Press release
Emerging Trends Influencing The Growth Of The GMP Cell Therapy Consumables Market: Advancements in GMP-Compliant CD34+ Hematopoietic Stem Cells: OrganaBio's Launch and Industry Developments
The GMP Cell Therapy Consumables Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].How Big Is the GMP Cell Therapy Consumables Market Size Expected to Be by 2034?
The market size for consumables used in GMP cell therapies has seen rapid expansion in the past few years. Predictions have it growing from $17.85 billion in 2024 to a staggering $22.87 billion in 2025, with a compound annual growth rate (CAGR) of 28.1%. This historic growth trend is due, in large part, to increased investments in regenerative medicine, improved cell therapy research, an uptick in clinical trials and approvals, a rising incidence of chronic diseases, and increasing consciousness about personalized medicine.
In the coming years, an explosive growth is expected in the GMP cell therapy consumables market, with a market value projected to hit $59 billion by 2029. This represents a compound annual growth rate (CAGR) of 26.7%. The projected expansion over the forecast period is primarily driven by factors such as broadened applications for cell therapy, an escalation in global cell therapy trials, regulatory backing for cell therapies, the advent of cutting-edge cell therapy platforms, and the global growth of cell therapy manufacture. Key trends expected during the forecast period comprise investment in manufacturing infrastructure, technological advancements in cell processing, collaborative efforts and partnerships, digitization and data amalgamation, and strategic alliances and mergers.
Purchase the full report for exclusive industry analysis:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=12014
What Are the Emerging Segments Within the GMP Cell Therapy Consumables Market?
The GMP cell therapy consumables market covered in this report is segmented -
1) By Product: Kits, Reagents Or Molecular Biology reagents, Growth Factors Or Cytokines And Interleukins, Other Products
2) By Cell Therapy: NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy, Other cell therapies
3) By Process: Cell Collection And Characterization Or Sorting And Separation, Cell Culture And Expansion Or Preparation, Cryopreservation, Cell Processing And Formulation, Cell Isolation And Activation, Cell Distribution Or Handling, Process Monitoring And Control Or Readministration Or Quality Assurance, Other Processes
4) By End-Use: Clinical, Commercial, Research
Subsegments:
1) By Kits: Cell Culture Kits, Cell Isolation Kits, Transfection Kits
2) By Reagents Or Molecular Biology Reagents: Buffers And Solutions, Enzymes, Staining Reagents
3) By Growth Factors Or Cytokines And Interleukins: Hematopoietic Growth Factors, Antibody Cytokines, Immunomodulatory Cytokines
3) By Other Products: Cell Culture Vessels, Bioreactors, Media And Supplements
Get your free sample here:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12014&type=smp
What Long-Term Drivers Are Shaping GMP Cell Therapy Consumables Market Trends?
The GMP cell therapy consumables market is anticipated to grow due to the escalating progress in drug discovery. Drug discovery involves the finding of potential new medical entities through the integration of computational, experimental, translational, and clinical models. Developments in fields like molecular biology, genetics, bioinformatics, and high-throughput screening have caused a paradigm shift in drug discovery. These advancements allow scientists to gain a deeper understanding of disease mechanisms, pinpoint potential drug targets, and conduct more efficient screenings for drug candidates across extensive compound libraries. This increase in drug discovery processes is influencing the expansion of the GMP cell therapy consumables market. For instance, in January 2023, the Food and Drug Administration, a U.S. federal department of Health and human services, reported that its Centre for Drug Evaluation and Research (CDER) approved 37 new drugs and therapeutic biological products for various indications in 2022 in the U.S. Consequently, this surge in drug discovery is fuelling the growth of the GMP cell therapy consumables market.
Who Are the Top Competitors in Key GMP Cell Therapy Consumables Market Segments?
Major companies operating in the GMP cell therapy consumables market include Thermo Fisher Scientific Inc., Fresenius Kabi AG, Danaher Corporation, Merck KGaA, Asahi Kasei Corporation, GE HealthCare Technologies Inc., Corning Incorporated, Avantor Inc., Lonza Group AG, Terumo Corporation, Catalent Inc., Sartorius AG, Bio-Techne Corp, Repligen Corporation, Miltenyi Biotec BV & Co KG, Genscript Biotech Corporation, Rentschler Biopharma SE, FUJIFILM Irvine Scientific Inc., BioLegend Inc., STEMCELL Technologies Inc., BioLife Solutions Inc., Abzena Ltd., Sino Biological Inc., ProBioGen AG, MaxCyte Inc., PromoCell GmbH, PeproTech Inc., Cellares Corp., Wilson Wolf Corporation, Cellexus Ltd.
What GMP Cell Therapy Consumables Market Trends Are Gaining Traction Across Different Segments?
