Press release
Abacavir Impurity Standards: Enhancing Drug Development, Research, and Quality Control
Abacavir plays a critical role in combating HIV-1 infections as a fundamental component of antiretroviral therapy. Its effectiveness and widespread usage make it a cornerstone of HIV treatment. However, ensuring the safety, efficacy, and purity of Abacavir formulations is paramount. Pharmaceutical companies and research organizations increasingly emphasize the need for sophisticated impurity standards to meet stringent regulatory and therapeutic requirements.The Importance of Abacavir Impurity Profiling
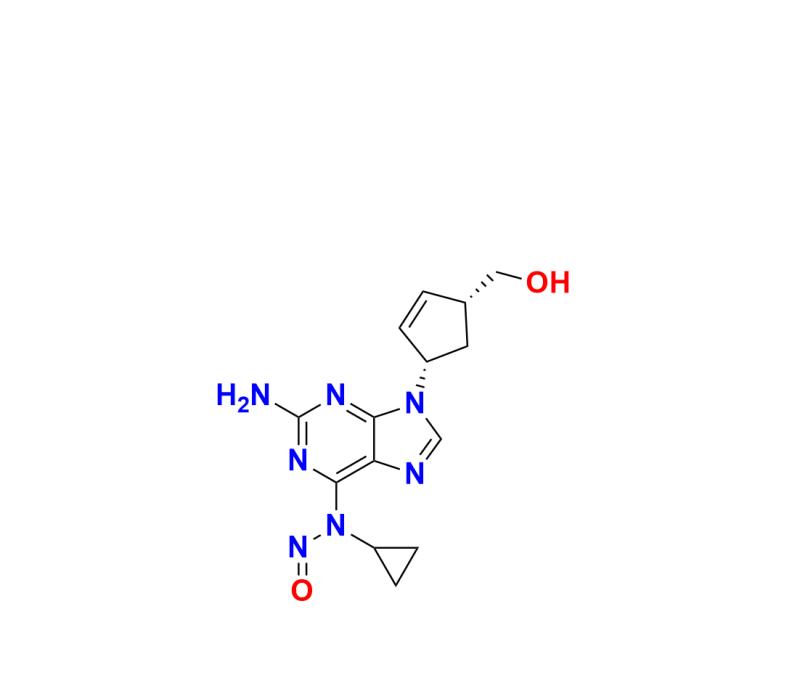
The detection, quantification, and control of impurities in active pharmaceutical ingredients (APIs) like Abacavir are essential for safeguarding patient health and upholding global regulatory compliance. One highly notable impurity, N-Nitroso Abacavir EP Impurity A2, underscores the industry's focus on ensuring that advanced impurity standards are readily available for accurate analysis. Identifying impurities like this prevents the risk of potential toxicological concerns by enabling manufacturers to precisely monitor contaminants during drug production processes.
To know more about Abacavir : https://aquigenbio.com/products/impurity-standards/abacavir-impurity-standards/
Moreover, the adoption of advanced impurity reference materials allows for seamless impurity profiling, a crucial component during formulation, stability studies, and regulatory submissions. For instance, impurities such as N-Nitroso Abacavir EP Impurity A must be carefully analyzed and controlled to meet strict thresholds set by international health authorities. Pharmaceutical companies rely heavily on high-quality reference standards to generate accurate impurity profiles and ensure that their drug substances and formulations do not pose a risk to patient safety.
Ensuring Safety and Compliance in Drug Development
The presence of impurities, even in minute quantities, can compromise drug safety if left unchecked. Accurate testing with premium impurity standards is pivotal for accelerating the approval of Abacavir-based products. These standards allow manufacturers to not only meet regulatory benchmarks but also establish their products as safe, consistent, and effective. Additionally, researchers can leverage these tools to investigate degradation pathways, optimize formulations, and enhance overall drug stability profiles-further building on the scientific foundation for improving antiretroviral treatments.
Role of Impurity Standards in Pharmaceutical Innovation
The importance of using authenticated impurity standards extends beyond regulatory compliance. These materials play a major role in fostering pharmaceutical advancements by assisting in the discovery of new formulations and dosage strategies. The use of high-purity impurity reference materials for Abacavir facilitates deeper research insights while giving manufacturers the confidence to scale their operations. This ensures a robust supply chain of safe and effective Abacavir-based therapies-strengthening global HIV treatment infrastructure.
