Press release
Linolenic Impurity Standards: Advancing Analytical Techniques for Detecting Trace-Level Impurities in Pharmaceutical Ingredients
Linolenic Impurity Standards are at the forefront of modern pharmaceutical research, enabling precise identification and quantification of trace-level impurities that can impact the safety, efficacy, and stability of drug products. As regulatory agencies impose increasingly stringent expectations for impurity characterization, pharmaceutical companies are turning to robust analytical solutions like Linolenic Impurity Standards to ensure compliance and deliver high-quality products.To know more about Linolenic Impurity Standards : https://aquigenbio.com/products/impurity-standards/linolenic/
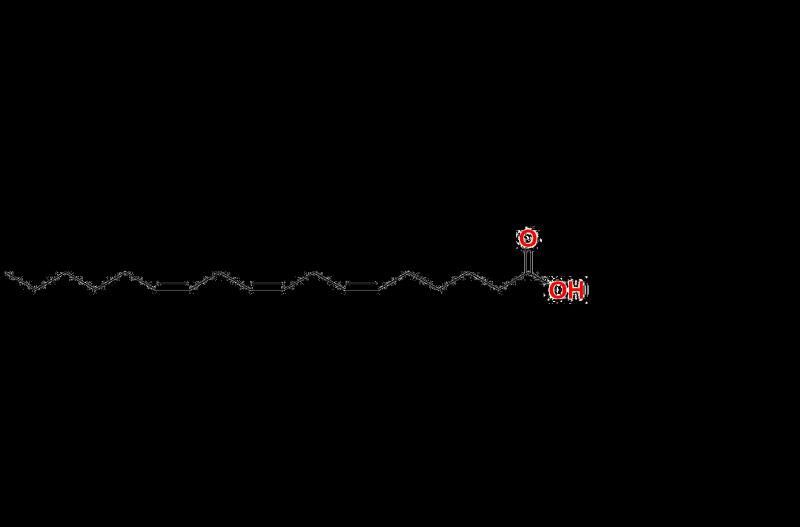
The ability to detect impurities such as Linolenic Acid and its derivatives is vital for the development and manufacture of Active Pharmaceutical Ingredients (APIs). Trace impurities, including those related to Gamma-Linolenic Acid, can arise from raw materials, degradation processes, or reactions during production. Identifying and controlling these impurities has become an indispensable part of modern pharmaceutical manufacturing in order to mitigate potential risks to patients.
Moreover, advanced methodologies are designed to track impurities even at extremely low concentrations. These analytical developments empower organizations to maintain quality, minimize toxicological risks, and stay ahead in delivering reliable therapeutic solutions to patients worldwide.
The Growing Importance of Linolenic Impurity Standards in Drug Development
With the advent of Linolenic Impurity Standards, researchers now have access to highly accurate reference materials to validate their testing methods. These standards allow testing labs to replicate impurity profiles, making it easier to analyze data and implement corrective actions when needed. Pharmaceutical players are increasingly adopting these standards to streamline manufacturing processes and adhere to Good Manufacturing Practices (GMP), ensuring the delivery of products that meet global regulatory requirements.
To know more about products :
Gamma-Linolenic Acid - https://aquigenbio.com/product/gamma-linolenic-acid/
Linolenic Acid - https://aquigenbio.com/product/linolenic-acid/
Methyl Gamma-linolenate - https://aquigenbio.com/product/methyl-gamma-linolenate/
The significance of Linolenic-based analysis extends beyond compliance. Effective impurity control has proven essential for ensuring the repeatability and scalability of production methods. Whether in the early stages of drug discovery or during the large-scale production of APIs, a robust focus on Linolenic Impurity Standards helps companies achieve superior product consistency.
Tackling Analytical Challenges with Linolenic Impurity Standards
The complexity of testing impurities related to compounds like Gamma-Linolenic Acid or its derivatives underscores the importance of precision. For instance, advanced techniques such as LC-MS, GC-MS, and NMR spectroscopy have been used successfully to detect Methyl Gamma-linolenate, which can act as a marker for reaction pathways or stability issues within formulations. These techniques are pushing analytical capabilities to new heights, giving scientists the tools to detect impurities that were previously beyond the limits of standard equipment.
The CEO of Aquigen Bio Sciences, highlights the challenges faced in detecting and controlling impurities: "Ensuring the complete removal or monitoring of trace Linolenic impurities is one of the hurdles in pharmaceutical manufacturing. The detection of these minuscule compounds demands highly specialized tools and methodologies, emphasizing the critical role of impurity standards in shaping the future of analytical chemistry. Advances in this field provide pharmaceutical manufacturers the clarity they need to achieve compliance and optimize patient safety."