Significant businesses in the GMP cell therapy consumables market are working on GMP-compliant CD34+ hematopoietic stem cells (HSCs) for the advancement of therapies. These CD34+ HSCs, produced under Good Manufacturing Practice (GMP), are a certain type of stem cell distinguished by the CD34 marker on their surface. To illustrate, in April 2024, the American pharmaceutical firm, OrganaBio, introduced HematoPAC-HSC-CB-GMP, an easily accessible source of GMP-compliant CD34+ HSCs originating from fresh human cord blood. Capitalizing on their comprehensive proficiency in cell segregation and GMP production, OrganaBio can generate high volumes of extremely viable CD34+ HSCs within a day from collection. The purpose of this initiative is to expedite the development of cutting-edge remedies, inclusive of those for blood cancers and genetic anomalies.
Get the full report for exclusive industry analysis:
https://www.thebusinessresearchcompany.com/report/gmp-cell-therapy-consumables-global-market-report
Which Regions Are Becoming Hubs for GMP Cell Therapy Consumables Market Innovation?
North America was the largest region in the GMP cell therapy consumables market in 2024. Asia-pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the GMP cell therapy consumables market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Frequently Asked Questions:
1. What Is the Market Size and Growth Rate of the GMP Cell Therapy Consumables Market?
2. What is the CAGR expected in the GMP Cell Therapy Consumables Market?
3. What Are the Key Innovations Transforming the GMP Cell Therapy Consumables Industry?
4. Which Region Is Leading the GMP Cell Therapy Consumables Market?
Why This Report Matters:
Competitive overview: This report analyzes the competitive landscape of the 3D imaging software market, evaluating key players on market share, revenue, and growth factors.
Informed Decisions: Understand key strategies related to products, segmentation, and industry trends.
Efficient Research: Quickly identify market growth, leading players, and major segments.
Connect with us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ.
Contact Us
Europe: +44 207 1930 708,
Asia: +91 88972 63534,
Americas: +1 315 623 0293 or
Email: mailto:info@tbrc.info
Learn More About The Business Research Company
With over 15,000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Our flagship product, the Global Market Model delivers comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Emerging Trends Influencing The Growth Of The GMP Cell Therapy Consumables Market: Advancements in GMP-Compliant CD34+ Hematopoietic Stem Cells: OrganaBio's Launch and Industry Developments here
News-ID: 3997113 • Views: …
More Releases from The Business Research Company
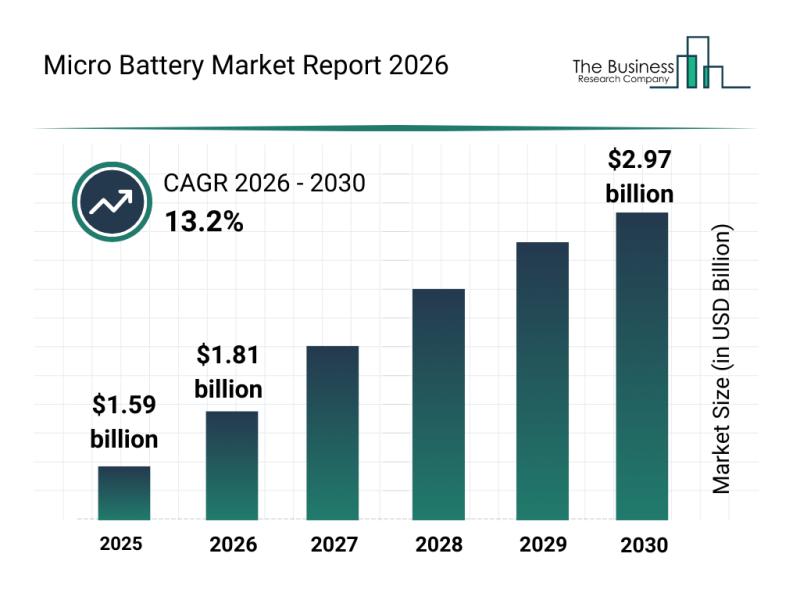
Competitive Landscape: Leading Companies and New Entrants in the Micro Battery M …
The micro battery market is on the verge of significant expansion as emerging technologies and growing applications drive demand. This sector is rapidly evolving with innovations that promise to deliver more efficient, compact, and versatile power solutions for a wide range of devices. Let's explore the current market size, key players, emerging trends, and segmentation details shaping the future of micro batteries.
Projected Market Value and Growth Trajectory of the Micro…
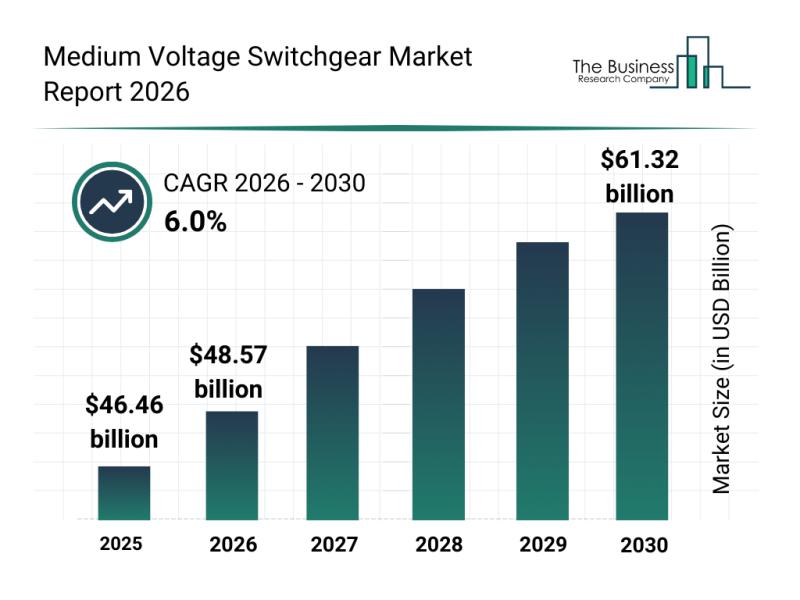
Growth Patterns, Market Segmentation, and Competitive Approaches Influencing the …
The medium voltage switchgear industry is on track for substantial growth as advancements in technology and infrastructure continue to drive demand. With the increasing need for efficient power distribution and smart management solutions, this sector is set to experience significant expansion through 2030. Let's examine the market's projected value, leading companies, important trends, and the key segments shaping this competitive landscape.