To Know more about the products :
1.N-Nitroso Abacavir EP Impurity A - https://aquigenbio.com/product/n-nitroso-abacavir-ep-impurity-a/
2.N-Nitroso Abacavir EP Impurity A 2 - https://aquigenbio.com/product/n-nitroso-abacavir-ep-impurity-a-2/
"The pharmaceutical landscape demands precision and adherence to global safety standards, especially for critical APIs such as Abacavir," said Aquigen Bio Sciences CEO. "We are committed to providing industry-leading impurity standards like N-Nitroso Abacavir EP Impurity A2 to empower manufacturers and researchers. By ensuring unmatched quality and reliability, we support our clients in expediting their drug development processes, achieving compliance, and delivering safer medications to patients worldwide."
As technology in pharmaceutical analysis evolves, impurity standards have become indispensable tools for both researchers and the industry. Whether for quantifying impurities like N-Nitroso Abacavir EP Impurity A, addressing regulatory requirements, or enhancing product stability, impurity standards are at the core of modern pharmaceutical development. Their application directly supports innovation while ensuring quality, safety, and sustainability in drug manufacturing.
Conclusion
The availability of high-purity Abacavir impurity standards has transformed pharmaceutical drug development by enabling more precise and reliable impurity analysis. From facilitating regulatory compliance to enhancing the safety and efficacy of life-saving therapies, impurity standards are critical to the success of the global pharmaceutical industry. High-quality impurity reference materials empower organizations to overcome challenges in drug formulation, testing, and compliance, ensuring patients receive safe, effective medications for HIV treatment.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences
Aquigen Bio Sciences stands as a trusted leader in India for Abacavir impurity standards. The company provides a comprehensive range of high-purity reference materials, including those for N-Nitroso Abacavir EP Impurity A2 and N-Nitroso Abacavir EP Impurity A, to meet the evolving needs of pharmaceutical manufacturers and researchers. With a commitment to quality, consistency, and reliability, Aquigen Bio Sciences plays a pivotal role in advancing drug development and quality assurance processes. Their expertise and focus on delivering superior impurity standards make them the partner of choice for drug manufacturers aiming to meet international regulatory standards and enhance the safety and performance of their products.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Abacavir Impurity Standards: Enhancing Drug Development, Research, and Quality Control here
News-ID: 3928369 • Views: …
More Releases from Aquigen Bio Sciences

Elevate Pharmaceutical R&D with Aquigen BioSciences' Precision‐Grade Flibanser …
Flibanserin Impurity B is a reference standard used in pharmaceutical research and development. It is primarily applied during the analysis and validation of drug substances to identify, quantify, and control impurities that may be present in the final product. This impurity is associated with the parent compound, Flibanserin, a medication approved for the treatment of hypoactive sexual desire disorder (HSDD) in premenopausal women.
Aquigen BioSciences offers Flibanserin Impurity B as a…

Estradiol Valerate EP Impurity A - Premium Reference Standard for Analytical Dev …
Estradiol Valerate EP Impurity A is a high-quality reference standard designed to meet the stringent requirements of pharmaceutical research, method validation, and quality control processes.
Explore Estradiol Valerate EP Impurity A :
https://aquigenbio.com/product/estradiol-valerate-ep-impurity-a/
Manufactured and characterized with precision, this impurity standard supports laboratories and manufacturers in achieving consistent, reliable, and reproducible results in critical analytical workflows.
