3-(morpholin-4-yl)propoxy]quinazolin-4(3H)-one
CAS Number: [CAS registered]
Purity: ≥98%
Analytical Data Included: HPLC, NMR, MS, COA
Packaging Sizes: Available in 10 mg, 25 mg, 50 mg, and 100 mg packs
Storage: Store in a cool, dry place; protect from light
The availability of this impurity in high purity enables precise and reproducible analytical testing, which is vital for maintaining the integrity of generic and branded Gefitinib formulations.
Why Impurity Profiling Matters in Pharmaceutical Development
Impurities can arise during synthesis, storage, or degradation of a pharmaceutical compound. Regulatory authorities such as the EMA, FDA, and ICH require comprehensive impurity profiling to ensure drugs are safe for human consumption.
Gefitinib EP Impurity A must be detected, quantified, and controlled within permissible limits throughout the drug's lifecycle. This ensures:
Patient safety by reducing toxicological risks
Therapeutic efficacy by maintaining the purity of the API
Regulatory compliance in accordance with pharmacopoeial standards
Aquigen Bio helps pharmaceutical scientists meet these goals by providing ready-to-use, validated impurity standards.
Applications of Gefitinib EP Impurity A
Aquigen Bio's Gefitinib EP Impurity A is suited for a range of laboratory applications, including:
Analytical Method Development
Supporting HPLC, UPLC, and LC-MS method validation
Defining specificity, accuracy, and robustness for impurity detection
Stability Testing
Assists in determining the shelf life and degradation profile of Gefitinib products
Regulatory Submissions
Includes complete documentation to facilitate DMF (Drug Master File) and ANDA (Abbreviated New Drug Application) filings
Quality Control (QC)
Utilized in batch release testing and specification setting for manufacturing
With global GMP and ICH compliance, Aquigen's impurity standards streamline workflows in R&D, QC, and regulatory departments.
Complementary Products in the Gefitinib Series
To offer a comprehensive impurity profiling suite, Aquigen Bio also provides several other impurities and related compounds in the Gefitinib family. These products are useful for thorough characterization and bioanalytical studies:
1. Gefitinib EP Impurity B
https://aquigenbio.com/product/gefitinib-ep-impurity-b/
Gefitinib EP Impurity B is another specified impurity in the EP monograph, relevant for stability and degradation pathway analysis. Often occurring during synthetic processing or under stress conditions, this impurity is crucial for assessing the drug's robustness and storage requirements.
2. Gefitinib d8
https://aquigenbio.com/product/gefitinib-d8/
This deuterated version of Gefitinib is widely used as an internal standard in LC-MS/MS assays. Its stable isotopic labeling allows for accurate quantification of Gefitinib in biological matrices, making it valuable in pharmacokinetic (PK) and bioequivalence (BE) studies.
3. 3-Desmorpholinyl-3-Hydroxyethylamino Gefitinib
https://aquigenbio.com/product/3-desmorpholinyl-3-hydroxyethylamino-gefitinib/
This compound is a key metabolite or degradation impurity of Gefitinib. It provides insight into the metabolic fate of the drug, helping researchers assess potential toxicity, efficacy, and formulation stability.
By combining these related impurity standards, researchers can perform comprehensive impurity mapping and method validation with confidence.
Why Choose Aquigen Bio?
Aquigen Bio has established itself as a trusted source of pharmaceutical reference materials by upholding the highest standards of quality, reliability, and scientific integrity. Here's what sets the company apart:
Strict Quality Control
All impurities undergo rigorous quality control including:
Nuclear Magnetic Resonance (NMR)
Mass Spectrometry (MS)
High-Performance Liquid Chromatography (HPLC)
Elemental analysis and stability testing
Global Regulatory Support
Each product is supplied with a Certificate of Analysis (COA) and full analytical data, ensuring smooth regulatory submissions for ANDA, NDA, or MAH applications.