Projected Market Value and Growth Factors in the Medium Voltage…
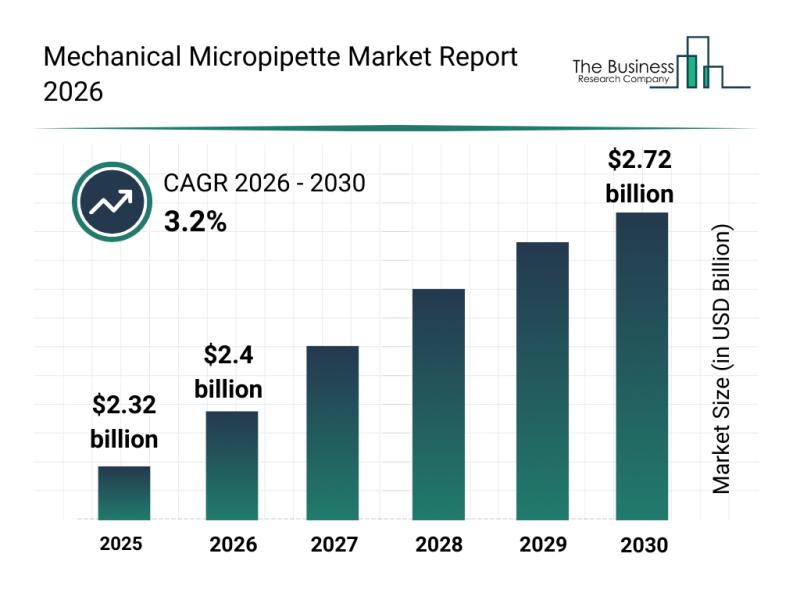
Leading Companies Reinforce Their Presence in the Mechanical Micropipette Market
The mechanical micropipette market is progressing steadily as new technologies and expanding applications continue to push demand. Driven by advancements in laboratory automation and increasing needs in biological research fields, this sector is set for notable growth over the coming years. Let's explore the expected market value, leading companies, emerging trends, and key market segments shaping this industry.
Projected Market Value and Growth Outlook for Mechanical Micropipettes
The mechanical micropipette market…
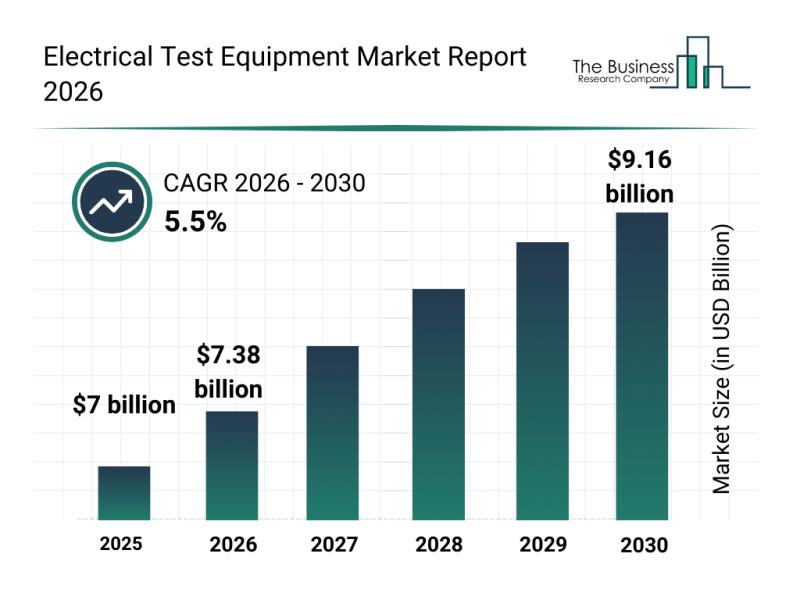
Top Players and Market Competition in the Electrical Test Equipment Industry
The electrical test equipment market is poised for significant expansion in the coming years, driven by rapid technological advancements and increasing demands across various industries. This growth reflects broader trends in electric vehicle infrastructure, renewable energy, and digital diagnostics, positioning the market for robust development through 2030.
Projected Market Size and Growth Trajectory of the Electrical Test Equipment Market
By 2030, the electrical test equipment market is expected to reach a…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…