With its exceptional purity and accurate characterization, Estradiol Valerate EP Impurity A plays a vital role…

High-Purity N-Nitroso Betahistine D3 for Precise Pharmaceutical Analysis | Deute …
Product Overview
N-Nitroso Betahistine D3 is a premium deuterated nitrosamine impurity standard, specifically developed for precise analytical testing in pharmaceutical laboratories. This reference standard is widely used for analytical method development, validation, and quality control processes to meet stringent regulatory guidelines. With exceptional purity, complete documentation, and reliable traceability, it is ideal for research, development, and compliance applications.
https://aquigenbio.com/product/n-nitroso-betahistine-d3/
Key Features and Benefits
Deuterated Design for Precision: The incorporation of deuterium improves mass spectrometric…

Aquigen Bio Strengthens Pharmaceutical Research with High-Purity Icatibant Impur …
Aquigen Bio, a trusted supplier of pharmaceutical reference standards, today announced the expansion of its Icatibant Impurity Standards portfolio, designed to support drug developers, analytical laboratories, and research organizations with reliable materials for impurity profiling and quality control.
Icatibant, a selective bradykinin B2 receptor antagonist, is widely used in the treatment of hereditary angioedema (HAE). Given its peptide-based structure, Icatibant is prone to the formation of impurities during synthesis and storage.…
More Releases for Abacavir
Abacavir Market is Booming Worldwide with GlaxoSmithKline, Viatris, Cipla
The latest study released on the Global Abacavir Market by HTF MI evaluates market size, trend, and forecast to 2031. The Abacavir market study covers significant research data and proofs to be a handy resource document for managers, analysts, industry experts and other key people to have ready-to-access and self-analyzed study to help understand market trends, growth drivers, opportunities and upcoming challenges and about the competitors.
Key Players in This Report…
Abacavir Market Size, Share, Growth, Analysis, Key Players, Revenue, | Valuates …
Abacavir Market Size
The global market for Abacavir was estimated to be worth US$ 688.2 million in 2023 and is forecast to a readjusted size of US$ 1194.5 million by 2030 with a CAGR of 8.2% during the forecast period 2024-2030
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-11N8047/Global_Abacavir_Market_Insights_and_Forecast_to_2028
Abacavir Market Trends
The Abacavir market size is experiencing steady growth due to the increasing prevalence of HIV/AIDS and the rising demand for antiretroviral therapies. Abacavir, a key nucleoside reverse…
Abacavir Market Size, Trends, Growth Analysis, and Forecast | Valuates Reports
Abacavir Market Size
The global market for Abacavir was estimated to be worth US$ 688.2 million in 2023 and is forecast to a readjusted size of US$ 1194.5 million by 2030 with a CAGR of 8.2% during the forecast period 2024-2030.
Get Free Sample: https://reports.valuates.com/request/sample/QYRE-Auto-11N8047/Global_Abacavir_Market_Insights_and_Forecast_to_2028
Major Trends
The global Abacavir market is driven by the increasing prevalence of HIV/AIDS, which necessitates effective antiretroviral therapies (ART) for managing the condition. Abacavir, a nucleoside reverse transcriptase…
Global Abacavir Market Current Trends Opportunities And Challenges by 2027-GSK, …
The global Abacavir market is carefully researched in the report while largely concentrating on top players and their business tactics, geographical expansion, market segments, competitive landscape, manufacturing, and pricing and cost structures. Each section of the research study is specially prepared to explore key aspects of the global Abacavir market. For instance, the market dynamics section digs deep into the drivers, restraints, trends, and opportunities of the global Abacavir Market.…
Abacavir Market is Thriving Worldwide with Top Companies like: Aurobindo, Novnau …
Los Angeles, United State, –The report titled Global Abacavir Market is one of the most comprehensive and important additions to QY Research’s archive of market research studies. It offers detailed research and analysis of key aspects of the global Abacavir market. The market analysts authoring this report have provided in-depth information on leading growth drivers, restraints, challenges, trends, and opportunities to offer a complete analysis of the global Abacavir market.…
Augmentation of Abacavir Market by 2025 | Growth, Size, Share and Types
This report studies the global Abacavir market status and forecast, categorizes the global Abacavir market size (value & volume) by key players, type, application, and region. This report focuses on the top players in North America, Europe, Asia-Pacific, South America, and Middle East & Africa.
Ask for Sample copy of this report at https://www.inforgrowth.com/samplerequest/global-abacavir-market-research-report-2018.
The following manufacturers are covered in this report: GSK, Aurobindo, Novnauki, Smilax, Hycultec, Atripla.
Abacavir Breakdown Data by…