Customization and Synthesis
Need a custom impurity or isotope-labeled compound? Aquigen's expert team offers custom synthesis services tailored to meet the specific demands of your project.
Rapid Global Shipping
With a streamlined supply chain and global logistics support, Aquigen Bio delivers to over 60 countries, ensuring timely availability for urgent R&D needs.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio
Aquigen Bio is a trusted manufacturer and global supplier of pharmaceutical impurities, isotope-labeled compounds, metabolites, and reference standards. Backed by a team of experienced scientists and a commitment to quality, Aquigen supports pharmaceutical companies, CROs, and academic institutions in over 60 countries. With a customer-first approach and dedication to scientific excellence, Aquigen Bio continues to drive innovation in analytical chemistry and regulatory science.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Aquigen Bio Launches High-Purity Gefitinib EP Impurity A: Elevating Standards in Pharmaceutical Research here
News-ID: 4020661 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Gefitinib
AquigenBio Advances Oncology Research with High-Purity Gefitinib and Certified I …
Pune, India - May 9, 2025 - AquigenBio, a leading provider of high-quality impurity standards, is proud to announce the expansion of its product portfolio with the addition of certified impurity standards for Gefitinib. These standards are essential for the pharmaceutical industry, particularly for developers of generic formulations, to ensure the safety, efficacy, and regulatory compliance of their products.
Introduction to Gefitinib and Its Importance in Oncology
https://aquigenbio.com/products/impurity-standards/gefitinib/
Gefitinib, marketed under the brand…
Pharmaceutical Grade Gefitinib Market Insights, Forecast to 2030
A complete study of the global Pharmaceutical Grade Gefitinib market is carried out by the analysts in this report, taking into consideration key factors like drivers, challenges, recent trends, opportunities, advancements, and competitive landscape. This report offers a clear understanding of the present as well as future scenarios of the global Pharmaceutical Grade Gefitinib industry. Research techniques like PESTLE and Porter's Five Forces analysis have been deployed by the researchers.…
Pharmaceutical Grade Gefitinib Market Size, Trends, Application, Growth Forecast …
LOS ANGELES, United States: The global Pharmaceutical Grade Gefitinib market is carefully researched in the report while largely concentrating on top players and their business tactics, geographical expansion, market segments, competitive landscape, manufacturing, and pricing and cost structures. Each section of the research study is specially prepared to explore key aspects of the global Pharmaceutical Grade Gefitinib market. For instance, the market dynamics section digs deep into the drivers, restraints,…
Latest Report Of Gefitinib Market Growth, Opportunities, Risk, Challenges, Gross …
[Latest Updated Report 2023] The Gefitinib Market 2023 Report makes available the current and forthcoming technical and financial details of the industry. It is one of the most comprehensive and important additions to the Prudent Markets archive of market research studies. It offers detailed research and analysis of key aspects of the global Gefitinib market. This report explores all the key factors affecting the growth of the global Gefitinib market,…
Global Gefitinib API Market to Witness a Pronounce Growth During 2025
LP INFORMATION recently released a research report on the Gefitinib API market analysis and elaborate the industry coverage, current market competitive status, and market outlook and forecast by 2025. Moreover, it categorizes the global Gefitinib APImarket by key players, product type, applications and regions,etc.
The main objective of this market research is to help the readers understand the structure of Gefitinib APImarket, market definition, overview, industry opportunities and trends, investment…
Gefitinib Market Size, Share, Development by 2024– Top Key players like AstraZ …
Market Research Report Store offers a latest published report on Gefitinib Market Analysis and Forecast 2019-2025 delivering key insights and providing a competitive advantage to clients through a detailed report. The report contains 91 pages which highly exhibit on current market analysis scenario, upcoming as well as future opportunities, revenue growth, pricing and profitability.
Click to view the full report TOC, figure and tables:
https://www.marketresearchreportstore.com/reports/858328/global-gefitinib-market-insights
The global Gefitinib market is valued at xx…