Moving forward, the role of Linolenic Impurity Standards will continue to expand as the pharmaceutical sector adopts more rigorous standards for quality control. Custom-designed impurity standards will be pivotal in assisting research institutions and drug manufacturers in meeting the evolving needs of regulatory frameworks, while also enabling them to efficiently address patient safety concerns during drug development.
Conclusion
Linolenic Impurity Standards have emerged as a cornerstone of pharmaceutical analytical science, driving advances in impurity detection, risk management, and regulatory compliance. By providing highly reliable tools for monitoring trace impurities, these standards bridge the gap between innovation and patient safety, enabling organizations to meet the growing complexities of modern drug development.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
Aquigen Bio Sciences is a trusted leader in providing customized solutions for pharmaceutical research and development. As the leading resource for Linolenic Impurity Standards in India, Aquigen supports the pharmaceutical sector with advanced impurity synthesis, reference materials, and analytical validation services. With expertise spanning impurities like Linolenic Acid and its derivatives, Aquigen empowers organizations to achieve excellence in impurity characterization, ensuring that every pharmaceutical product adheres to the highest standards of quality and safety.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Linolenic Impurity Standards: Advancing Analytical Techniques for Detecting Trace-Level Impurities in Pharmaceutical Ingredients here
News-ID: 3946976 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Linolenic
Gamma Linolenic Acid Market Examine of Key Developments, Geodemographic Study by …
New York, Global Gamma Linolenic Acid Market report from Global Insight Services is the single authoritative source of intelligence on Gamma Linolenic Acid Market . The report will provide you with analysis of impact of latest market disruptions such as Russia-Ukraine war and Covid-19 on the market. Report provides qualitative analysis of the market using various frameworks such as Porters' and PESTLE analysis. Report includes in-depth segmentation and market size…
Linolenic Acid Market Analysis 2019 By Top Key Players- Cayman, Eastman, Acme, P …
Linolenic acid is a type of fatty acid. It can refer to either of two octadecatrienoic acids (i.e. with an 18-carbon chain and three double bonds, which are found in the cis configuration), or a mixture of the two.
Linoleic acid is a fatty acid that is essential for healthy skin and hair, as well as normal brain function and can’t be produced in the body naturally and thus has to…
Global Alpha Linolenic Acid(ALA) Market 2019 - Cayman, Eastman, Charkit Chemical …
Alpha linolenic acid is a kind of plant ligan most notably found in flaxseed.Alpha linolenic acid is classified as a phytoestrogen since it is a plant-derived, nonsteroid compound that possesses estrogen-like activity.
Scope of the Report:
Alpha linolenic acid(ALA)is a polyunsaturated fatty acid, which is essential to human health but cannot be manufactured by human body.
The worldwide market for Alpha Linolenic Acid(ALA) is expected to grow at a CAGR of roughly xx%…
Global Gamma Linolenic Acid(GLA) Market 2019 - Merck and Millipore, Charkit Chem …
Gamma Linolenic Acid(GLA) extract from evening primrose.Evening primrose is a North American native biennial plant.
Scope of the Report:
Gamma linolenic acid can reduce the blood lipid, cholesterin and triglyceride, it can also prevent for apoplexy, hyperension, blood clots, arteriosclerosis and arrhythmia.
The worldwide market for Gamma Linolenic Acid(GLA) is expected to grow at a CAGR of roughly xx% over the next five years, will reach xx million US$ in 2023,…
Linolenic Acid Market Industry 2018-2025 Growth and Business demand| Key Vendors …
Global Linolenic Acid Market report is designed in a method that helps readers to acquire a complete knowledge about the overall market scenario for the projected period. This Market report consists of the brief profile of key players in the industry and their upcoming market plans and current developments during the forecasted period 2019 to 2025.
Linolenic Acid Market Report provides a basic overview of the industry including definitions,…
Global Linolenic Acid Market Trend Survey and Prospects Report 2018-2023
2018-2023 Linolenic Acid Market research report presents a comprehensive study of the Linolenic Acid Market in Global Industry; the study scans the progression of the market trailed over the past few years and the forecasts of growth in the coming years, primarily in terms of sales. The research report highlights the reasons liable for the disparities in the market and investigates them methodically. The Linolenic Acid Market effect factors have…